 Back
BackAtoms: Structure, Properties, and Laws of Matter
Study Guide - Smart Notes

Atoms and Matter
Definition and Composition of Matter
Matter is defined as anything that has mass and occupies space. Chemistry is the science that seeks to understand matter and its properties. Matter is composed of particles, including subatomic particles (neutrons, protons, electrons), atoms, and molecules. The arrangement and interaction of these particles determine the physical properties of matter.
Atoms: Basic submicroscopic particles that constitute the fundamental building blocks of ordinary matter.
Molecules: Particles formed when two or more atoms bond together in specific geometric arrangements.
Example: Water molecule consists of two hydrogen atoms and one oxygen atom.
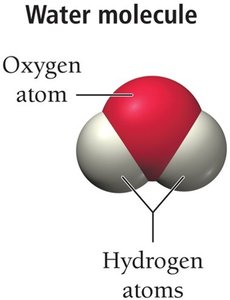
Classification of Matter
States of Matter
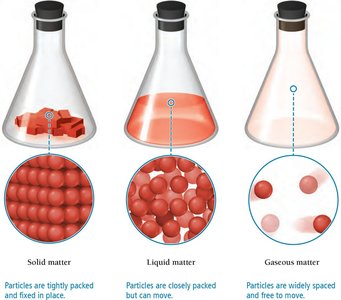
Matter can exist in three primary states: solid, liquid, and gas. The state depends on the arrangement and movement of particles and changes with temperature.
Solid: Atoms or molecules are closely packed in fixed locations, resulting in a fixed volume and rigid shape.
Liquid: Atoms or molecules are closely packed but can move relative to each other, giving liquids a fixed volume but not a fixed shape.
Gas: Atoms or molecules are widely spaced and free to move, making gases compressible and able to fill the shape of their container.

Classification by Composition
Matter can be classified as pure substances or mixtures based on its composition.
Pure Substance: Made up of only one component; composition is invariant.
Mixture: Composed of two or more components in proportions that can vary.
Elements: Substances that cannot be chemically broken down into simpler substances.
Compounds: Substances composed of two or more elements in fixed, definite proportions.
Heterogeneous Mixture: Composition varies from one region to another (e.g., salt and sand mixture).
Homogeneous Mixture: Uniform composition throughout (e.g., sweetened tea).

The Scientific Approach to Knowledge
The Scientific Method
The scientific method is a systematic process for understanding nature through observation and experimentation. It involves:
Observations: Descriptions about the characteristics or behavior of nature.
Hypothesis: Tentative interpretation or explanation of observations; must be falsifiable.
Scientific Law: Brief statement summarizing past observations and predicting future ones (e.g., law of conservation of mass).
Theory: Well-established explanation for why natural phenomena occur; validated by experiments but never conclusively proven.
Early Atomic Theory and Laws
Historical Development
Early philosophers like Democritus proposed that matter was composed of small, indestructible particles called atoms. Dalton later provided evidence for the atomic theory and formulated several key laws:
Law of Conservation of Mass: In a chemical reaction, matter is neither created nor destroyed.
Law of Definite Proportions: All samples of a given compound have the same proportions of their constituent elements.
Law of Multiple Proportions: When two elements form different compounds, the masses of one element that combine with 1 gram of the other can be expressed as a ratio of small whole numbers.


Structure of the Atom
Discovery of Subatomic Particles
Atoms are composed of three main subatomic particles:
Protons: Positively charged particles found in the nucleus.
Neutrons: Neutral particles found in the nucleus; similar mass to protons.
Electrons: Negatively charged particles found outside the nucleus.
Table: Subatomic Particles
Particle | Mass (kg) | Mass (amu) | Charge (relative) | Charge (C) |
|---|---|---|---|---|
Proton | 1.67262 × 10-27 | 1.00727 | +1 | +1.60218 × 10-19 |
Neutron | 1.67493 × 10-27 | 1.00866 | 0 | 0 |
Electron | 0.00091 × 10-27 | 0.00055 | -1 | -1.60218 × 10-19 |
Atomic Number and Isotopes
The atomic number (Z) is the number of protons in the nucleus and defines the element. Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers (A).
Example: Neon has three isotopes: 20Ne, 21Ne, 22Ne.
Notation: Isotopes are represented as AZX or X-A.
Table: Neon Isotopes
Symbol | Protons | Neutrons | Mass Number (A) | Natural Abundance (%) |
|---|---|---|---|---|
20Ne | 10 | 10 | 20 | 90.48 |
21Ne | 10 | 11 | 21 | 0.27 |
22Ne | 10 | 12 | 22 | 9.25 |
Ions and Atomic Mass
Ions: Charged Atoms
Atoms can lose or gain electrons to become ions:
Cations: Positively charged ions (loss of electrons).
Anions: Negatively charged ions (gain of electrons).
Atomic Mass and Molar Mass
The atomic mass of an element is the weighted average of the masses of its isotopes, based on their natural abundance. The molar mass (grams per mole) is numerically equal to the atomic mass in amu.
Formula:
Example: Chlorine's atomic mass is calculated from its two isotopes: amu
The Mole and Counting Atoms
Definition of the Mole
A mole (mol) is a counting unit for atoms, molecules, or other particles. One mole contains particles (Avogadro's number).
Relationship: particles
Example:
Converting Between Mass, Moles, and Particles
To count atoms by weighing, use the molar mass as a conversion factor between mass (grams) and amount (moles).
Formula:
Formula:
Example:
Summary Table: Classification of Matter
Type | Description | Example |
|---|---|---|
Element | Cannot be broken down; composed of one type of atom | Helium |
Compound | Composed of two or more elements in fixed proportions | Water |
Heterogeneous Mixture | Composition varies; distinct regions | Wet sand |
Homogeneous Mixture | Uniform composition throughout | Sweetened tea |

Key Equations
Law of Definite Proportions:
Law of Multiple Proportions:
Atomic Mass:
Mole Conversion: particles
Mass to Moles:
Additional info:
All content is directly relevant to Chapter 1: Atoms, and covers foundational concepts for general chemistry.
Images included are only those that visually clarify the structure of matter, classification, and key laws.
