 Back
BackAtoms, Molecules, and Ions: Foundations of Chemical Structure and Nomenclature
Study Guide - Smart Notes

Atoms, Molecules, and Ions
Chemistry, Matter, and Its Properties
Chemistry is the study of matter, its properties, the changes it undergoes, and the energy associated with these changes. Matter is anything that has both mass and volume, encompassing all physical substances in the universe. The properties of matter are the characteristics that give each substance a unique identity, while composition refers to the types and amounts of simpler substances that make up a sample of matter.
Matter exists in three primary physical states: solid, liquid, and gas.
Properties include physical (e.g., melting point, density) and chemical (e.g., reactivity) characteristics.
Composition determines the identity and behavior of a substance.
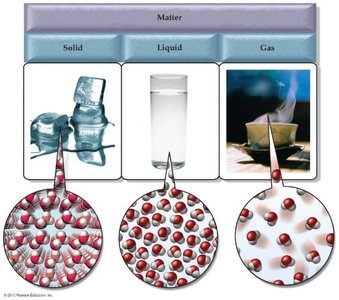
Physical States of Matter
The three main states of matter differ in the arrangement and motion of their particles:
Solid: Particles are closely packed in a fixed arrangement and vibrate about fixed positions.
Liquid: Particles are close together but can move past one another, giving liquids an indefinite shape but definite volume.
Gas: Particles are far apart and move freely, resulting in both indefinite shape and volume.
State | Atomic/Molecular Motion | Atomic/Molecular Spacing | Shape | Volume | Compressibility |
|---|---|---|---|---|---|
Solid | Oscillation/vibration about fixed point | Close together | Definite | Definite | Incompressible |
Liquid | Free to move relative to one another | Close together | Indefinite | Definite | Incompressible |
Gas | Free to move relative to one another | Far apart | Indefinite | Indefinite | Compressible |
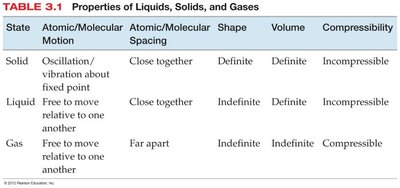
The Periodic Table and Classification of Elements
The Periodic Table
The periodic table organizes elements by increasing atomic number and groups them by similar chemical properties. Dmitri Mendeleev first arranged elements by mass, but the modern table uses atomic number. The table is divided into metals, nonmetals, and metalloids, with transition metals occupying the center block (groups 1B–8B) and main group elements in groups 1A–8A.
Metals: Good conductors, malleable, ductile, and typically solid at room temperature.
Nonmetals: Poor conductors, can be gases, liquids, or brittle solids.
Metalloids: Exhibit properties intermediate between metals and nonmetals.
Transition Metals: Found in the center of the table, often form colored compounds and have variable oxidation states.
General Reactivity Patterns
Group 1A (Alkali Metals) and Group 7A (Halogens) are highly reactive.
Group 2A (Alkaline Earth Metals) and Group 6A (Chalcogens) are fairly reactive.
Group 8A (Noble Gases) are generally unreactive due to their stable electron configurations.
Atomic Theory and Structure
Observations Supporting Atomic Theory
Law of Mass Conservation: Matter is neither created nor destroyed in a chemical reaction.
Law of Definite Composition: All samples of a compound have the same proportions of their constituent elements.
Law of Multiple Proportions: When two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in small whole-number ratios.
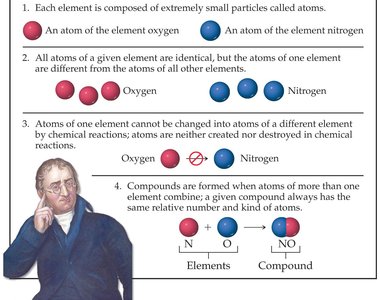
Dalton’s Atomic Theory
John Dalton proposed a theory to explain the nature of matter:
All matter is composed of indivisible atoms.
Atoms of a given element are identical; atoms of different elements are different.
Atoms cannot be created or destroyed in chemical reactions.
Compounds are formed by the combination of atoms in fixed ratios.
Discovery of Atomic Structure
Cathode-Ray Tubes (J.J. Thomson): Discovered the electron and measured its mass-to-charge ratio.
Millikan’s Oil Drop Experiment: Measured the charge of the electron and calculated its mass.
Rutherford’s Gold Foil Experiment: Discovered the atomic nucleus, showing that atoms are mostly empty space with a dense central nucleus.
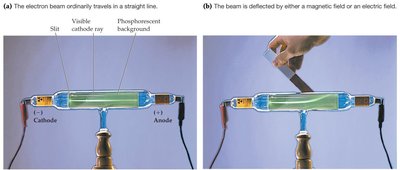
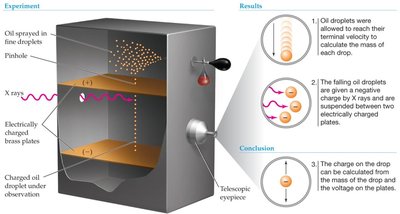
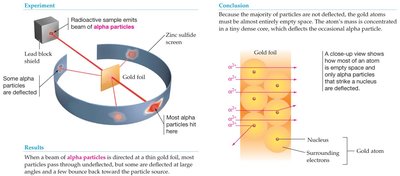
Subatomic Particles
Protons: Positively charged particles in the nucleus.
