 Back
BackAtoms and Elements: Foundations of Modern Chemistry
Study Guide - Smart Notes

Atoms and Elements
Imaging and Moving Individual Atoms
Atoms are the fundamental building blocks of matter, connecting the macroscopic and microscopic worlds. An atom is the smallest identifiable unit of an element, and there are about 91 naturally occurring elements, with over 20 synthetic elements created by scientists.
Early Ideas About the Building Blocks of Matter
Historical Perspectives
Leucippus and Democritus first proposed that matter was composed of small, indestructible particles called atoms. They suggested that different kinds of atoms existed, each with unique shapes and sizes, moving randomly through empty space. Plato and Aristotle, however, believed matter had no smallest parts and was composed of fire, air, earth, and water. The scientific approach later prevailed, with John Dalton providing evidence for the atomic theory.
Modern Atomic Theory and the Laws That Led to It
Key Laws
Law of Conservation of Mass: Matter is neither created nor destroyed in a chemical reaction. The total mass of substances remains unchanged.
Law of Definite Proportions: All samples of a given compound have the same proportions of their constituent elements.
Law of Multiple Proportions: When two elements form different compounds, the masses of one element that combine with 1 gram of the other can be expressed as a ratio of small whole numbers.
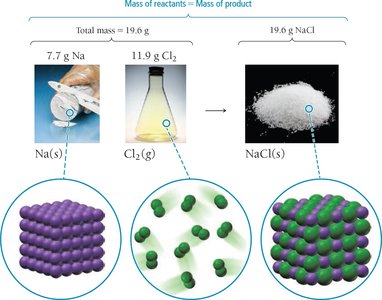
Example: Law of Definite Proportions
Two samples of carbon dioxide decomposed yield the same oxygen-to-carbon mass ratio, demonstrating the law of definite proportions:
Sample 1:
Sample 2:

Example: Law of Multiple Proportions
Carbon dioxide and carbon monoxide show different mass ratios of oxygen to carbon:
CO2: 2.67 g O per 1 g C
CO: 1.33 g O per 1 g C
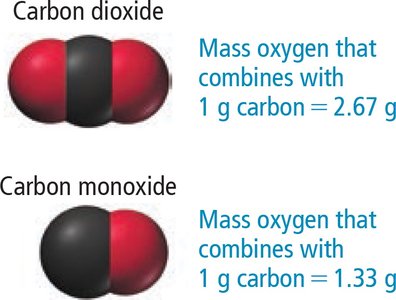
Atomic Theory and Subatomic Particles
Dalton’s Atomic Theory
Each element is composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties.
Atoms combine in simple, whole-number ratios to form compounds.
Atoms of one element cannot change into atoms of another element.
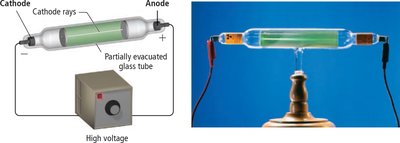
The Discovery of the Electron
J. J. Thomson’s cathode ray experiments led to the discovery of the electron, a negatively charged, low-mass particle present in all atoms.
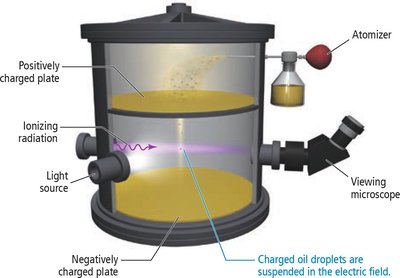
Millikan’s Oil Drop Experiment
Robert Millikan determined the charge of a single electron using the oil drop experiment, finding it to be C.
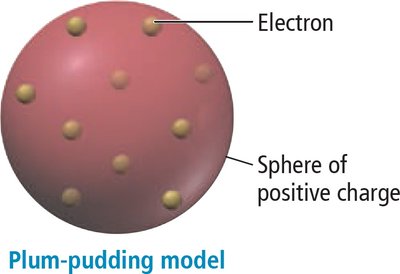
Plum-Pudding Model
Thomson proposed that electrons were embedded in a positively charged sphere, known as the plum-pudding model.
