 Back
BackAtoms: Structure, Properties, and Fundamental Laws in Chemistry
Study Guide - Smart Notes

Atoms and the Particulate Nature of Matter
Structure Determines Properties
The physical and chemical properties of matter are determined by the way its constituent particles—atoms and molecules—are arranged and interact. Chemistry seeks to understand these properties by studying matter at the particulate level.
Matter: Anything that has mass and occupies space.
Atoms: Fundamental building blocks of matter; submicroscopic particles composed of protons, neutrons, and electrons.
Molecules: Groups of two or more atoms bonded in specific geometric arrangements.
Example: Water (H2O) is a molecule made of two hydrogen atoms and one oxygen atom.
Classification of Matter
States of Matter
Matter exists in three primary states: solid, liquid, and gas. The arrangement and movement of particles in each state determine its properties.
Solid: Fixed volume and shape; particles vibrate but do not move past each other.
Liquid: Fixed volume, variable shape; particles move freely relative to each other.
Gas: Variable volume and shape; particles are far apart and move freely, making gases compressible.
Classification by Composition
Matter can be classified as pure substances or mixtures, based on its composition.
Pure Substance: Composed of only one component; composition is invariant.
Mixture: Composed of two or more components; composition can vary.
Types of Pure Substances
Element: Cannot be chemically broken down into simpler substances; composed of one type of atom.
Compound: Composed of two or more elements in fixed, definite proportions.
Types of Mixtures
Heterogeneous Mixture: Composition varies from one region to another; components are visibly distinct.
Homogeneous Mixture: Uniform composition throughout; appears as a single substance.
The Scientific Approach to Knowledge
Scientific Method
The scientific method is a systematic process for understanding nature, based on observation, hypothesis formation, experimentation, and development of laws and theories.
Observation: Collection of data about nature.
Hypothesis: Tentative explanation of observations; must be falsifiable.
Law: Statement summarizing past observations and predicting future ones (e.g., Law of Conservation of Mass).
Theory: Well-established explanation of why phenomena occur; validated by experiments but never conclusively proven.
Early and Modern Atomic Theory
Historical Development
Early philosophers like Democritus proposed the existence of atoms, while Dalton provided experimental evidence and formulated atomic theory.
Dalton's Atomic Theory:
Matter is composed of atoms.
Atoms of an element are identical in mass and properties.
Atoms of different elements differ in properties.
Compounds are formed by atoms in fixed ratios.
Atoms are neither created nor destroyed in chemical reactions.
Fundamental Laws
Law of Conservation of Mass: Matter is neither created nor destroyed in a chemical reaction.
Law of Definite Proportions: All samples of a compound have the same proportions of constituent elements.
Law of Multiple Proportions: When two elements form different compounds, the mass ratios of one element to a fixed mass of the other are small whole numbers.
Example: Carbon dioxide and carbon monoxide illustrate the law of multiple proportions.


Discovery of Subatomic Particles
Discovery of the Electron
J.J. Thomson's cathode ray experiments revealed the existence of electrons, negatively charged particles much lighter than atoms.
Charge-to-mass ratio:
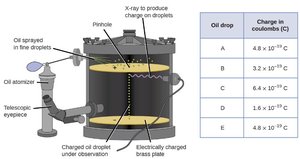
Millikan's Oil Drop Experiment
Millikan determined the charge of a single electron using oil droplets and an electric field.
Charge of electron:
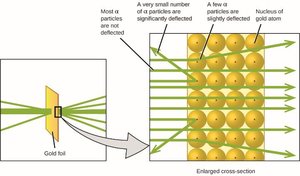
Rutherford's Gold Foil Experiment
Rutherford's experiment led to the nuclear model of the atom, with a dense, positively charged nucleus surrounded by electrons.
Nucleus: Contains protons and neutrons; most of the atom's mass.
Electrons: Negatively charged, occupy most of the atom's volume.
Subatomic Particles and Atomic Structure
Protons, Neutrons, and Electrons
Proton: Mass = 1.0073 amu, Charge = +1
Neutron: Mass = 1.0087 amu, Charge = 0
Electron: Mass = 0.00055 amu, Charge = -1
Atomic Number and Mass Number
Atomic Number (Z): Number of protons; defines the element.
Mass Number (A): Number of protons + number of neutrons.
Number of Neutrons:
Neutral Atoms and Ions
Neutral Atom: Number of protons equals number of electrons.
Ion: Atom with unequal numbers of protons and electrons.
Cation: Positively charged ion (loss of electrons).
Anion: Negatively charged ion (gain of electrons).
Chemical Symbols and the Periodic Table
Element Symbols
Chemical symbols are abbreviations for elements, often derived from their English or Latin names. Only the first letter is capitalized.

Isotopes
Definition and Representation
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers.
Notation: or X-A (where X is the element symbol, A is mass number, Z is atomic number)
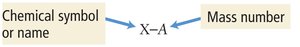
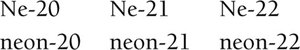
Atomic Mass and Mass Spectrometry
Atomic Mass
The atomic mass listed in the periodic table is the weighted average of all naturally occurring isotopes of an element.
Calculation:
The Mole and Avogadro's Number
Definition and Importance
The mole is a counting unit for atoms, molecules, or ions, defined as the number of entities in 12 grams of carbon-12.
Avogadro's Number: entities per mole
Mole Conversions
Atoms to Moles:
Moles to Atoms:
Converting Between Mass, Moles, and Number of Particles
Molar Mass
The molar mass is the mass in grams of one mole of a substance, numerically equal to its atomic or formula mass in amu.
Mass to Moles:
Moles to Mass:
Chemical Formulas
Molecular and Empirical Formulas
Molecular Formula: Shows the actual number of atoms of each element in a molecule.
Empirical Formula: Shows the simplest whole-number ratio of atoms in a compound.
Example: Benzene: Molecular formula = C6H6, Empirical formula = CH
Summary Table: Subatomic Particles
Particle | Mass (amu) | Charge |
|---|---|---|
Proton | 1.0073 | +1 |
Neutron | 1.0087 | 0 |
Electron | 0.00055 | -1 |
Additional info: Academic context and examples have been added to clarify and expand upon the original lecture notes, ensuring completeness and self-contained study guidance.
