 Back
BackAtoms, Elements, and the Periodic Table: Structure and Properties
Study Guide - Smart Notes

Atoms & Elements
Structure of the Atom
The atom is the fundamental unit of matter, composed of a nucleus containing protons and neutrons, surrounded by an electron cloud. The arrangement and behavior of these subatomic particles determine the chemical properties of each element.
Protons: Positively charged particles found in the nucleus; define the atomic number.
Neutrons: Neutral particles found in the nucleus; contribute to atomic mass.
Electrons: Negatively charged particles found in the electron cloud; occupy specific energy levels.
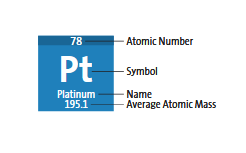
Atomic Number (Z): Number of protons in the nucleus; unique for each element.
Mass Number (A): Total number of protons and neutrons in the nucleus.
Isotopes: Atoms of the same element with different numbers of neutrons; same atomic number, different mass number.


Example: Chlorine has two common isotopes: chlorine-35 and chlorine-37.
Periodic Table
Organization and Classification
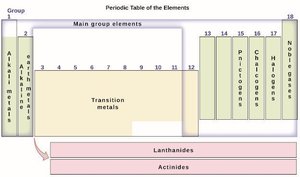
The periodic table arranges elements by increasing atomic number and groups them based on similar chemical properties. It is divided into periods (rows) and groups (columns).
Periods: Horizontal rows; indicate repeating patterns in properties.
Groups: Vertical columns; elements in the same group have similar chemical behaviors.
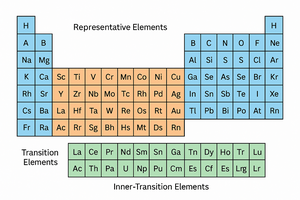
Main Group Elements: Groups 1, 2, and 13–18; include metals, nonmetals, and metalloids.
Transition Elements: Groups 3–12; all are metals.
Inner-Transition Elements: Lanthanides and actinides; many are radioactive or rare earth metals.
Types of Elements
Metals: Shiny, malleable, good conductors of heat and electricity.
Nonmetals: Dull, poor conductors of heat and electricity.
Metalloids: Possess properties of both metals and nonmetals.
Subatomic Particles & Atomic Properties
Atomic Number and Mass Number
The atomic number identifies the element, while the mass number distinguishes isotopes.
Atomic Number (Z): Number of protons; determines element identity.
Mass Number (A): Number of protons plus neutrons; unique to each isotope.
Neutrons: Calculated as mass number minus atomic number.
Atomic Weight
Atomic weight is the weighted average of all naturally occurring isotopes of an element, reflecting their relative abundance.
Example: Lithium's atomic weight is 6.94 amu, due to the higher abundance of lithium-7 compared to lithium-6.
Bohr Model and Quantum Theory
Bohr Model of the Atom
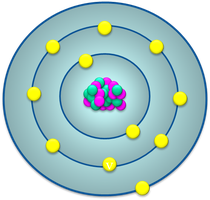
The Bohr model describes electrons as orbiting the nucleus in fixed energy levels or shells. These energy levels are quantized, meaning electrons can only occupy specific energy states.
Energy Levels: Concentric layers around the nucleus; the closest layer has the lowest energy.
Valence Electrons: Electrons in the outermost shell; most chemical reactions involve these electrons.
Quantum Numbers and Orbitals
Quantum numbers describe the size, shape, and orientation of atomic orbitals.
Principal Quantum Number (n): Specifies the energy level and size of the orbital; n = 1, 2, 3, ...
Angular Momentum Quantum Number (l): Specifies the shape; l = 0 (s), 1 (p), 2 (d), 3 (f).
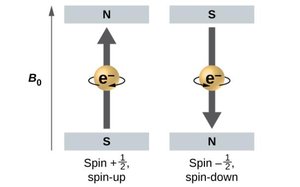
Magnetic Quantum Number (ml): Specifies the orientation; ml ranges from -l to +l.
Spin Quantum Number (ms): Specifies electron spin; ms = +½ or –½.
Orbital Shapes and Subshells
s orbital: Spherical shape
p orbitals: Dumbbell shapes
d orbitals: Cloverleaf shapes
f orbitals: Complex shapes
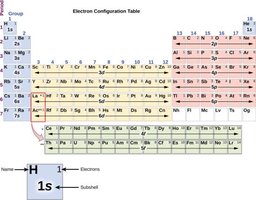
Electron Configurations
Writing Electron Configurations
Electron configurations show the arrangement of electrons in an atom's orbitals, using numbers and letters to indicate energy levels and subshells.
Format: Principal quantum number (n), subshell letter (l), superscript for number of electrons.
Example: Sodium: 1s22s22p63s1

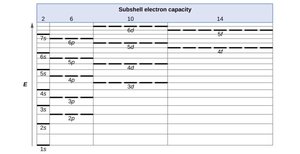
Orbital Filling Order
Electrons fill orbitals in order of increasing energy, following the Aufbau principle. Subshell energies can overlap, such as 4s being lower than 3d.
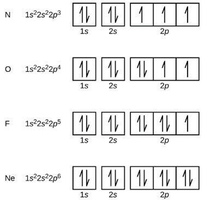
Orbital Diagrams
Orbital diagrams visually represent electron configurations, showing individual orbitals as boxes and electrons as arrows. Hund's Rule states that electrons fill each orbital singly before pairing.
Valence Electrons and Ions
Valence Electrons
Valence electrons are those in the highest energy level and are responsible for chemical reactivity. Their configuration explains similarities in chemical behavior among elements in the same group.
Representative Elements: Last electron enters s or p orbital.
Transition Metals: Last electron enters d orbital.
Inner Transition Metals: Last electron enters f orbital.
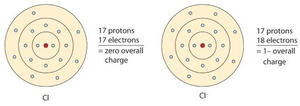
Ions
Ions are formed when atoms gain or lose electrons to achieve a full valence shell, resulting in a positive (cation) or negative (anion) charge.
Cation: Positive charge; electrons lost.
Anion: Negative charge; electrons gained.
Periodic Properties
Atomic and Ionic Radius
Atomic radius is the distance from the nucleus to the valence electrons. It decreases across a period and increases down a group. Ionic radius follows similar trends, with cations being smaller and anions larger than their neutral atoms.

Ionization Energy
Ionization energy is the minimum energy required to remove a valence electron from an atom in the gaseous state. It increases across a period and decreases down a group.
Equation:
Trend: Higher for nonmetals, lower for metals.

Summary Table: Classification of Elements
Type | Groups | Properties |
|---|---|---|
Representative Elements | 1, 2, 13–18 | Include metals, nonmetals, metalloids; main group chemistry |
Transition Elements | 3–12 | All metals; variable oxidation states |
Inner-Transition Elements | Lanthanides, Actinides | Many are radioactive; rare earth metals |
Additional info: This summary covers foundational concepts in atomic structure, periodic table organization, quantum theory, electron configurations, and periodic properties, suitable for exam preparation in general chemistry.