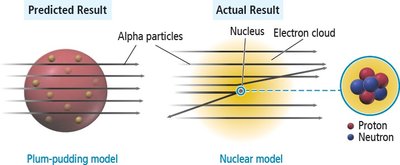
Rutherford’s Gold Foil Experiment
Ernest Rutherford’s experiment showed that most of the atom is empty space, with a dense nucleus containing protons. This led to the nuclear model of the atom.
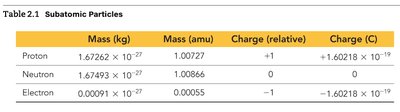
Subatomic Particles
Atoms are composed of protons, neutrons, and electrons. Protons and neutrons have nearly identical masses, while electrons are much lighter. The charge of the proton and electron are equal in magnitude but opposite in sign; neutrons have no charge.
Particle | Mass (kg) | Mass (amu) | Charge (relative) | Charge (C) |
|---|---|---|---|---|
Proton | 1.67262 × 10-27 | 1.00727 | +1 | +1.60218 × 10-19 |
Neutron | 1.67493 × 10-27 | 1.00866 | 0 | 0 |
Electron | 0.00091 × 10-27 | 0.00055 | -1 | -1.60218 × 10-19 |
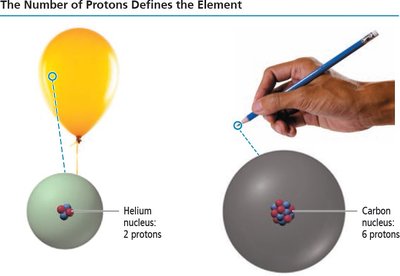
Elements and the Periodic Table
Defining Elements by Number of Protons
The number of protons in an atom’s nucleus (atomic number, Z) defines the element. Each element has a unique atomic number and chemical symbol.
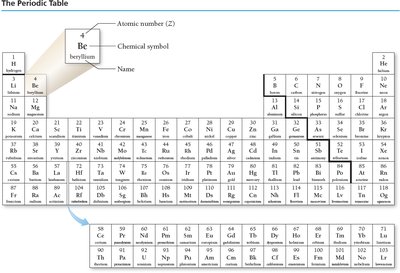
The Periodic Table
The periodic table organizes elements by increasing atomic number. Each element is represented by its atomic number, chemical symbol, and name.
Isotopes and Atomic Mass
Isotopes
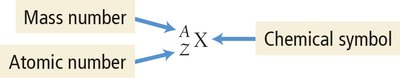
Isotopes are atoms of the same element with different numbers of neutrons. The mass number (A) is the sum of protons and neutrons. Isotopes are represented as or as X-A.
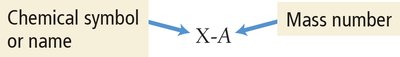
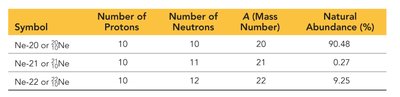
Symbol | Number of Protons | Number of Neutrons | Mass Number (A) | Natural Abundance (%) |
|---|---|---|---|---|
Ne-20 | 10 | 10 | 20 | 90.48 |
Ne-21 | 10 | 11 | 21 | 0.27 |
Ne-22 | 10 | 12 | 22 | 9.25 |
Atomic Mass
Atomic mass is the weighted average mass of an element’s isotopes, based on their natural abundance. It is listed beneath the element’s symbol in the periodic table.
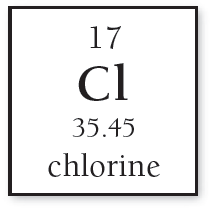
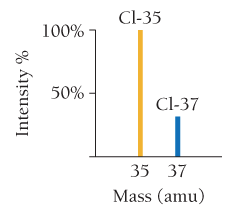
For example, chlorine’s atomic mass is calculated as:
Cl-35: amu
Cl-37: amu
Atomic mass Cl = amu
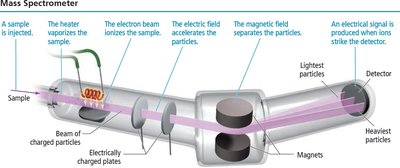
Mass Spectrometry
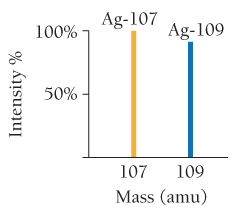
Mass spectrometry is used to determine the masses and abundances of isotopes, producing characteristic patterns for each element.
