 Back
BackCh. 16
Study Guide - Smart Notes

Carboxylic Acids
Structure and Nomenclature
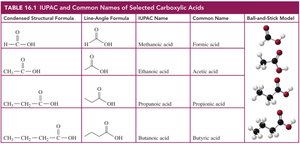
Carboxylic acids are organic compounds containing a carboxyl group (–COOH), which consists of a carbonyl group (C=O) attached to a hydroxyl group (–OH). They are named systematically using IUPAC rules, but many also have common names derived from historical sources.
Carboxyl Group: The functional group –COOH is characteristic of carboxylic acids.
IUPAC Naming: The parent alkane name is modified by replacing the final -e with -oic acid.
Common Names: Many carboxylic acids are known by traditional names (e.g., formic acid, acetic acid).
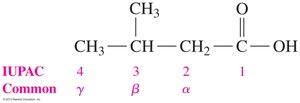
Numbering: In IUPAC, the carboxyl carbon is always carbon 1. In common naming, Greek letters (α, β, γ) are used for carbons adjacent to the carboxyl group.
Examples and Applications
Formic Acid: Found in ant stings and causes skin irritation.
Acetic Acid: The main component of vinegar.


Carboxylic Acids with Aromatic Rings
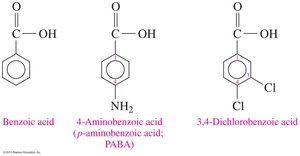
Carboxylic acids can be attached to aromatic rings, such as benzoic acid. Substituents on the ring are named to give the lowest possible numbers, and prefixes like ortho (o-), meta (m-), and para (p-) indicate relative positions.
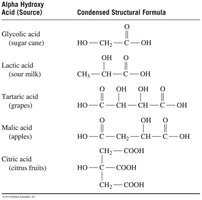
Alpha Hydroxy Acids (AHAs)
Alpha hydroxy acids are carboxylic acids with a hydroxyl group on the alpha carbon. They occur naturally in fruits, milk, and sugar cane, and are used in skin care products for exfoliation and rejuvenation.

Practice: Naming and Drawing Carboxylic Acids
2-Methylbutanoic acid and 3-chlorobenzoic acid are examples of substituted carboxylic acids.
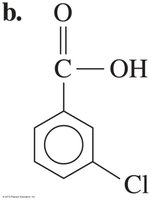
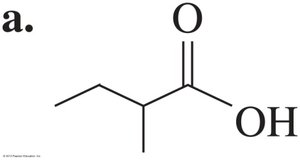
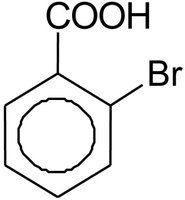
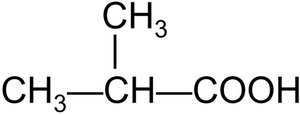
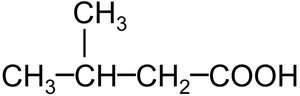

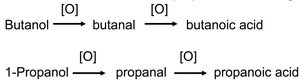
Preparation of Carboxylic Acids
Carboxylic acids are commonly prepared by the oxidation of primary alcohols or aldehydes. For example, ethanol is oxidized to ethanoic acid (acetic acid).
General Reaction:

Properties of Carboxylic Acids
Polarity and Boiling Points
Carboxylic acids are strongly polar due to the presence of both a carbonyl and a hydroxyl group. They form hydrogen-bonded dimers, resulting in higher boiling points than alcohols, ketones, or aldehydes of similar mass.
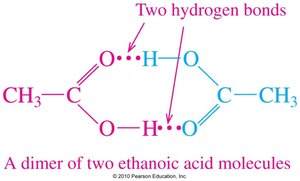
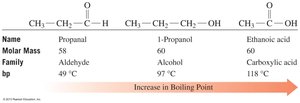
Solubility in Water
Carboxylic acids with 1–4 carbon atoms are very soluble in water due to their ability to form hydrogen bonds with water molecules. Solubility decreases as the hydrocarbon chain length increases.
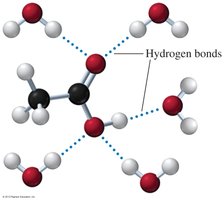
Acidity and Ionization
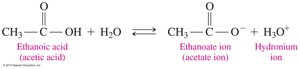
Carboxylic acids are weak acids that partially ionize in water to produce carboxylate ions and hydronium ions. Their acid dissociation constants (Ka) are small, indicating weak acidity.
General Ionization Reaction:
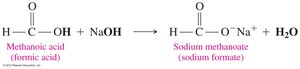
Neutralization Reactions
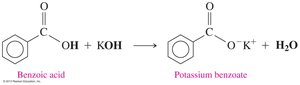
Carboxylic acids react with strong bases to form carboxylate salts and water. These salts are often used as preservatives and flavor enhancers in food.
General Neutralization Reaction:

