 Back
BackBasic Concepts of Chemical Bonding: Ionic, Covalent, and Metallic Bonds, Lewis Structures, and Molecular Geometry
Study Guide - Smart Notes

Basic Concepts of Chemical Bonding
Types of Chemical Bonds
Chemical bonds are the forces that hold atoms together in compounds. There are three primary types of chemical bonds:
Ionic Bonds: Formed by the electrostatic attraction between oppositely charged ions, typically between metals and nonmetals.
Covalent Bonds: Involve the sharing of electron pairs between atoms, usually between nonmetals.
Metallic Bonds: Occur between metal atoms, where electrons are delocalized over many atoms.

Energetics of Ionic Bonding
Formation of Ionic Compounds
The formation of ionic compounds involves several energetic steps:
Ionization Energy (IE): The energy required to remove an electron from a gaseous atom (e.g., Na → Na+ + e-).
Electron Affinity (EA): The energy change when an electron is added to a gaseous atom (e.g., Cl + e- → Cl-).
Lattice Energy (U): The energy released when gaseous ions combine to form an ionic solid. It is governed by Coulomb's law:
Lattice energy increases with higher ionic charges and decreases with larger ionic radii.
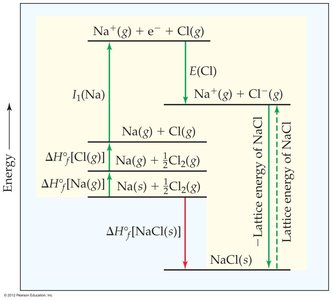
Born-Haber Cycle
The Born-Haber cycle is used to analyze the energetics of ionic compound formation by accounting for all energy changes (ionization, electron affinity, lattice energy, etc.).
Lewis Symbols and the Octet Rule
Lewis Symbols
Lewis symbols (or electron-dot symbols) represent valence electrons as dots around the chemical symbol of an element. These are useful for tracking electrons during bond formation.

The Octet Rule
Atoms tend to gain, lose, or share electrons to achieve eight valence electrons, attaining a noble gas configuration (s2p6). This is known as the octet rule. However, elements in periods 3-7 can have more than eight electrons (expanded octets).
Covalent Bonding and Lewis Structures
Lewis Electron Pair Theory
G.N. Lewis proposed that atoms achieve noble-gas configurations by sharing electrons. A covalent bond is formed by sharing a pair of electrons (bonding pair), while unshared pairs are called lone pairs.
Drawing Lewis Structures
Lewis structures visually represent the arrangement of electrons in molecules:
Add up all valence electrons for all atoms in the molecule or ion.
Identify the central atom (usually the least electronegative, except hydrogen).
Connect outer atoms to the central atom with single bonds.
Fill the octets of the outer atoms, then the central atom.
If the central atom lacks an octet, form multiple bonds as needed.

Multiple Bonds
Atoms can share more than one pair of electrons to complete their octets:
Single bond: one shared pair
Double bond: two shared pairs
Triple bond: three shared pairs
Multiple bonds are shorter and stronger than single bonds. The strength of a bond is measured by its bond enthalpy (D), the energy required to break the bond.
Bond Polarity and Electronegativity
Bond Polarity
Bond polarity describes the sharing of electrons in a covalent bond:
Non-polar covalent bond: Electrons are shared equally (e.g., Cl2).
Polar covalent bond: One atom attracts electrons more strongly (e.g., HCl).
Electronegativity
Electronegativity is the ability of an atom in a molecule to attract electrons to itself. It increases across a period and decreases down a group in the periodic table. The greater the difference in electronegativity, the more polar the bond.
Compound | Electronegativity Difference | Type of Bond |
|---|---|---|
F2 | 4.0 – 4.0 = 0 | Non-Polar Covalent |
HF | 4.0 – 2.1 = 1.9 | Polar Covalent |
LiF | 4.0 – 1.0 = 3.0 | Ionic |
Dipole Moment
A dipole moment () is a quantitative measure of bond polarity:
where Q is the magnitude of charge and r is the distance between charges. Dipole moments are measured in debyes (D).
Formal Charge and Resonance Structures
Formal Charge
The formal charge of an atom in a molecule is calculated as:
Formal charge = (number of valence electrons in the free atom) – (number of nonbonding electrons + 1/2 number of bonding electrons)
The best Lewis structure minimizes formal charges and places negative charges on the most electronegative atoms.
Resonance Structures
Some molecules cannot be adequately represented by a single Lewis structure. Resonance structures are multiple valid Lewis structures for the same molecule, differing only in the arrangement of electrons. The actual structure is a hybrid of these forms.
Exceptions to the Octet Rule
There are three main exceptions to the octet rule:
Odd number of electrons: Molecules with an odd number of electrons (e.g., NO, NO2).
Less than an octet: Molecules where an atom (often B or Be) has fewer than eight electrons (e.g., BF3).
More than an octet: Atoms from period 3 and beyond can have expanded valence shells (e.g., PCl5, SF4).
VSEPR Theory and Molecular Geometry
Valence Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory predicts the three-dimensional arrangement of atoms in a molecule by minimizing repulsions between electron domains (regions of electron density around a central atom).
Electron domain geometry: Arrangement of all electron domains (bonding and nonbonding) around the central atom.
Molecular geometry: Arrangement of only the atoms (ignoring lone pairs).
Multiple bonds and lone pairs exert greater repulsive forces than single bonds, affecting bond angles and molecular shape.
Steps for Determining Molecular Geometry
Draw the Lewis structure.
Count the total number of electron domains around the central atom.
Arrange the electron domains to minimize repulsion (choose the appropriate geometry).
Ignore lone pairs when describing the molecular geometry.
Effect of Non-Bonding Electrons and Multiple Bonds
Lone pairs and multiple bonds occupy more space than bonding pairs, leading to smaller bond angles in molecules with lone pairs or multiple bonds.
Molecular Polarity
The overall polarity of a molecule depends on both the polarity of individual bonds and the molecular geometry. For molecules with more than two atoms, the vector sum of bond dipoles determines the molecular dipole moment.
Non-polar molecule: Bond dipoles cancel due to symmetry (e.g., CO2).
Polar molecule: Bond dipoles do not cancel (e.g., H2O).
Summary Table: Bond Type and Polarity
Bond Type | Electronegativity Difference | Polarity |
|---|---|---|
Non-polar covalent | 0 | Non-polar |
Polar covalent | 0 < x < 2 | Polar |
Ionic | > 2 | Very polar (ionic) |
Practice: Determining Molecular Polarity
For each molecule or ion (e.g., SO2, SO3, NF3, ClF3, IF5, SF4, SnCl3-):
Draw the Lewis structure.
Determine the electron domain geometry.
Determine the molecular geometry.
Identify all bond dipoles.
Determine the overall polarity of the molecule.
