Back
BackChapter 1: Chemistry and Measurements – Study Notes
Study Guide - Smart Notes

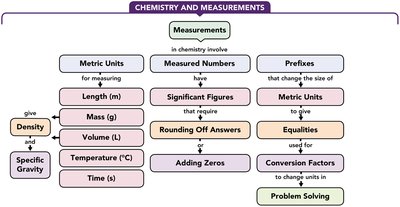
Chemistry and Measurements
Introduction to Chemistry and Measurements
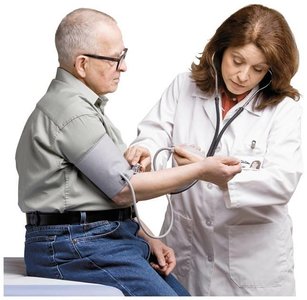
Chemistry is the study of matter, its properties, and the changes it undergoes. Accurate measurement is fundamental in chemistry, as it allows scientists to quantify observations and communicate results effectively. Measurements are essential in both laboratory and clinical settings, such as monitoring patient health and administering medications.
Units of Measurement
Metric and SI Units
Scientists use the metric system and the International System of Units (SI) as standardized methods for measuring physical quantities. These systems provide consistency in scientific communication worldwide.
Length: meter (m)
Volume: liter (L) in metric, cubic meter (m3) in SI
Mass: gram (g) in metric, kilogram (kg) in SI
Temperature: degree Celsius (°C) in metric, kelvin (K) in SI
Time: second (s)

Measurement | Metric Unit | SI Unit |
|---|---|---|
Volume | liter (L) | cubic meter (m3) |
Length | meter (m) | meter (m) |
Mass | gram (g) | kilogram (kg) |
Temperature | degree Celsius (°C) | kelvin (K) |
Time | second (s) | second (s) |
Measured Numbers and Significant Figures
Measured vs. Exact Numbers
Measured numbers are obtained through measurement and contain some degree of uncertainty. Exact numbers are obtained by counting or by definition and have no uncertainty.
Measured Number: Determined using instruments (e.g., length, mass, temperature).
Exact Number: Obtained by counting (e.g., 3 apples) or defined relationships (e.g., 1 ft = 12 in.).
Reading Measurements and Estimation
When measuring, the last digit is always an estimate, reflecting the uncertainty in the measurement. For example, when using a ruler, the value between the smallest marks is estimated.

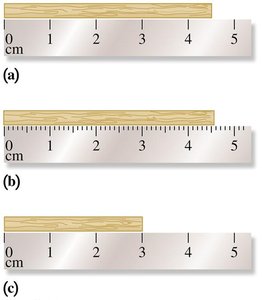
Significant Figures (SFs)
Significant figures reflect the precision of a measured number. They include all certain digits plus one estimated digit.
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Zeros at the end of a decimal number are significant.
Leading zeros are not significant.
Placeholder zeros in large numbers without a decimal are not significant.
Rule | Example | SFs |
|---|---|---|
Nonzero digits | 4.5 g | 2 |
Zeros between digits | 205 °C | 3 |
Zeros at end of decimal | 16.00 mL | 4 |
Leading zeros | 0.0004 s | 1 |
Placeholder zeros | 850 000 m | 2 |
Scientific Notation and Significant Zeros
Scientific notation is used to clearly indicate significant zeros in large or small numbers. For example, m has two significant figures.
Rounding Off and Calculations with Significant Figures
When performing calculations, the number of significant figures in the result depends on the operation:
Multiplication/Division: The result has the same number of SFs as the measurement with the fewest SFs.
Addition/Subtraction: The result has the same number of decimal places as the measurement with the fewest decimal places.

Prefixes and Equalities
Metric Prefixes
Prefixes are used to express multiples or fractions of units in the metric system. They make it easier to represent very large or very small quantities.
Prefix | Symbol | Value | Scientific Notation |
|---|---|---|---|
kilo | k | 1,000 | |
centi | c | 0.01 | |
milli | m | 0.001 | |
micro | μ (mc in medicine) | 0.000001 | |
nano | n | 0.000000001 |
Equalities and Conversion Factors
Equalities express the relationship between two units that describe the same quantity. Conversion factors are fractions derived from equalities and are used to convert between units.
Example: gives conversion factors and .


Temperature
Temperature Scales
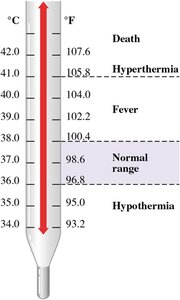
Temperature is a measure of how hot or cold an object is. The three main temperature scales are Celsius (°C), Fahrenheit (°F), and Kelvin (K).
Celsius: Freezing point of water is 0°C, boiling point is 100°C.
Fahrenheit: Freezing point of water is 32°F, boiling point is 212°F.
Kelvin: Absolute zero is 0 K (no negative values), same unit size as Celsius.

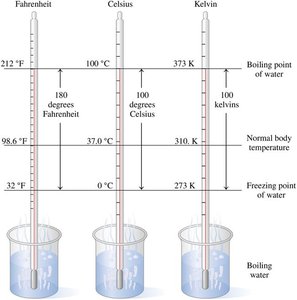
Temperature Conversions
Celsius to Fahrenheit:
Fahrenheit to Celsius:
Celsius to Kelvin:
Problem Solving Using Unit Conversion
Unit Conversion Method
To solve problems involving unit conversions, identify the given quantity and units, determine the needed units, and use appropriate conversion factors to reach the answer. Multiple conversion factors may be needed for complex problems.

Chemistry Link to Health: Toxicology
LD50 and Toxicity
The LD50 (lethal dose, 50%) is the amount of a substance that causes death in 50% of test animals, usually expressed in mg/kg of body mass. Lower LD50 values indicate higher toxicity.

Substance | LD50 (mg/kg) |
|---|---|
Table sugar | 29,700 |
Boric acid | 5,140 |
Baking soda | 4,220 |
Table salt | 3,300 |
Ethanol | 2,080 |
Aspirin | 1,100 |
Codeine | 800 |
Oxycodone | 480 |
Caffeine | 192 |
DDT | 113 |
Cocaine (injected) | 95 |
Dichlorvos | 56 |
Ricin | 30 |
Sodium cyanide | 6 |
Parathion | 3 |
Density
Definition and Calculation
Density is the ratio of mass to volume and is a characteristic property of substances. It is used to identify substances and determine whether objects will float or sink in a fluid.
Formula:
Units: g/mL (solids and liquids), g/L (gases)

Substance | Density (g/mL or g/L) |
|---|---|
Cork | 0.26 |
Ice | 0.92 |
Water | 1.00 |
Aluminum | 2.70 |
Lead | 11.3 |
Air (dry, g/L) | 1.29 |
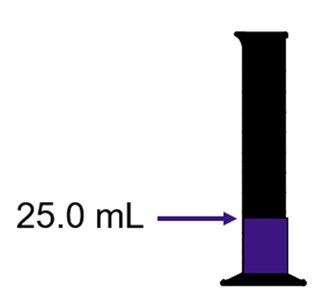
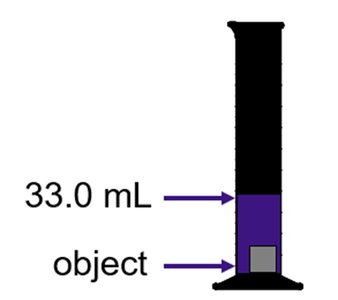
Volume Displacement Method
The volume of an irregular solid can be determined by the amount of water it displaces. The density is then calculated using the measured mass and displaced volume.

Concept Map: Chemistry and Measurements