 Back
BackAtoms, Molecules, and Ions: Foundations of Chemical Structure and Nomenclature
Study Guide - Smart Notes

Atoms, Molecules, and Ions
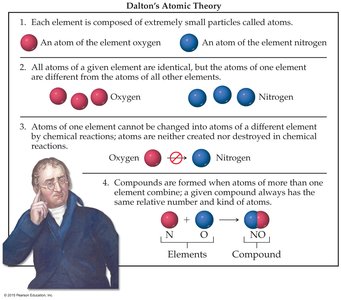
Dalton’s Atomic Theory
Dalton’s Atomic Theory laid the groundwork for modern chemistry by describing the nature of atoms and their role in chemical reactions.
Atoms: Each element is composed of extremely small particles called atoms.
Elemental Identity: All atoms of a given element are identical in mass and properties, but differ from atoms of other elements.
Conservation of Atoms: Atoms cannot be created or destroyed in chemical reactions; they are simply rearranged.
Compound Formation: Compounds are formed when atoms of more than one element combine in fixed, simple ratios.
Law of Conservation of Mass
The total mass of substances present at the end of a chemical process is the same as the mass of substances present before the process took place. This principle is fundamental to all chemical reactions.
Law of Multiple Proportions
If two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in the ratio of small whole numbers. This law supports the concept of atoms combining in simple, definite ratios.
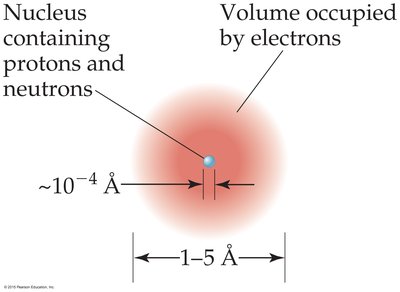
Structure of the Atom
Atoms consist of a dense nucleus containing protons and neutrons, surrounded by electrons occupying most of the atom’s volume.
Nucleus: Contains protons (positively charged) and neutrons (neutral).
Electrons: Negatively charged particles found outside the nucleus.
Atomic Neutrality: In a neutral atom, the number of protons equals the number of electrons.
Subatomic Particles: Properties and Comparison
The three main subatomic particles differ incharge and mass:
1 Particle | Charge | Mass (amu) |
|---|---|---|
Proton | Positive (1+) | 1.0073 |
Neutron | None (neutral) | 1.0087 |
Electron | Negative (1–) | 5.486 × 10–4 |

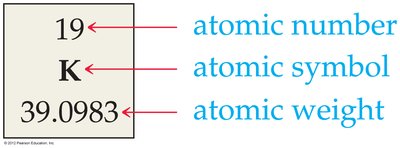
Atomic Number and Mass Number
Atoms are identified by their atomic number (Z), which is the number of protons in the nucleus. The mass number is the total number of protons and neutrons.
Atomic Number (Z): Written as a subscript before the element symbol.
Mass Number: Written as a superscript before the element symbol.
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers but identical chemical properties.
Atomic Mass Unit (amu)
The atomic mass unit is defined as one-twelfth the mass of a carbon-12 atom:
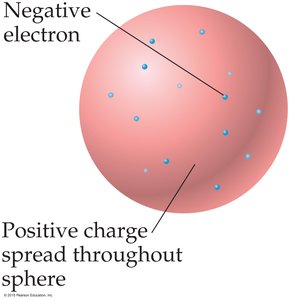
Models of the Atom
Early models of the atom, such as the "plum pudding" model, depicted electrons embedded in a sphere of positive charge. Later, Rutherford’s nuclear model placed a dense nucleus at the center with electrons around it.
