 Back
BackCh.13 - Solutions: Properties, Concentrations, and Colligative Effects
Study Guide - Smart Notes

Solutions: Solubility and Intermolecular Forces
Definition and Types of Solutions
Solutions are homogeneous mixtures formed when a solute dissolves in a solvent. The solubility of a substance is a physical property describing its ability to become miscible in a solvent. If a solute cannot dissolve, a heterogeneous mixture results.
Homogeneous mixture: Uniform composition throughout; solute is dissolved.
Heterogeneous mixture: Non-uniform; solute is not dissolved.
"Likes dissolve likes": Substances with similar polarity and intermolecular forces tend to form solutions together.
Types of Intermolecular Forces
Intermolecular forces determine solubility and miscibility. The main types are:
Ion-Dipole: Major force in ionic compounds dissolved in polar solvents.
Hydrogen Bonding: Occurs in compounds with hydrogen directly bonded to fluorine, oxygen, or nitrogen.
Dipole-Dipole: Present in polar covalent compounds.
London Dispersion (van der Waals): Found in all molecules, especially nonpolar covalent compounds.
Solubility of Gases in Water
The solubility of nonpolar gases in water varies and is influenced by intermolecular forces and polarity.
Example: N2 and O2 have different solubilities; F2 is expected to have a solubility value between those of N2 and O2.
Types of Aqueous Solutions
Saturated, Unsaturated, and Supersaturated Solutions
When solutes dissolve in water, equilibrium is established between dissolution and recrystallization. The amount of dissolved solute at equilibrium defines the solution type:
Saturated: Maximum solute dissolved; at equilibrium concentration.
Unsaturated: Less solute than equilibrium; more can be dissolved.
Supersaturated: More solute than equilibrium; unstable and may precipitate.
Concentration Units
Molality (m)
Molality is a concentration unit independent of temperature, defined as moles of solute per kilogram of solvent.
Formula:
Example: Calculating molality for a solution with known mass of solute and solvent.
Osmolality
Osmolality measures the total number of dissolved particles (ions) in a solution. For ionic compounds, count the number of ions produced and multiply by the molality.
Formula:
Example: For NaCl, which dissociates into Na+ and Cl-, osmolality is 2 × molality.
Parts Per Million (ppm) and Parts Per Billion (ppb)
Extremely dilute concentrations are expressed in ppm or ppb, which can be mass or volume based.
ppm (mass):
ppb (mass):
In aqueous solutions: 1 ppm = 1 mg/L; 1 ppb = 1 µg/L.
Mole Fraction (X)
Mole fraction represents the ratio of moles of solute to total moles in the solution.
Formula:
Mass Percent
Mass percent is the mass of solute per mass of solution, multiplied by 100.
Formula:
Colligative Properties
Overview of Colligative Properties
Colligative properties depend on the number of solute particles, not their identity. The four main colligative properties are:
Boiling Point Elevation
Freezing Point Depression
Vapor Pressure Lowering
Osmotic Pressure
Boiling Point Elevation
Adding a solute increases the boiling point of a solvent. The change is calculated using:
Formula:
Variables: = van't Hoff factor (number of particles), = boiling point constant, = molality
Freezing Point Depression
Adding a solute decreases the freezing point of a solvent. The change is calculated using:
Formula:
Variables: = van't Hoff factor, = freezing point constant, = molality
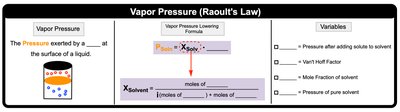
Vapor Pressure Lowering (Raoult's Law)
The vapor pressure of a solution is lower than that of the pure solvent. Raoult's Law describes this effect:
Formula:
Variables: = mole fraction of solvent, = vapor pressure of pure solvent
Osmosis and Osmotic Pressure
Osmosis is the net movement of solvent across a semipermeable membrane from low to high solute concentration. Osmotic pressure is the force driving this movement.
Formula:
Variables: = osmotic pressure, = van't Hoff factor, = molarity, = gas constant, = temperature (K)
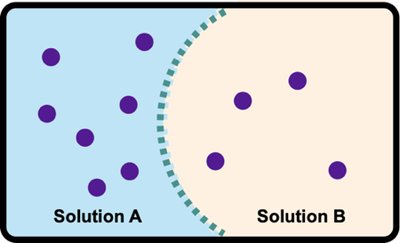
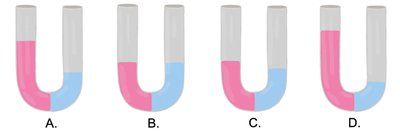
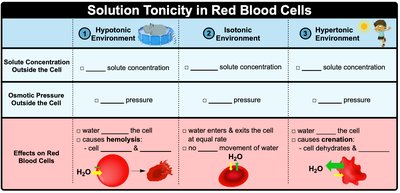
Tonicity and Effects on Cells
Hypotonic, Isotonic, and Hypertonic Solutions
Tonicity describes the relative solute concentration of solutions separated by a membrane. It affects cell volume and osmotic pressure:
Hypotonic: Lower solute concentration outside the cell; water enters, causing cell swelling (hemolysis).
Isotonic: Equal solute concentration; no net movement of water.
Hypertonic: Higher solute concentration outside; water leaves, causing cell shrinkage (crenation).

Summary Table: Types of Aqueous Solutions
The following table summarizes the types of aqueous solutions:
Type of Solution | Amount of Dissolved Solute | Equilibrium Concentration | Example |
|---|---|---|---|
Saturated | Maximum solute dissolved | At equilibrium | 20 g/100 mL water |
Unsaturated | Less solute can be dissolved | Below equilibrium | 10 g/100 mL water |
Supersaturated | More solute dissolved | Above equilibrium | 40 g/100 mL water |
Additional info: All equations are provided in LaTeX format for clarity and exam preparation.
