 Back
BackBasic Concepts of Chemical Bonding: Ionic, Covalent, and Metallic Bonds
Study Guide - Smart Notes

Basic Concepts of Chemical Bonding
Types of Chemical Bonds
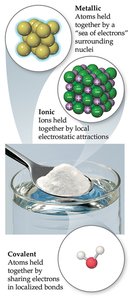
Chemical bonds are the forces that hold atoms together in compounds. There are three primary types of chemical bonds: ionic, covalent, and metallic. Each type involves different mechanisms of electron interaction and results in distinct physical and chemical properties.
Ionic Bonds: Formed by electrostatic attraction between oppositely charged ions, typically between metals and nonmetals.
Covalent Bonds: Formed by sharing pairs of electrons between atoms, usually between nonmetals.
Metallic Bonds: Involve a 'sea' of delocalized electrons that hold metal atoms together.
Lewis Symbols and the Octet Rule
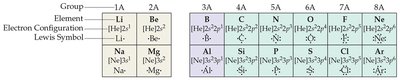
Lewis Symbols
Lewis symbols are a simple way to represent the valence electrons of an atom. Each dot represents a valence electron, and the element symbol is used as the core.
Valence Electrons: Electrons in the outermost shell, important for bonding.
Lewis Symbol: Element symbol surrounded by dots indicating valence electrons.
The Octet Rule
The octet rule states that atoms tend to gain, lose, or share electrons to achieve eight valence electrons, resembling the electron configuration of a noble gas.
Application: Most main group elements follow the octet rule when forming compounds.
Exceptions: Hydrogen (2 electrons), some elements with expanded octets.
Ionic Bonding
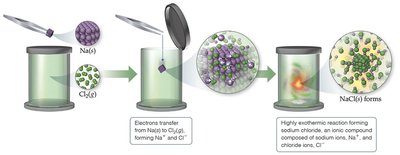
Formation of Ionic Bonds
Ionic bonds are formed when electrons are transferred from a metal atom to a nonmetal atom, resulting in the formation of cations and anions.
Electron Transfer: Metal loses electrons (forms cation), nonmetal gains electrons (forms anion).
Example: Sodium (Na) reacts with chlorine (Cl) to form sodium chloride (NaCl).
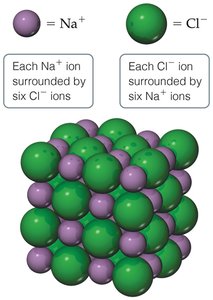
Properties of Ionic Substances
Ionic compounds exhibit characteristic physical properties due to their three-dimensional lattice structure.
Brittle: Ionic solids break along smooth planes.
High Melting Points: Strong electrostatic forces require significant energy to break.
Crystalline: Well-defined, repeating lattice structure.
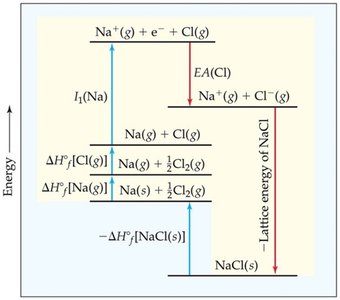
Energetics of Ionic Bonding: The Born–Haber Cycle
The Born–Haber cycle is used to analyze the energetics of ionic compound formation, including ionization energy, electron affinity, and lattice energy.
Lattice Energy: The energy released when ions form a solid lattice from gaseous ions.
Factors Affecting Lattice Energy: Increases with higher ionic charge and decreases with larger ionic radius.
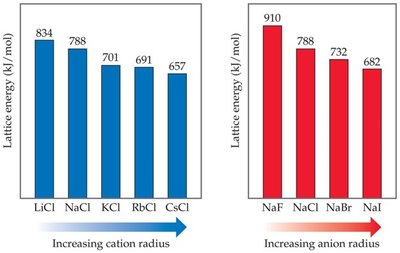
Trends in Lattice Energy
Lattice energy varies with the size and charge of the ions involved. Smaller ions and higher charges result in greater lattice energies.
Increasing Cation Radius: Lattice energy decreases.
Increasing Anion Radius: Lattice energy decreases.
