 Back
BackCalculations Involving Weak Acids and Polyprotic Acids
Study Guide - Smart Notes

Calculations Involving Weak Acids
Introduction to Weak Acids and Equilibrium
Weak acids are substances that partially dissociate in aqueous solution, establishing a dynamic equilibrium between the undissociated acid and its ions. Understanding the behavior of weak acids is essential for calculating the pH of their solutions and for determining their acid dissociation constants (Ka).
Weak Acid Dissociation: The general equation for the dissociation of a weak acid (HA) in water is:
Acid Dissociation Constant (Ka): The equilibrium constant for this reaction is called the acid dissociation constant, Ka:
pH Calculation: To find the pH of a weak acid solution, determine the equilibrium concentration of H+ (or H3O+).
Example: Calculating pH of a Weak Acid Solution
Given: The Ka for niacin is . What is the pH of a 0.010 M solution of niacin?
Set up the equilibrium expression for niacin (HA):
Initial: [HA] = 0.010 M, [H+] = 0, [A-] = 0 Change: [HA] = -x, [H+] = +x, [A-] = +x Equilibrium: [HA] = 0.010 - x, [H+] = x, [A-] = x
Assume x << 0.010, so Solve for x (which is [H+]), then calculate pH:
Example Application: This method is used for any weak acid where the initial concentration and Ka are known.
Calculating Ka from pH
It is also possible to determine the acid dissociation constant if the equilibrium concentrations are known. For example, if a 0.10 M solution of formic acid (HCOOH) has a pH of 2.38 at 25°C, the [H+] can be calculated from the pH, and then Ka can be determined using the equilibrium expression.
Step 1: Calculate [H+] from pH:
Step 2: Use the equilibrium table to find [A-] and [HA] at equilibrium.
Step 3: Substitute values into the Ka expression.
Polyprotic Acids
Definition and Properties
Polyprotic acids are acids that can donate more than one proton (H+) per molecule. Each dissociation step has its own acid dissociation constant (Ka1, Ka2, Ka3, etc.), and the first dissociation is usually the strongest.
Examples: Sulfuric acid (H2SO4), phosphoric acid (H3PO4), and carbonic acid (H2CO3).
Stepwise Dissociation: Each proton is lost in a separate step, each with a unique Ka value.
Example: pH Calculation for Polyprotic Acids
To calculate the pH of a solution of a polyprotic acid, consider the first dissociation step as the major contributor to [H+], especially if Ka1 >> Ka2. For example, for a 0.0037 M solution of H2CO3 (carbonic acid), use Ka1 for the initial calculation.
Acid-Dissociation Constants of Common Polyprotic Acids
The table below summarizes the acid-dissociation constants for several common polyprotic acids. These values are essential for solving equilibrium and pH problems involving these acids.
Name | Formula | Ka1 | Ka2 | Ka3 |
|---|---|---|---|---|
Ascorbic | H2C6H6O6 | 8.0 × 10–5 | 1.6 × 10–12 | — |
Carbonic | H2CO3 | 4.3 × 10–7 | 5.6 × 10–11 | — |
Citric | H3C6H5O7 | 7.4 × 10–4 | 1.7 × 10–5 | 4.0 × 10–7 |
Oxalic | HOOC—COOH | 5.9 × 10–2 | 6.4 × 10–5 | — |
Phosphoric | H3PO4 | 7.5 × 10–3 | 6.2 × 10–8 | 4.2 × 10–13 |
Sulfurous | H2SO3 | 1.7 × 10–2 | 6.4 × 10–8 | — |
Sulfuric | H2SO4 | Large | 1.2 × 10–2 | — |
Tartaric | C4H6O6(COOH)2 | 1.0 × 10–3 | 4.6 × 10–5 | — |
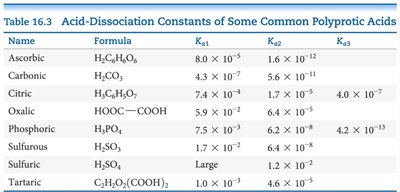
Additional info: For polyprotic acids, the first dissociation constant (Ka1) is typically much larger than the subsequent ones, meaning the first proton is lost much more easily than the second or third. In most pH calculations, only the first dissociation is considered unless high accuracy is required or the acid is very dilute.
