 Back
BackAtoms, Elements, and the Structure of Matter: CHEM131 Study Guide
Study Guide - Smart Notes

Atoms & Elements
Discovery of Subatomic Particles
The structure of the atom was revealed through a series of key experiments in the late 19th and early 20th centuries. These discoveries laid the foundation for modern chemistry by identifying the electron, proton, and neutron.
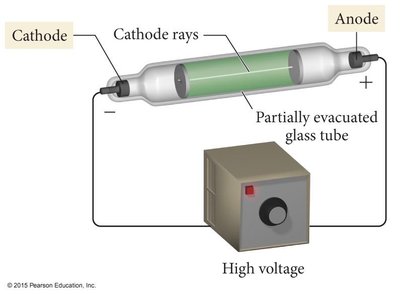
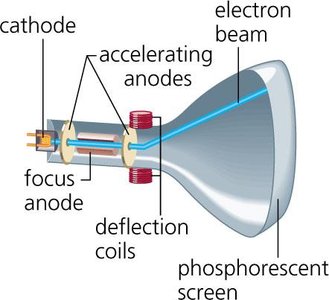
Cathode Ray Experiment (1897): J.J. Thomson discovered the electron and its charge-to-mass ratio using cathode rays. Electrons are much smaller than hydrogen atoms.
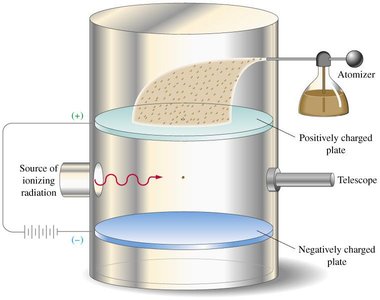
Oil Drop Experiment (1909): Robert Millikan determined the charge of a single electron and its mass, confirming electrons are smaller than hydrogen atoms.
Plum Pudding Model (1904): Thomson proposed that atoms are composed of electrons embedded in a positively charged sphere.
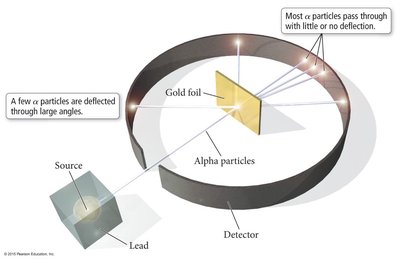
Gold Foil Experiment (1909): Ernest Rutherford discovered the nucleus, showing that most of the atom's mass is concentrated in a dense center, with most of the volume being empty space.

Components of the Atom
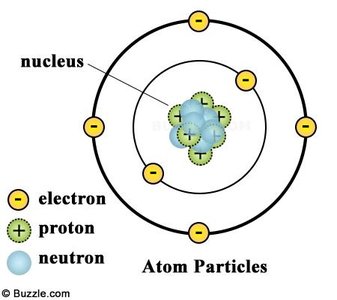
Atoms are composed of three fundamental subatomic particles: protons, neutrons, and electrons. Their arrangement determines the chemical properties of elements.
Proton: Positively charged particle found in the nucleus.
Neutron: Neutral particle found in the nucleus.
Electron: Negatively charged particle found outside the nucleus.
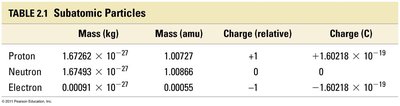
Particle | Mass (kg) | Mass (amu) | Charge (relative) | Charge (C) |
|---|---|---|---|---|
Proton | 1.67262 × 10-27 | 1.00727 | +1 | +1.60218 × 10-19 |
Neutron | 1.67493 × 10-27 | 1.00866 | 0 | 0 |
Electron | 0.00091 × 10-27 | 0.00055 | -1 | -1.60218 × 10-19 |
Matter, Measurement & Problem Solving
Types of Matter
Matter is classified based on its composition and physical state. Atoms and molecules are the basic building blocks.
Atoms: Smallest unit of an element, composed of protons, neutrons, and electrons. Examples: hydrogen (H), oxygen (O), gold (Au).
Molecules: Groups of two or more atoms bonded together. Examples: water (H2O), oxygen (O2), hydrogen (H2).


States of Matter
Matter exists in three primary states: solid, liquid, and gas. Each state is defined by the arrangement and movement of atoms or molecules.
Solid: Atoms or molecules are closely packed in fixed locations, vibrate but do not move past each other. Solids have fixed volume and shape. Examples: ice, aluminum, diamond.
Liquid: Atoms or molecules are close but can move relative to each other. Liquids have fixed volume but not fixed shape, and flow to fill containers. Examples: water, alcohol, gasoline.
Gas: Atoms or molecules have much space between them and move freely. Gases are compressible and fill the shape of their container.


