 Back
BackChapter 1 & 2 Study Notes: Matter, Measurement, Problem Solving, Atoms & Elements
Study Guide - Smart Notes

Matter, Measurement, and Problem Solving
Atoms and Molecules: The Foundation of Chemistry
The properties of matter are determined by the properties of molecules and atoms. Understanding matter at the molecular level allows chemists to control and manipulate substances for various applications.
Atoms are the fundamental building blocks of matter. Free atoms are rare; they usually bind together in specific arrangements to form molecules.
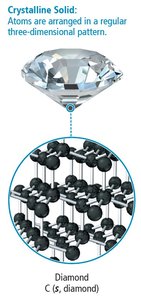
Small differences in atomic or molecular structure can result in large differences in properties (e.g., graphite vs. diamond).
Chemistry is the science that seeks to understand the behavior of matter by studying atoms and molecules.
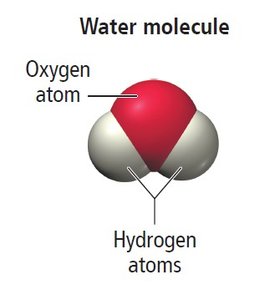
Example: Water molecules (H2O) consist of two hydrogen atoms and one oxygen atom held together by chemical bonds.

The Scientific Method
The scientific method is an empirical approach based on observation and experimentation. It is used to understand nature and its behavior.
Observation: Gathering data about characteristics or behavior of nature.
Hypothesis: A tentative explanation of observations; must be falsifiable.
Law: A brief statement summarizing past observations and predicting future ones (e.g., Law of Conservation of Mass).
Theory: A model explaining why nature behaves as it does; validated by experiments but never conclusively proven.

The Classification of Matter
States of Matter
Matter can exist as a solid, liquid, or gas, each with distinct properties due to differences in atomic or molecular structure.
Solid: Fixed volume and shape; atoms/molecules are closely packed and vibrate in place.
Liquid: Fixed volume but no fixed shape; atoms/molecules are close but can move past each other.
Gas: No fixed volume or shape; atoms/molecules are far apart and compressible.


Classification by Composition
Matter is classified as pure substances or mixtures based on its composition.
Pure Substance: Made of only one component; composition is invariant.
Mixture: Composed of two or more components; proportions can vary.
Elements: Cannot be chemically broken down; basic building blocks.
Compounds: Composed of two or more elements in fixed proportions.
Heterogeneous Mixture: Composition varies throughout (e.g., sand and salt).
Homogeneous Mixture: Uniform composition (e.g., sweetened tea).
Separating Mixtures
Mixtures can be separated using physical techniques based on differences in properties.
Decanting: Pouring off liquid from a solid-liquid mixture.
Distillation: Separating liquids by boiling off the more volatile component.
Filtration: Separating solids from liquids using filter paper.


