 Back
BackAtoms and Elements: Laws, Atomic Theory, and Atomic Structure
Study Guide - Smart Notes

Atoms and Elements
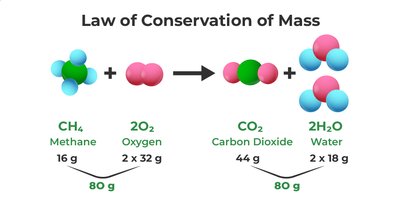
Law of Conservation of Mass
The Law of Conservation of Mass states that in a chemical reaction, matter is neither created nor destroyed. The total mass of reactants equals the total mass of products. This principle is fundamental to all chemical reactions and is used to balance chemical equations.
Key Point: The mass of the substances before a reaction is equal to the mass after the reaction.
Example: In the reaction of methane (CH4) and oxygen (O2) to produce carbon dioxide (CO2) and water (H2O), the total mass of reactants (80 g) equals the total mass of products (80 g).
Law of Definite Proportions
The Law of Definite Proportions (Joseph Proust) states that all samples of a given compound, regardless of their source or preparation, have the same proportions by mass of their constituent elements.
Key Point: The ratio of elements in a compound is always constant.
Example: Sodium chloride (NaCl) always contains 39.3% sodium and 60.7% chlorine by mass, regardless of sample size.
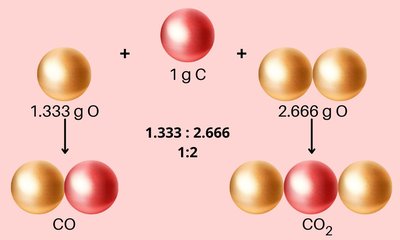
Law of Multiple Proportions
The Law of Multiple Proportions (John Dalton) states that when two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in ratios of small whole numbers.
Key Point: This law helps distinguish different compounds formed by the same elements.
Example: Carbon and oxygen form CO and CO2. For 1 g of carbon, CO contains 1.333 g of oxygen, and CO2 contains 2.666 g of oxygen. The ratio 1.333:2.666 simplifies to 1:2.
Atomic Theory
John Dalton's Atomic Theory laid the foundation for modern chemistry:
Each element is composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties.
Atoms combine in simple whole-number ratios to form compounds.
Atoms of one element cannot change into atoms of another element in chemical reactions; they only rearrange.
Discovery of Subatomic Particles
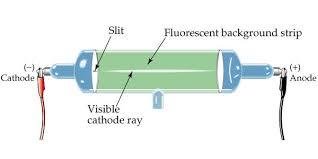
Electrons: Cathode Ray Experiments
J.J. Thomson discovered the electron using cathode ray tubes. He found that cathode rays are made of negatively charged particles much lighter than hydrogen atoms.
Key Point: Electrons are fundamental, negatively charged subatomic particles present in all atoms.
Charge-to-mass ratio: C/g
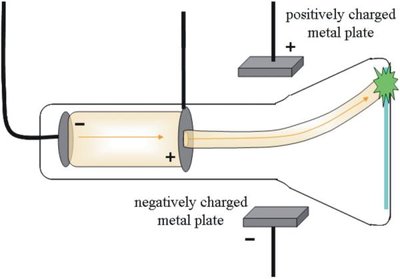
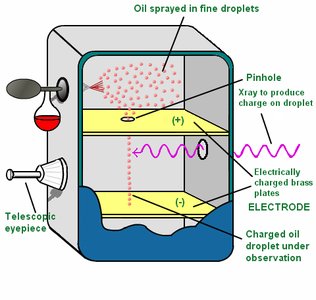
Millikan Oil Drop Experiment
Robert Millikan determined the charge and mass of the electron using the oil drop experiment.
Electron charge: C
Electron mass: g
Discovery of the Nucleus: Gold Foil Experiment
Ernest Rutherford's gold foil experiment showed that atoms have a small, dense, positively charged nucleus. Most of the atom's volume is empty space, with electrons dispersed throughout.
Key Point: The nucleus contains most of the atom's mass and all its positive charge.


Structure of the Atom
Atoms consist of three main subatomic particles:
Protons: Positively charged, located in the nucleus, define the atomic number (Z).
Neutrons: Neutral, located in the nucleus, contribute to atomic mass.
Electrons: Negatively charged, orbit the nucleus.

Isotopes
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers (A).
Isotope notation: or X-A, where X is the element symbol, Z is the atomic number, and A is the mass number.
Example: Hydrogen has three isotopes: protium (H), deuterium (H), and tritium (H).

Atomic Mass and Natural Abundance
The atomic mass of an element is the weighted average of the masses of its naturally occurring isotopes, based on their relative abundances.
Formula:
Example: Neon has three isotopes: Ne, Ne, and Ne. Their atomic mass is calculated using their masses and percent abundances.

The Mole and Molar Mass
The mole is a counting unit in chemistry, defined as the amount of substance containing particles (Avogadro's number). The molar mass of an element (g/mol) is numerically equal to its atomic mass in unified atomic mass units (u).
Key Point: 1 mole of carbon-12 atoms has a mass of exactly 12 grams.
Conversions:
From mass to moles:
From moles to mass:
From moles to atoms:
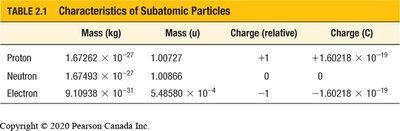
Summary Table: Characteristics of Subatomic Particles
Particle | Mass (kg) | Mass (u) | Charge (relative) | Charge (C) |
|---|---|---|---|---|
Proton | 1.67262 × 10−27 | 1.00727 | +1 | +1.60218 × 10−19 |
Neutron | 1.67493 × 10−27 | 1.00866 | 0 | 0 |
Electron | 9.10938 × 10−31 | 5.48580 × 10−4 | −1 | −1.60218 × 10−19 |

Practice Problems and Applications
Example: Calculate the number of atoms in 95.8 g of Fe (Molar mass = 55.85 g/mol): mol atoms
Example: Calculate the mass of Ag in 0.342 mol Ag (Molar mass = 107.87 g/mol): g
Summary
This chapter covers the foundational laws of chemistry, the structure and properties of atoms, the concept of isotopes, atomic mass, and the mole. Understanding these principles is essential for further study in general chemistry.
