 Back
BackAtoms, Molecules, and Matter: Foundations of General Chemistry
Study Guide - Smart Notes

Atoms, Molecules, and Ions
Basic Units of Matter
Atoms are the fundamental building blocks of matter. They combine to form molecules, which are groups of atoms held together by chemical bonds. Ions are atoms or molecules that have gained or lost electrons, resulting in a net charge.
Atom: The smallest unit of an element, retaining its chemical properties.
Molecule: Two or more atoms bonded together, forming a distinct chemical entity.
Ion: An atom or molecule with a net electrical charge due to electron gain or loss.

Structure of Atoms and Molecules
Atomic Arrangement and Properties
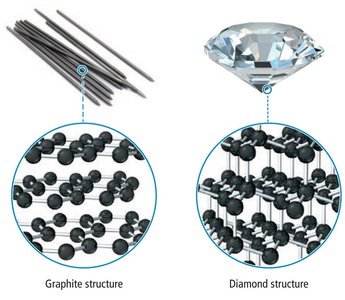
Small differences in atomic arrangement can result in large differences in the properties of substances. For example, graphite and diamond are both composed of carbon atoms, but their atomic arrangements differ:
Graphite: Carbon atoms are arranged in sheets, allowing layers to slide over each other.
Diamond: Carbon atoms are bonded in a three-dimensional network, making diamond extremely hard.
States of Matter
Solid, Liquid, and Gas
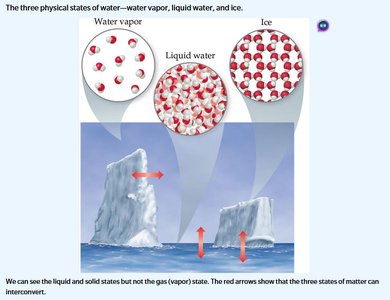
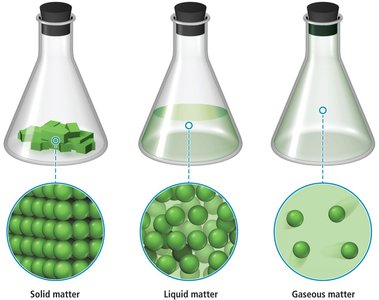
Matter exists in three primary states: solid, liquid, and gas. The state depends on temperature and the arrangement of particles.
Solid: Definite shape and volume; particles are closely packed.
Liquid: Definite volume but no definite shape; particles are less tightly packed and can flow.
Gas: No definite shape or volume; particles are far apart and move freely.
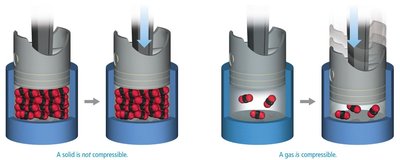
Compressibility of States
Solids are not compressible, while gases are highly compressible. Liquids are not easily compressed.
Solid: Not compressible due to tightly packed particles.
Gas: Compressible because particles are far apart.
Liquid: Not easily compressed; particles are close but can move.
Classification of Matter
Pure Substances and Mixtures
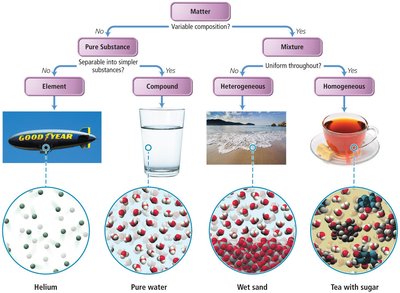
Matter can be classified as pure substances or mixtures. Pure substances have a fixed composition, while mixtures contain two or more substances.
Pure Substance: Matter with distinct properties and constant composition (e.g., water, sodium chloride).
Mixture: Combination of two or more substances with variable composition.
Elements and Compounds
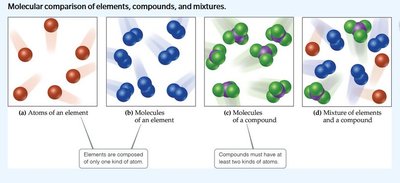
All substances are either elements or compounds:
Element: Cannot be decomposed into simpler substances; composed of one kind of atom.
Compound: Composed of two or more elements; contains two or more kinds of atoms.
Physical and Chemical Changes
Physical Changes
Physical changes alter only the state or appearance of a substance, not its composition. The identity of the atoms or molecules remains unchanged.
Example: Boiling water changes it from liquid to gas, but the molecules remain H2O.

