 Back
BackAtoms and Elements: Structure, Properties, and the Periodic Table
Study Guide - Smart Notes

Atoms and Elements
Elements and Their Symbols
Elements are pure substances that serve as the fundamental building blocks of all matter. Each element is represented by a unique chemical symbol, which is a one- or two-letter abbreviation derived from its name. Learning the names and symbols of elements is essential for understanding chemical reactions and properties.
Element Names: Often named after planets, mythological figures, minerals, geographic locations, or famous people.
Chemical Symbols: The first letter is always capitalized; the second letter (if present) is lowercase. For example, Co is cobalt, while CO represents carbon and oxygen.
Examples: Aluminum (Al), Silver (Ag), Gold (Au), Sulfur (S).




Table: Names and Symbols of Common Elements
Name of Element | Element Symbol | Name of Element | Element Symbol |
|---|---|---|---|
Aluminum | Al | Hydrogen | H |
Barium | Ba | Iodine | I |
Carbon | C | Nitrogen | N |
Calcium | Ca | Oxygen | O |
Chlorine | Cl | Phosphorus | P |
Table: Some Elements, Symbols, and Source of Names
Element | Symbol | Source of Name |
|---|---|---|
Uranium | U | The planet Uranus |
Titanium | Ti | Titans (mythology) |
Chlorine | Cl | Chloros: "greenish yellow" (Greek) |
Iodine | I | Ioeides: "violet" (Greek) |
Magnesium | Mg | Magnesia, a mineral |
Californium | Cf | California |
Curium | Cm | Marie and Pierre Curie |
Copernicium | Cn | Nicolaus Copernicus |
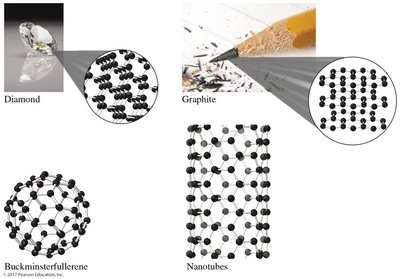
Allotropes of Carbon
Carbon atoms can arrange themselves in various structures, resulting in different substances with unique properties. These arrangements are called allotropes.
Diamond: Carbon atoms are arranged in a rigid, three-dimensional structure, making diamonds transparent and extremely hard.
Graphite: Carbon atoms are connected in planes of hexagonal rings, used in lubricants and pencils.
Buckminsterfullerene: Spherical arrangement of carbon atoms, also known as "buckyballs."
Nanotubes: Cylindrical arrangement of carbon atoms, important in nanotechnology.
The Periodic Table
Structure and Organization
The periodic table is a systematic arrangement of elements based on their atomic number and properties. Created by Dmitri Mendeleev in 1872, it organizes elements into groups (vertical columns) and periods (horizontal rows).
Groups: Elements with similar properties are placed in the same group.
Periods: Horizontal rows; period 1 contains two elements (H and He), periods 2 and 3 contain eight elements each.
Group Numbers: Written as 1A–8A for representative elements and 1–18 for all columns.
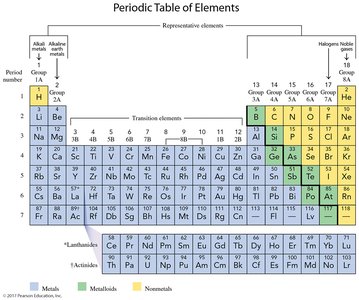
Representative Elements and Group Names
Representative elements are found in groups 1A–8A and have common names:
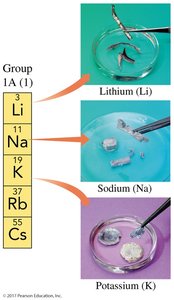
Group 1A (1): Alkali metals (Li, Na, K, Rb, Cs, Fr)
Group 2A (2): Alkaline earth metals (Be, Mg, Ca, Sr, Ba, Ra)
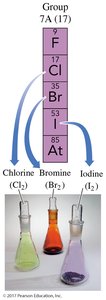
Group 7A (17): Halogens (F, Cl, Br, I, At)
Group 8A (18): Noble gases (He, Ne, Ar, Kr, Xe, Rn)
Metals, Nonmetals, and Metalloids
The periodic table is divided by a zigzag line that separates metals from nonmetals. Metalloids border this line and exhibit properties of both metals and nonmetals.
Metals: Shiny, ductile, malleable, good conductors, usually solid at room temperature (except mercury).
Nonmetals: Not shiny, poor conductors, low melting points and densities.
Metalloids: Intermediate properties, semiconductors, include B, Si, Ge, As, Sb, Te, Po, At.
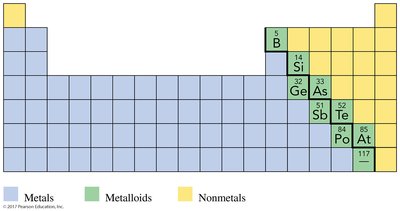
Table: Characteristics of a Metal, Metalloid, and Nonmetal
Silver (Ag) | Antimony (Sb) | Sulfur (S) |
|---|---|---|
Metal | Metalloid | Nonmetal |
Shiny | Blue-gray, shiny | Dull, yellow |
Extremely ductile | Brittle | Brittle |
Can be hammered into sheets (malleable) | Shatters when hammered | Shatters when hammered |
Good conductor of heat and electricity | Poor conductor of heat and electricity | Poor conductor of heat and electricity, good insulator |
Used in coins, jewelry, tableware | Used to harden lead, color glass and plastics | Used in gunpowder, rubber, fungicides |
Density 10.5 g/mL | Density 6.7 g/mL | Density 2.1 g/mL |
Melting point 962°C | Melting point 630°C | Melting point 113°C |