Physical and Chemical Changes
Changes in matter are classified as physical or chemical.
Physical Change: Alters state or appearance, not composition (e.g., boiling water).
Chemical Change: Alters composition; atoms rearrange to form new substances (e.g., rusting iron).
Physical Properties: Observed without changing composition (e.g., odor, color, melting point).
Chemical Properties: Observed only by changing composition (e.g., flammability, acidity).
Energy in Chemistry
Energy is the capacity to do work and is conserved in physical and chemical changes.
Kinetic Energy: Energy of motion.
Potential Energy: Energy due to position or composition.
Thermal Energy: Energy associated with temperature; a type of kinetic energy.
Law of Conservation of Energy: Energy is neither created nor destroyed.
Units of Measurement
Chemistry relies on standard units for measurement, primarily the International System of Units (SI).
Length: Meter (m)
Mass: Kilogram (kg)
Time: Second (s)
Temperature: Kelvin (K)
Amount: Mole (mol)
Quantity | Unit | Symbol |
|---|---|---|
Length | Meter | m |
Mass | Kilogram | kg |
Time | Second | s |
Temperature | Kelvin | K |
Amount | Mole | mol |
Prefix Multipliers
SI units use prefix multipliers to represent powers of ten.
Prefix | Symbol | Multiplier |
|---|---|---|
kilo | k | 103 |
centi | c | 10-2 |
milli | m | 10-3 |
micro | μ | 10-6 |
nano | n | 10-9 |
mega | M | 106 |
Derived Units: Volume and Density
Volume and density are derived units in chemistry.
Volume:
Density:
Substance | Density (g/cm3) |
|---|---|
Water | 1.00 |
Ice | 0.917 |
Aluminum | 2.70 |
Iron | 7.86 |
Gold | 19.3 |
Intensive and Extensive Properties
Intensive Property: Independent of amount (e.g., density).
Extensive Property: Dependent on amount (e.g., mass).
Significant Figures and Measurement Reliability
Significant figures reflect the precision of measurements. Rules for counting and using significant figures ensure accuracy in calculations.
All nonzero digits are significant.
Interior zeroes are significant.
Leading zeroes are not significant.
Trailing zeroes are significant if after a decimal point.
Multiplication/Division: Result has same number of significant figures as the factor with the fewest.
Addition/Subtraction: Result has same number of decimal places as the quantity with the fewest.
Precision and Accuracy
Accuracy: Closeness to the true value.
Precision: Consistency among measurements.
Random Error: Equal probability of being too high or low.
Systematic Error: Tends toward being consistently high or low.
Dimensional Analysis
Dimensional analysis uses units as a guide to solve conversion problems.
Conversion Factor: Fractional quantity from a unit equation.
General Form:
Atoms and Elements
Atomic Theory and Laws
Atomic theory explains the fundamental nature of matter and is supported by several key laws.
Law of Conservation of Mass: Matter is neither created nor destroyed in a chemical reaction.
Law of Definite Proportions: All samples of a compound have the same proportions of constituent elements.
Law of Multiple Proportions: When two elements form different compounds, the mass ratios of one element to a fixed mass of the other are small whole numbers.
Dalton's Atomic Theory
Elements are composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties.
Atoms combine in simple, whole-number ratios to form compounds.
Atoms of one element cannot change into atoms of another element.
Discovery of Subatomic Particles
Electron: Discovered by J.J. Thomson via cathode ray experiments; negatively charged, low mass.
Proton: Positively charged particle in the nucleus.
Neutron: Neutral particle in the nucleus; similar mass to proton.
Particle | Mass (kg) | Charge |
|---|---|---|
Proton | 1.67262 × 10-27 | +1 |
Neutron | 1.67493 × 10-27 | 0 |
Electron | 0.00091 × 10-27 | -1 |
Structure of the Atom
Most of the atom's mass and all positive charge are in the nucleus.
Electrons are dispersed in empty space around the nucleus.
Atoms are electrically neutral: number of protons equals number of electrons.
Elements and the Periodic Table
Each element is defined by its atomic number (number of protons).
Periodic table organizes elements by increasing atomic number and groups elements with similar properties.
Elements are classified as metals, nonmetals, or metalloids.
Isotopes
Atoms of the same element with different numbers of neutrons are called isotopes.
Mass number (A) = number of protons + number of neutrons.
Natural abundance refers to the relative percentage of each isotope in a sample.
Isotope | Protons | Neutrons | Mass Number | Natural Abundance (%) |
|---|---|---|---|---|
Ne-20 | 10 | 10 | 20 | 90.48 |
Ne-21 | 10 | 11 | 21 | 0.27 |
Ne-22 | 10 | 12 | 22 | 9.25 |
Ions
Atoms can lose or gain electrons to form ions.
Cations: Positively charged (lost electrons).
Anions: Negatively charged (gained electrons).
Atomic Mass and the Mole
Atomic mass is the weighted average of the masses of an element's isotopes.
The mole (mol) is a counting unit: 1 mol = particles (Avogadro's number).
Molar mass (g/mol) is numerically equal to atomic mass (amu).
To convert between mass, moles, and number of atoms:
Mass (g) → Moles (mol) → Number of atoms
Example: To find the number of atoms in 12.01 g of carbon:
12.01 g C → 1 mol C → C atoms
----------------------------------------
