 Back
BackCHM113, ch2. Atoms, Molecules, and Ions: Foundations of Chemical Structure and Nomenclature
Study Guide - Smart Notes

Atoms, Molecules, and Ions
Introduction
This chapter introduces the fundamental concepts of atomic theory, the structure of atoms, the periodic table, and the basics of chemical nomenclature. Understanding these principles is essential for studying chemical reactions and properties of matter.
Laws of Chemical Combination
Law of Conservation of Mass
The Law of Conservation of Mass states that the total mass of substances present before a chemical reaction is equal to the total mass after the reaction. This principle, discovered by Antoine Lavoisier, is foundational to chemical equations and stoichiometry.
Key Point: Mass is neither created nor destroyed in a chemical reaction.
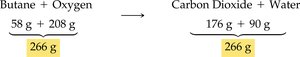
Example: The combustion of butane: 58 g butane + 208 g oxygen → 176 g carbon dioxide + 90 g water (total mass = 266 g before and after).
Law of Constant Composition
The Law of Constant Composition (Joseph Proust) states that a compound always contains the same proportion of elements by mass. For example, every molecule of carbon dioxide (CO2) contains two oxygen atoms for every carbon atom, with a mass ratio of 2.66:1 (O:C).
Key Point: The composition of a compound is always fixed.
Example: CO2 always has a mass ratio of O to C of 32:12.01 = 2.66.
Law of Multiple Proportions
The Law of Multiple Proportions (John Dalton) states that if two elements form more than one compound, the ratios of the masses of the second element that combine with a fixed mass of the first element are small whole numbers.
Key Point: Different compounds formed from the same elements have mass ratios that are simple multiples of each other.
Example: O:C in CO2 is 2.66, in CO is 1.33; 2.66/1.33 = 2/1.
Dalton’s Atomic Theory
Postulates of Dalton’s Atomic Theory
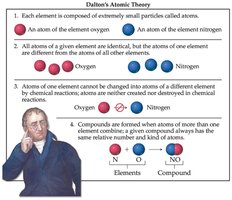
Dalton’s atomic theory provides the basis for understanding chemical reactions and the nature of matter:
Each element is composed of extremely small particles called atoms.
All atoms of a given element are identical in mass and properties, but differ from atoms of other elements.
Atoms are not created or destroyed in chemical reactions.
Atoms of different elements combine in simple whole-number ratios to form compounds.
Structure of the Atom
Subatomic Particles
Atoms are composed of three main subatomic particles:
Protons: Positively charged, mass ≈ 1 amu, located in the nucleus.
Neutrons: Neutral, mass ≈ 1 amu, located in the nucleus.
Electrons: Negatively charged, mass ≈ 1/1836 amu, move around the nucleus.
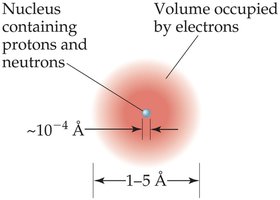
The Nuclear Atom
Ernest Rutherford’s experiments led to the nuclear model of the atom, where a dense, positively charged nucleus is surrounded by electrons.
Atoms are 1–5 Å (100–500 pm) in diameter; the nucleus is much smaller (~10–4 Å).
Atomic Number, Mass Number, and Isotopes
Atomic Number and Mass Number
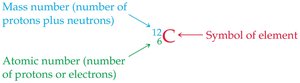
The atomic number (Z) is the number of protons in the nucleus and defines the element. The mass number (A) is the sum of protons and neutrons.
Atomic number is written as a subscript, mass number as a superscript before the element symbol.
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons, and thus different mass numbers.
Isotopes have identical chemical properties but different physical properties (e.g., mass).
Example: Carbon-12, Carbon-13, and Carbon-14 are isotopes of carbon.
Atomic Mass and Atomic Weight
Atomic Mass Unit (amu)
The atomic mass unit (amu) is defined as 1/12 the mass of a carbon-12 atom. 1 amu ≈ 1.66054 × 10–24 g.
Atomic Weight
The atomic weight of an element is the weighted average of the masses of its naturally occurring isotopes.
Calculated as:
Example: Chlorine atomic weight calculation using 35Cl and 37Cl isotopes.
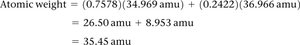
The Periodic Table
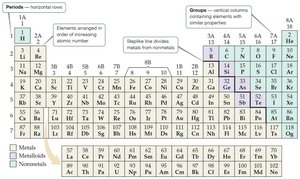
Organization of the Periodic Table
The periodic table arranges elements in order of increasing atomic number. Rows are called periods, columns are groups or families. Elements in the same group often have similar chemical properties.
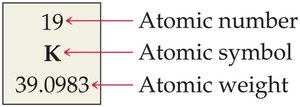
Reading the Periodic Table
Each box shows the atomic number (top), symbol (center), and atomic weight (bottom).
Classification of Elements
Metals: Left side, shiny, conduct heat/electricity, mostly solids.
Nonmetals: Right side, can be solid, liquid, or gas.
Metalloids: Along the staircase line, properties intermediate between metals and nonmetals.