Atomic Structure
Dalton's Atomic Theory
John Dalton proposed the atomic theory in 1808, which forms the foundation of modern chemistry:
All matter is made up of tiny particles called atoms.
All atoms of a given element are identical and different from atoms of other elements.
Atoms of different elements combine to form compounds, always in fixed ratios.
Chemical reactions involve rearrangement, separation, or combination of atoms; atoms are not created or destroyed.
Subatomic Particles
Atoms are composed of three main subatomic particles:
Protons: Positive charge, located in the nucleus.
Neutrons: No charge, located in the nucleus.
Electrons: Negative charge, located outside the nucleus.
Atoms are electrically neutral, having equal numbers of protons and electrons.
Atomic Number and Mass Number
The atomic number is the number of protons in an atom and defines the element. The mass number is the sum of protons and neutrons in the nucleus.
Atomic Number (Z): Appears above the element symbol in the periodic table.
Mass Number (A):
Number of Neutrons:
Isotopes and Atomic Mass
Isotopes
Isotopes are atoms of the same element with different mass numbers due to varying numbers of neutrons. The atomic symbol for an isotope shows the mass number and atomic number.
Example: Carbon has three naturally occurring isotopes: , , .
Calculating Atomic Mass
The atomic mass of an element is the weighted average of the masses of its naturally occurring isotopes, based on their percent abundance.
Formula:
Example (Magnesium):
:
:
:
Atomic mass of Mg:
Example (Chlorine):
:
:
Atomic mass of Cl:
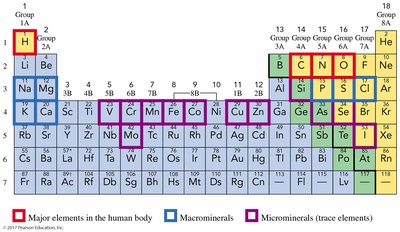
Elements Essential to Life
Major Elements in the Human Body
Only 20 elements are essential for human health. Four elements—oxygen, carbon, hydrogen, and nitrogen—make up 96% of body mass. Macrominerals such as calcium, phosphorus, potassium, chlorine, sulfur, sodium, and magnesium are vital for bone formation and bodily functions.
Summary Concept Map
The relationships between atoms, elements, isotopes, and the periodic table can be visualized in a concept map, showing how these fundamental ideas connect in chemistry.
*Additional info: The concept map (not shown here) would typically illustrate the connections between atomic structure, element classification, isotopes, and their roles in chemical reactions and biological systems.*