Chemical Changes
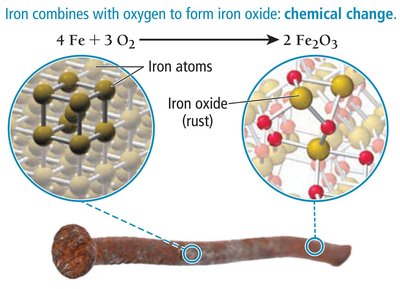
Chemical changes alter the composition of matter. Atoms rearrange, forming new substances.
Example: Rusting of iron forms iron oxide (Fe2O3).
Physical and Chemical Properties
Definitions and Examples
Physical Property: Observed without changing composition (e.g., odor, color, melting point).
Chemical Property: Observed only by changing composition via chemical reaction (e.g., flammability, acidity).
Famous Chemists and Atomic Theory
Antoine Lavoisier
Antoine Lavoisier is known as the Father of Modern Chemistry. He defined elements, proved water is a compound, and established the law of conservation of mass. 
John Dalton and Atomic Theory
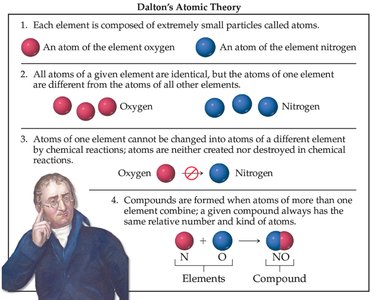
Dalton's atomic theory laid the foundation for understanding matter:
Elements are composed of atoms.
Atoms of a given element are identical.
Atoms cannot be changed into atoms of another element by chemical reactions.
Compounds are formed from atoms of more than one element.
Subatomic Particles
Protons, Neutrons, and Electrons
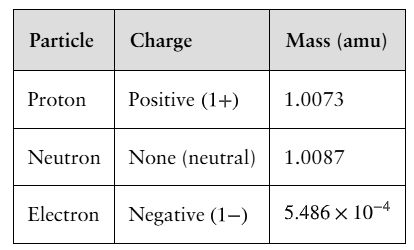
Atoms are composed of protons, neutrons, and electrons. Protons and neutrons have similar masses, while electrons are much lighter.
Particle | Charge | Mass (amu) |
|---|---|---|
Proton | Positive (1+) | 1.0073 |
Neutron | None (neutral) | 1.0087 |
Electron | Negative (1−) | 5.486 \times 10^{-4} |
Atomic Number and Element Identity
Defining Elements
The number of protons in an atom's nucleus (atomic number, Z) defines the element. For example, helium has 2 protons, carbon has 6. 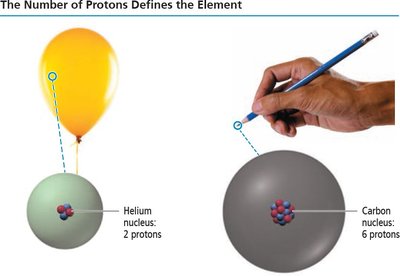
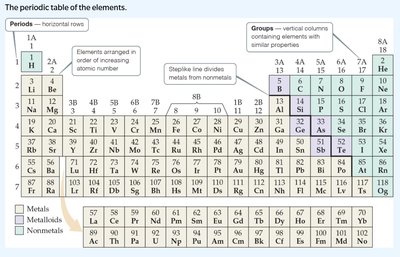
The Periodic Table
Structure and Importance
The periodic table organizes elements by increasing atomic number and groups elements with similar properties. It predicts reactivity, identifies trends, and is foundational for chemistry.
Groups: Vertical columns with similar properties.
Periods: Horizontal rows.
Trends: Atomic size, electronegativity, ionization energy.
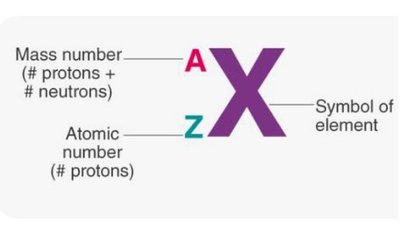
Isotopes
Definition and Notation
Isotopes are atoms of the same element (same number of protons) but different numbers of neutrons. Isotope notation includes mass number (A) and atomic number (Z).
Example: Carbon-12 (6 protons, 6 neutrons), Carbon-14 (6 protons, 8 neutrons).
Chemical Formulas and Compounds
Molecular and Empirical Formulas
Molecular Formula: Shows the actual number of atoms in a molecule (e.g., H2O2).
Empirical Formula: Shows the simplest ratio of atoms (e.g., HO).
Ions and Ionic Compounds
Formation and Properties
When atoms gain or lose electrons, they form ions. Ionic compounds are composed of cations (positive ions) and anions (negative ions).
Cation: Positive ion formed by loss of electrons.
Anion: Negative ion formed by gain of electrons.
Balancing Chemical Equations
Law of Conservation of Mass
Chemical equations must be balanced to reflect the conservation of mass. The number of atoms of each element must be the same on both sides of the equation.
Example:
Mole and Avogadro's Number
Definition and Calculation
A mole is the amount of substance containing entities (Avogadro's number). The molar mass is the mass of one mole of a substance, numerically equal to its atomic or molecular weight in grams.
Example: 1 mol of Cl has a mass of 35.5 g.
Mass versus Weight
Definitions and Differences
Mass: Amount of matter in an object; constant everywhere; measured in kilograms.
Weight: Gravitational force acting on mass; varies by location; measured in Newtons.
