 Back
BackAtoms and Elements: Foundations of Modern Atomic Theory
Study Guide - Smart Notes

Atoms and Elements
Introduction to Atomic Theory
The study of atoms and elements forms the foundation of modern chemistry. This chapter explores the historical development of atomic theory, the structure of the atom, and the organization of elements in the periodic table.
The Laws of Chemical Combination
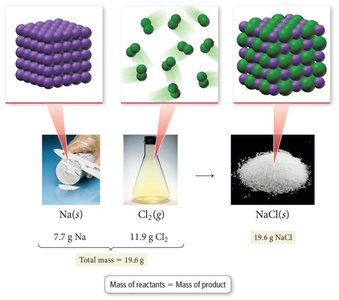
Law of Conservation of Mass
The law of conservation of mass, formulated by Antoine Lavoisier, states that matter is neither created nor destroyed in a chemical reaction. The total mass of reactants equals the total mass of products.
Key Point: Mass remains constant during chemical changes.
Example: When sodium reacts with chlorine to form sodium chloride, the combined mass of sodium and chlorine equals the mass of sodium chloride produced.
Law of Definite Proportions
Joseph Proust's law of definite proportions (or law of constant composition) states that all samples of a given compound have the same proportions of their constituent elements, regardless of source or preparation method.
Key Point: The composition of a compound is always fixed.
Example: Water (H2O) always decomposes into hydrogen and oxygen in a mass ratio of 2:16 (or 1:8).
Law of Multiple Proportions
John Dalton's law of multiple proportions states that when two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in ratios of small whole numbers.
Key Point: Elements combine in simple, whole-number ratios to form compounds.
Development of Atomic Theory
Dalton's Atomic Theory
John Dalton formalized atomic theory in 1808, proposing that:
Each element is composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties.
Atoms combine in simple, whole-number ratios to form compounds.
Atoms cannot be subdivided, created, or destroyed in chemical reactions.
In chemical reactions, atoms are combined, separated, or rearranged.
Discovery of Subatomic Particles
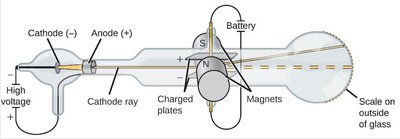
Thomson's Cathode Ray Experiment
J. J. Thomson discovered the electron using cathode ray tubes, showing that atoms contain smaller, negatively charged particles.
Electrons are much lighter than atoms and are present in all substances.
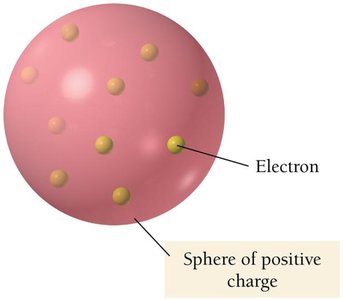
Thomson proposed the "plum pudding model," where electrons are embedded in a sphere of positive charge.