Atoms & Elements
Atomic Number, Mass Number, and Isotopes
Each element is defined by its atomic number (number of protons). The mass number is the sum of protons and neutrons. Isotopes are atoms of the same element with different numbers of neutrons.
Atomic Number (Z): Number of protons in the nucleus.
Mass Number (A): Number of protons plus neutrons.
Isotopes: Atoms with the same number of protons but different numbers of neutrons, resulting in different masses.

Calculating Subatomic Particles
To determine the number of protons, neutrons, and electrons in an atom or ion:
Protons: Equal to atomic number.
Neutrons: Mass number minus atomic number.
Electrons: Equal to protons for neutral atoms; adjust for ions.
Isotopes and Weighted Average Mass
The atomic mass listed on the periodic table is a weighted average of all naturally occurring isotopes of an element.
Example: Chlorine has two main isotopes: 35Cl (75.77%, mass = 34.97 g/mol) and 37Cl (24.23%, mass = 36.97 g/mol).
Weighted Average Formula:

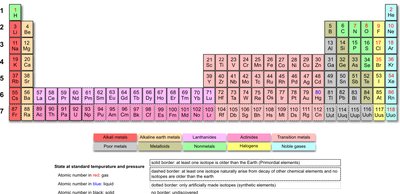
The Periodic Table
History and Structure
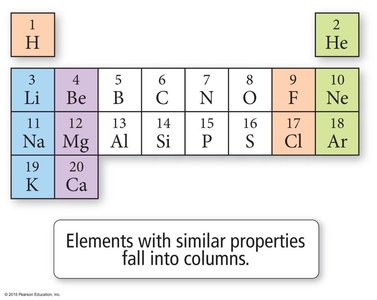
The periodic table organizes elements by increasing atomic number and groups elements with similar properties into columns. Dmitri Mendeleev created the first version in 1869.
Groups: Vertical columns with similar properties.
Periods: Horizontal rows.
Element Symbols: One- or two-letter abbreviations for each element.
Classification of Elements
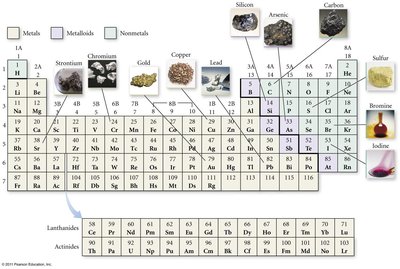
Elements are classified as metals, nonmetals, or metalloids based on their properties.
Metals: Conduct heat and electricity, malleable, ductile, shiny, tend to lose electrons and form cations. Located on the left side of the periodic table.
Alkali Metals (Group 1A): Highly reactive, one valence electron, form +1 cations.
Alkaline Earth Metals (Group 2A): Form alkaline solutions, two valence electrons, form +2 cations.
Metalloids: Mixed properties, semiconductors. Examples: B, Si, Ge, As, Sb, Te, At.
Nonmetals: Poor conductors, variable properties, tend to gain electrons and form anions, located on the right side.
Halogens (Group 7A): Reactive nonmetals, seven valence electrons, form -1 anions, exist as diatomic molecules.
Noble Gases (Group 8A): Inert, eight valence electrons, follow the octet rule.
Bonus: Mathematical Operations and Functions
Scientific Calculator Usage
A scientific calculator is essential for solving chemistry problems involving mathematical operations, such as calculating molar masses, concentrations, and performing conversions.
Example: TI-36X Pro is a recommended non-programmable calculator for exams and homework.

Bonus: Lab Techniques and Procedures
Understanding Experimental Apparatus
Familiarity with classic experimental setups, such as the cathode ray tube and oil drop apparatus, is important for understanding the discovery of atomic structure.
Cathode Ray Tube: Used to discover the electron.
Oil Drop Apparatus: Used to measure the charge of the electron.


Summary Table: Subatomic Particles
Particle | Location | Charge | Mass (amu) |
|---|---|---|---|
Proton | Nucleus | +1 | 1.00727 |
Neutron | Nucleus | 0 | 1.00866 |
Electron | Outside nucleus | -1 | 0.00055 |
Practice Problem Example
Weighted Average Calculation
Given two isotopes of bromine:
79Br: 78.9183371 amu
81Br: 80.9162906 amu
Let x = fraction of 79Br, (1-x) = fraction of 81Br. If the average atomic mass is known, set up the equation:
Solve for x to find percent natural abundance.
Tips for Success in General Chemistry
Practice problems regularly, not just memorization.
Use the textbook as a resource for explanations and examples.
Work through old exams and homework sets for exam preparation.
Attend office hours and guided study sessions for additional support.