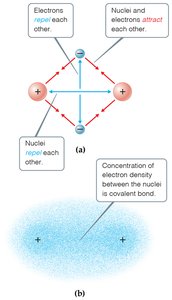
Covalent Bonding
Nature of Covalent Bonds
Covalent bonds involve the sharing of electron pairs between atoms, primarily nonmetals. The stability of the bond depends on the balance between attractive and repulsive forces.
Attractions: Between electrons and nuclei.
Repulsions: Between electrons and between nuclei.
Bond Formation: Occurs when attractions outweigh repulsions.
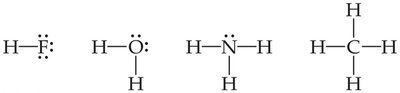
Lewis Structures
Lewis structures visually represent the sharing of electrons in covalent bonds. Each line or pair of dots represents a shared pair of electrons.
Bonding Pairs: Shared electrons between atoms.
Lone Pairs: Unshared electrons localized on one atom.

Number of Bonds for Nonmetals
Nonmetals form a characteristic number of bonds based on their valence electrons and the octet rule.
Hydrogen: Forms one bond.
Oxygen: Forms two bonds.
Nitrogen: Forms three bonds.
Carbon: Forms four bonds.
Polar and Nonpolar Covalent Bonds
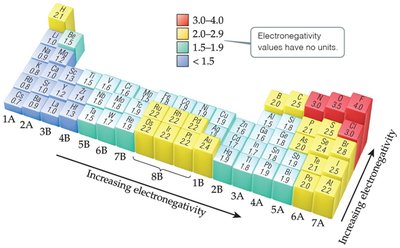
Electronegativity
Electronegativity is the ability of an atom in a molecule to attract electrons to itself. It determines the distribution of electron density in a bond.
Trend: Increases across a period, decreases down a group.
Most Electronegative Element: Fluorine (F).
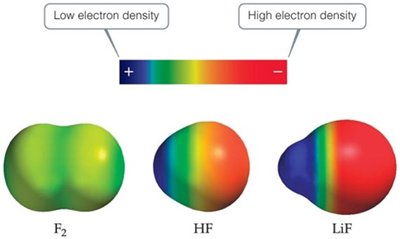
Polar Covalent Bonds
When two atoms share electrons unequally due to differences in electronegativity, a polar covalent bond forms. The more electronegative atom acquires a partial negative charge, while the other becomes partially positive.
Electronegativity Difference: The greater the difference, the more polar the bond.
Partial Charges: Represented by δ+ and δ−.
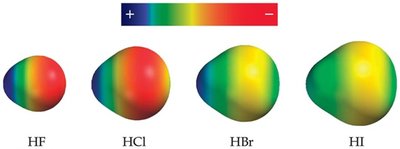
Bond Polarity in Hydrogen Halides
Hydrogen halides (HF, HCl, HBr, HI) demonstrate varying bond polarities based on electronegativity differences and bond lengths.
HF: Most polar, shortest bond length.
HI: Least polar, longest bond length.
Dipole Moments
Definition and Calculation
A dipole moment arises when two equal but opposite charges are separated by a distance. It is a measure of bond polarity and is calculated as:
Formula:
Units: Debye (D)
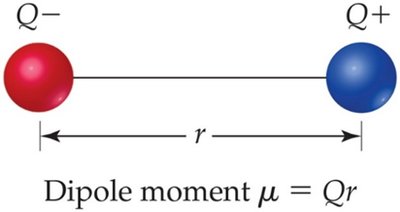
Comparing Ionic and Covalent Bonding
Bonding Continuum
There is a gradual transition between ionic and covalent bonding. The distinction is based on the degree of electron transfer versus sharing, guided by electronegativity differences and physical properties.
Ionic: Complete electron transfer, typically metal + nonmetal, electronegativity difference > 2.0.
Covalent: Electron pair sharing, typically two nonmetals, lower electronegativity difference.
Exceptions: High oxidation states in metals can lead to covalent character; physical properties (e.g., melting point) also provide clues.
Example: LiF is ionic, HF is polar covalent, F2 is nonpolar covalent.
Additional info: These concepts are foundational for understanding molecular structure, reactivity, and properties in general chemistry.