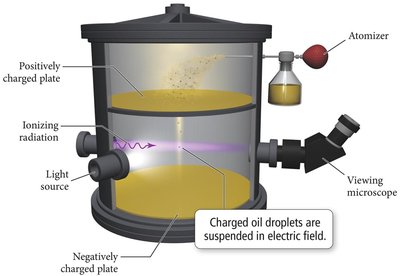
Millikan's Oil Drop Experiment
Robert Millikan measured the charge of the electron using the oil drop experiment, confirming the quantized nature of electric charge.
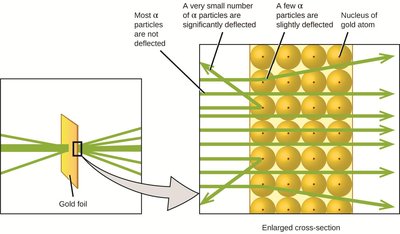
Rutherford's Nuclear Model
Ernest Rutherford's gold foil experiment demonstrated that atoms have a small, dense, positively charged nucleus, with electrons occupying the surrounding space.
Most of the atom's mass and all positive charge are in the nucleus.
Electrons move through the empty space around the nucleus.
The number of protons equals the number of electrons in a neutral atom.
Discovery of the Neutron
James Chadwick confirmed the existence of the neutron, a neutral particle in the nucleus with a mass similar to the proton.
Structure of the Atom
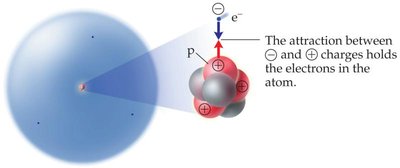
Protons: Positively charged, found in the nucleus.
Neutrons: Neutral, found in the nucleus.
Electrons: Negatively charged, found outside the nucleus.
Atomic Number and Mass Number
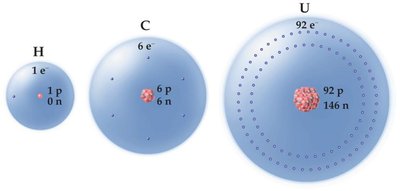
Atomic Number (Z): Number of protons in the nucleus; defines the element.
Mass Number (A): Total number of protons and neutrons in the nucleus.
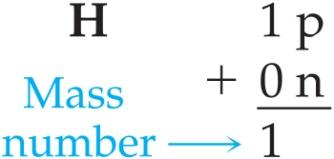
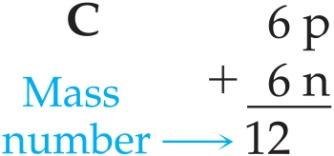
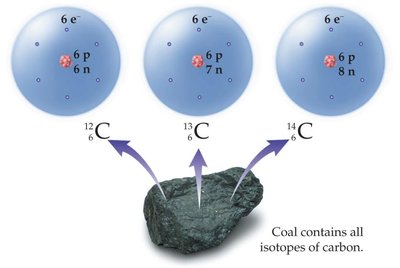
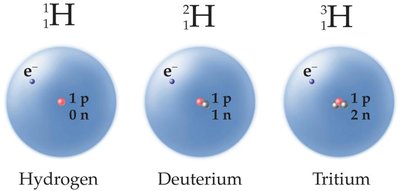
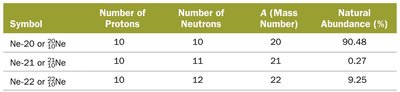
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons (and thus different mass numbers).
Isotopes are represented as Element-Mass Number (e.g., C-12, C-13, C-14).
Natural elements often exist as mixtures of isotopes with different natural abundances.
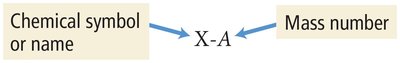
Ions
Formation of Ions
Atoms can gain or lose electrons to form ions:
Cations: Positively charged ions (loss of electrons).
Anions: Negatively charged ions (gain of electrons).
The charge of an ion is given by:

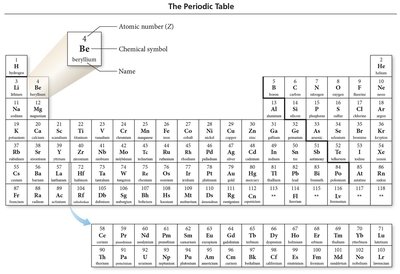
The Periodic Table
Organization and Classification
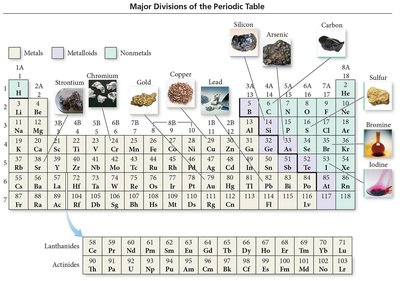
The periodic table arranges elements by increasing atomic number, grouping elements with similar properties into columns (groups or families).
Metals: Good conductors, malleable, ductile, shiny, tend to lose electrons.
Nonmetals: Poor conductors, varied states, tend to gain electrons.
Metalloids (Semiconductors): Intermediate properties, useful in electronics.
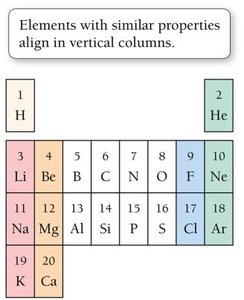
Groups of Elements
Alkali Metals (Group 1A): Highly reactive metals (e.g., Na, K).
Alkaline Earth Metals (Group 2A): Fairly reactive metals (e.g., Mg, Ca).
Halogens (Group 7A): Very reactive nonmetals (e.g., F, Cl).
Noble Gases (Group 8A): Unreactive, stable gases (e.g., He, Ne).
Atomic Mass and the Mole
Atomic Mass
The atomic mass of an element is the weighted average of the masses of its naturally occurring isotopes.
Example: Chlorine has two main isotopes, Cl-35 and Cl-37. Its atomic mass reflects their relative abundances.
The Mole and Avogadro's Number
The mole (mol) is the SI unit for amount of substance, defined as containing exactly particles (Avogadro's number).
1 mol of atoms = atoms.
The mass of 1 mol of an element (in grams) equals its atomic mass (in amu).
Converting Between Mass, Moles, and Number of Atoms
To convert between mass and moles, use the molar mass as a conversion factor.
To convert between moles and number of atoms, use Avogadro's number.
Example: Calculate the number of copper atoms in 2.45 mol of copper.
Example: Calculate the amount of carbon (in moles) in 0.0265 g of graphite.