The Periodic Law and Classification of Elements
The Periodic Law
When elements are arranged in order of increasing mass, certain sets of properties recur periodically. Mendeleev organized elements so that those with similar properties fall in the same columns.
Classification of Elements
Elements are classified as metals, nonmetals, or metalloids. Metals are good conductors, malleable, ductile, and tend to lose electrons. Nonmetals are poor conductors, not malleable or ductile, and tend to gain electrons. Metalloids have mixed properties and are often semiconductors.
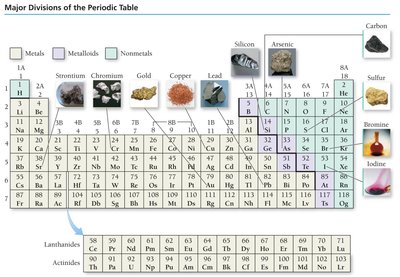
Periodic Table Groups and Periods
The periodic table is divided into vertical columns (groups) and horizontal rows (periods). Main-group elements have predictable properties, while transition elements are less predictable.
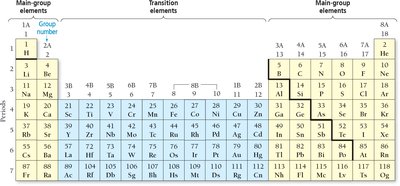
Special Groups
Noble Gases (Group 8A): Mostly unreactive, e.g., helium, neon, argon.
Alkali Metals (Group 1A): Highly reactive metals, e.g., sodium, lithium.
Alkaline Earth Metals (Group 2A): Fairly reactive, e.g., calcium, magnesium.
Halogens (Group 7A): Very reactive nonmetals, e.g., fluorine, chlorine.
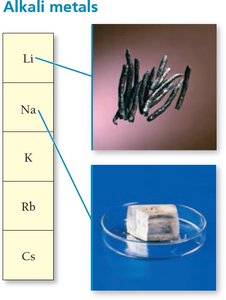

Ions and the Periodic Table
Formation of Ions
Atoms can lose or gain electrons to become ions. Cations are positively charged (e.g., Na+), and anions are negatively charged (e.g., F-). Main-group metals tend to lose electrons, forming cations; main-group nonmetals tend to gain electrons, forming anions.
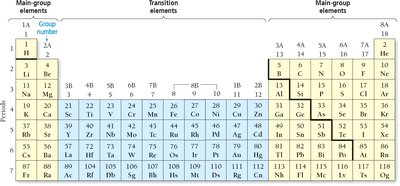
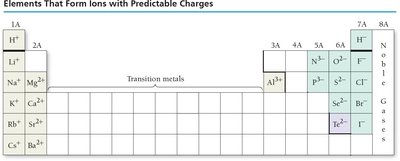
Predicting Ion Charges
Alkali metals (1A): Lose one electron, form 1+ ions.
Alkaline earth metals (2A): Lose two electrons, form 2+ ions.
Halogens (7A): Gain one electron, form 1- ions.
Oxygen family (6A): Gain two electrons, form 2- ions.
Molar Mass and Counting Atoms
The Mole Concept
The mole (mol) is a unit for counting atoms, defined as particles (Avogadro’s number). The mass of 1 mole of atoms (molar mass) in grams is numerically equal to the atomic mass in amu.
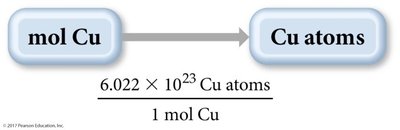
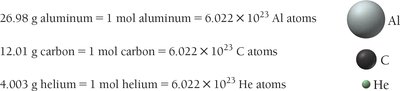
Converting Between Mass, Moles, and Number of Atoms
To count atoms by weighing, use the molar mass as a conversion factor between mass and moles, and Avogadro’s number to convert between moles and atoms.
Number of moles =
Number of atoms = number of moles
Example Calculation
Calculate the number of copper atoms in 2.45 mol of copper:
atoms
Additional info: These notes cover the foundational concepts of atomic theory, laws of chemical combination, subatomic particles, isotopes, atomic mass, periodic table structure, ion formation, and the mole concept, all essential for general chemistry students.