Esters
Structure and Naming
Esters are derived from carboxylic acids by replacing the hydrogen in the –OH group with an alkyl group. They are named by stating the alkyl group from the alcohol first, followed by the acid part with an -ate ending.
General Structure: RCOOR'
Naming Example: Methyl ethanoate (from methanol and ethanoic acid)
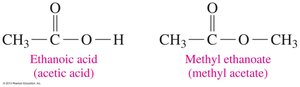
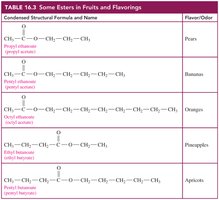
Esters in Nature and Industry
Esters are responsible for the pleasant fragrances and flavors of many fruits and flowers. They are widely used in food flavorings and perfumes.

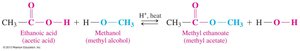
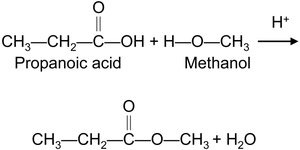
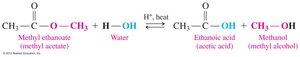
Esterification
Esterification is the reaction of a carboxylic acid with an alcohol in the presence of an acid catalyst to produce an ester and water.
General Reaction:
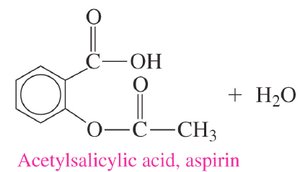
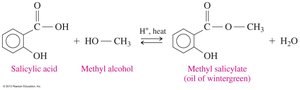
Important Esters: Aspirin and Oil of Wintergreen
Aspirin (acetylsalicylic acid): An ester of salicylic acid and acetic acid, used as an analgesic and anti-inflammatory drug.
Methyl salicylate (oil of wintergreen): An ester of salicylic acid and methanol, used in topical pain relief products.
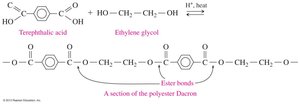
Polyesters
Polyesters are polymers formed by the reaction of dicarboxylic acids and diols, resulting in long chains with repeating ester bonds. Dacron is a common example of a synthetic polyester fiber.
Properties and Reactions of Esters
Physical Properties
Boiling Points: Esters have higher boiling points than alkanes but lower than alcohols and carboxylic acids of similar mass, due to their inability to form hydrogen bonds with each other.
Solubility: Esters with 1–5 carbon atoms are soluble in water; solubility decreases with increasing chain length.

Hydrolysis of Esters
Acid Hydrolysis: Esters react with water and an acid catalyst to produce a carboxylic acid and an alcohol.
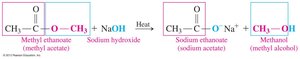
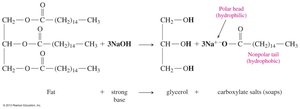
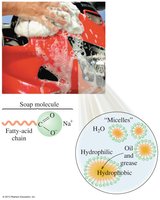
Base Hydrolysis (Saponification): Esters react with a strong base to produce a carboxylate salt and an alcohol. This reaction is the basis for soap production.
Practice: Naming and Drawing Esters
Example: Methyl butanoate and ethyl propanoate are named based on their alcohol and acid components.
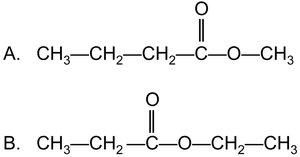
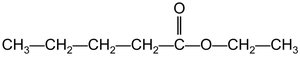
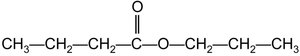
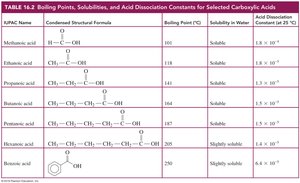
Summary Table: Carboxylic Acids and Esters
Key properties, boiling points, and solubilities of selected carboxylic acids and esters are summarized in the following tables:


Additional info: This guide covers the structure, nomenclature, properties, and reactions of carboxylic acids and esters, including their importance in biological and industrial contexts. Practice problems and tables are included to reinforce key concepts.