Neutrons: Neutral particles in the nucleus.
Electrons: Negatively charged particles orbiting the nucleus.
Atoms of the same element with different numbers of neutrons are called isotopes.
Atomic Mass, the Mole, and Chemical Calculations
Atomic Mass and Formula Mass
Atomic Mass: The weighted average mass of all naturally occurring isotopes of an element (units: amu).
Formula Mass: The sum of atomic masses in a chemical formula.
Example calculation for formula mass:
The Mole and Avogadro’s Number
Mole (mol): The amount of substance containing particles (Avogadro’s number).
Used as a conversion factor between atoms/molecules and grams.
Example: How many molecules are in 0.500 mol of water?
Example: How many moles are in atoms of iron (Fe)?
Classification of Matter and Chemical Bonds
Classification of Matter
Element: A pure substance made of only one kind of atom.
Compound: A pure substance composed of two or more elements chemically combined.
Homogeneous Mixture (Solution): Uniform composition throughout.
Heterogeneous Mixture: Non-uniform composition; distinct phases are visible.
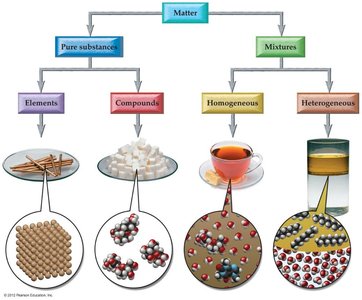
Types of Chemical Bonds
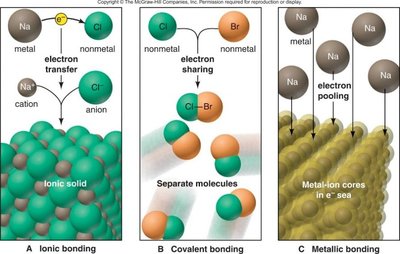
Ionic Bonding: Involves the transfer of electrons from a metal to a nonmetal, forming cations and anions.
Covalent Bonding: Involves the sharing of electrons between nonmetals.
Metallic Bonding: Involves a 'sea' of delocalized electrons shared among metal atoms.
Ions, Ionic Compounds, and Nomenclature
Ions and Ionic Compounds
Ion: An atom or molecule with a net electric charge due to the loss or gain of electrons.
Cation: Positively charged ion (loss of electrons).
Anion: Negatively charged ion (gain of electrons).
Binary Ionic Compound: Composed of two different elements: a metal (cation) and a nonmetal (anion).
Coulomb’s Law: The energy of attraction (or repulsion) between two charged particles is directly proportional to the product of their charges and inversely proportional to the distance between them:
Naming Ions and Ionic Compounds
Monoatomic Ions: Named by the element (for cations) or by adding “-ide” to the root of the element name (for anions).
Polyatomic Ions: Ions composed of more than one atom, often containing oxygen (oxyanions).
Naming Ionic Compounds: Name the cation first, then the anion.
Hydrates: Ionic compounds that include water molecules in their structure.
Naming Acids
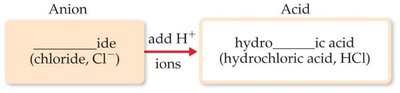
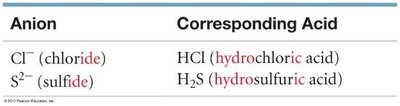
Binary Acids: Contain hydrogen and a monoatomic anion; named as “hydro-____-ic acid.”
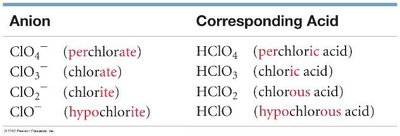
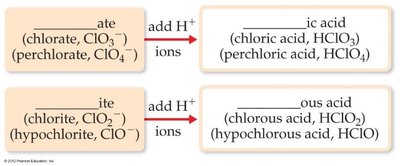
Oxoacids: Contain hydrogen and a polyatomic oxyanion; named based on the anion’s suffix:
“-ate” becomes “-ic acid” (e.g., chlorate → chloric acid)
“-ite” becomes “-ous acid” (e.g., chlorite → chlorous acid)
Naming Binary Covalent Compounds
Use prefixes to indicate the number of each atom (e.g., CO2 is carbon dioxide).
The more metallic element is named first, followed by the less metallic element with an “-ide” suffix.
Summary Table: Key Terms and Concepts
Term | Definition |
|---|---|
Atom | Smallest unit of an element retaining its properties |
Isotope | Atoms of the same element with different numbers of neutrons |
Mole | 6.02 × 1023 particles of a substance |
Ionic Bond | Electrostatic attraction between cations and anions |
Covalent Bond | Sharing of electrons between atoms |
Metallic Bond | Delocalized electrons shared among metal atoms |