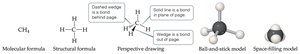
Chemical Formulas and Models
Chemical Formulas
Chemical formulas indicate the types and numbers of atoms in a compound. Subscripts show the number of each atom.
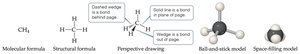
Molecular formulas: Exact number of atoms (e.g., H2O).
Empirical formulas: Simplest whole-number ratio (e.g., CH2O for glucose).
Diatomic Molecules
Seven elements exist naturally as diatomic molecules: H2, N2, O2, F2, Cl2, Br2, I2.

Structural and Molecular Models
Structural formulas show how atoms are connected; perspective, ball-and-stick, and space-filling models show three-dimensional arrangements.
Ions and Ionic Compounds
Formation of Ions
Atoms gain or lose electrons to form ions:
Cations: Positively charged, formed by loss of electrons (usually metals).
Anions: Negatively charged, formed by gain of electrons (usually nonmetals).
Writing Chemical Symbols for Ions
Symbol includes element, mass number (superscript), atomic number (subscript), and charge (superscript).
Example: for titanium ion with 22 protons, 26 neutrons, and 19 electrons.
Predicting Ionic Charges
Stable ions often have the same electron configuration as the nearest noble gas. Group 1 metals form +1 ions, group 2 metals form +2 ions, group 17 nonmetals form –1 ions, etc.
Common Ions
Some ions are so common they should be memorized, including polyatomic ions like NO3– (nitrate), SO42– (sulfate), and NH4+ (ammonium).
Ionic Compounds
Ionic compounds are formed from metals and nonmetals (or contain NH4+). The formula is written to ensure electrical neutrality.
Chemical Nomenclature
Naming Ionic Compounds
Name the cation first, then the anion.
For metals with variable charge, indicate the charge with Roman numerals (e.g., FeCl3 is iron(III) chloride).
Monatomic anions end in –ide (e.g., Cl– is chloride).
Polyatomic ions have specific names (e.g., NO3– is nitrate).
Naming Acids
If the anion ends in –ide, the acid name is hydro–[root]–ic acid (e.g., HCl is hydrochloric acid).
If the anion ends in –ite, the acid name is [root]–ous acid (e.g., HClO2 is chlorous acid).
If the anion ends in –ate, the acid name is [root]–ic acid (e.g., HNO3 is nitric acid).
Naming Binary Molecular Compounds
The element farther left or lower in the periodic table is named first.
Prefixes indicate the number of atoms (mono-, di-, tri-, etc.).
The second element ends in –ide (e.g., CO2 is carbon dioxide).
Summary Table: Common Ions
Cation | Formula | Anion | Formula |
|---|---|---|---|
Sodium | Na+ | Chloride | Cl– |
Calcium | Ca2+ | Sulfate | SO42– |
Ammonium | NH4+ | Nitrate | NO3– |
Iron(III) | Fe3+ | Oxide | O2– |
Conclusion
This chapter provides the essential foundation for understanding the structure of matter, the organization of elements, and the rules for naming chemical substances. Mastery of these concepts is critical for success in all areas of general chemistry.