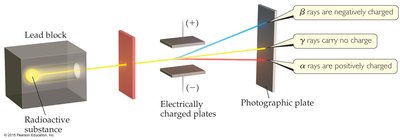
Radioactivity
Radioactive substances emit different types of radiation, which can be separated by their charge:
Alpha (α) rays: Positively charged
Beta (β) rays: Negatively charged
Gamma (γ) rays: No charge
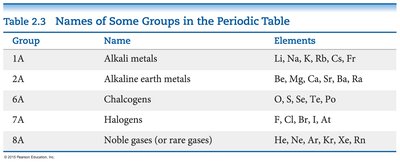
The Periodic Table: Groups and Families
The periodic table organizes elements into groups (columns) with similar chemical properties. Some important groups include:
Group | Name | Elements |
|---|---|---|
1A | Alkali metals | Li, Na, K, Rb, Cs, Fr |
2A | Alkaline earth metals | Be, Mg, Ca, Sr, Ba, Ra |
6A | Chalcogens | O, S, Se, Te, Po |
7A | Halogens | F, Cl, Br, I, At |
8A | Noble gases | He, Ne, Ar, Kr, Xe, Rn |
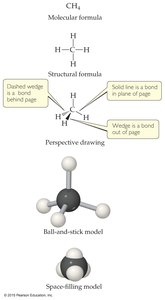
Chemical Formulas and Models
Chemical compounds can be represented in several ways, including molecular formulas, structural formulas, and three-dimensional models.
Molecular Formula: Shows the types and numbers of atoms (e.g., CH4 for methane).
Structural Formula: Shows how atoms are bonded.
Perspective,Ball-and-Stick, and Space-Filling Models: Visualize the three-dimensional arrangement of atoms.
. 
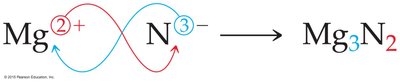
Naming Ions and Compounds
Compounds are electrically neutral. The formula can be determined by balancing the charges of the cation and anion. For example, magnesium nitride is formed by combining Mg2+ and N3– ions:
Naming Oxyanions and Acids
Oxyanions are polyatomic ions containing oxygen. Their names depend on the number of oxygen atoms:
Fewer oxygens: ends in -ite (e.g., NO2–: nitrite)
More oxygens: ends in -ate (e.g., NO3–: nitrate)
Fewest oxygens: prefix hypo- and ends in -ite (e.g., ClO–: hypochlorite)
Most oxygens: prefix per- and ends in -ate (e.g., ClO4–: perchlorate)
Acid names are derived from their anions:
Anion ends in -ide: hydro-...ic acid (e.g., HCl: hydrochloric acid)
Anion ends in -ite: ...ous acid (e.g., HClO: hypochlorous acid)
Anion ends in -ate: ...ic acid (e.g., HClO3: chloric acid)
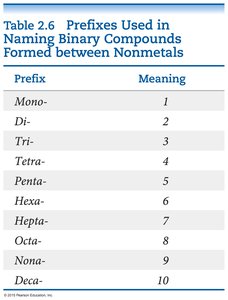
Prefixes for Binary Compounds of Nonmetals
Binary compounds formed between nonmetals use prefixes to indicate the number of each atom:
Prefix | Meaning |
|---|---|
Mono- | 1 |
Di- | 2 |
Tri- | 3 |
Tetra- | 4 |
Penta- | 5 |
Hexa- | 6 |
Hepta- | 7 |
Octa- | 8 |
Nona- | 9 |
Deca- | 10 |
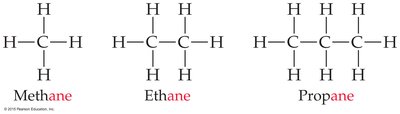
Hydrocarbons and Functional Groups
The simplest hydrocarbons are alkanes, which contain only carbon and hydrogen. Their names are based on the number of carbon atoms (meth- = 1, eth- = 2, prop- = 3, etc.).
Alkanes: Saturated hydrocarbons with single bonds only.
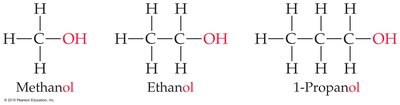
When a hydrogen atom in an alkane is replaced by a functional group (such as -OH), the compound’s name changes accordingly. Alcohols, for example, end in -ol.
Summary Table: Key Laws and Concepts
Law/Concept | Description |
|---|---|
Law of Conservation of Mass | Mass is neither created nor destroyed in chemical reactions. |
Law of Multiple Proportions | Elements combine in small whole-number ratios. |
Dalton’s Atomic Theory | Atoms are indivisible, unique to each element, and combine in fixed ratios. |
Atomic Number (Z) | Number of protons in the nucleus. |
Mass Number | Total number of protons and neutrons. |
