 Back
BackChapter 1: Chemistry in Our Lives – Foundations and Essential Skills
Study Guide - Smart Notes

Chapter 1: Chemistry in Our Lives
1.1 Chemistry and Chemicals
Chemistry is the scientific study of matter, focusing on its composition, structure, properties, and the changes it undergoes during reactions. Chemistry is present in everyday activities such as cooking, cleaning, and even in the functioning of our bodies.
Composition: What substances are made of (elements, compounds).
Structure: How atoms and molecules are arranged.
Properties: Characteristics such as color, odor, melting point, and reactivity.
Reactions: How substances interact and transform into new substances.
Chemicals are substances with a definite composition and set of properties. Everyday items like soaps, lotions, toothpaste, and clothes are made of chemicals.
Branches of Chemistry: General, Organic, Biochemistry, Geochemistry, Physical Chemistry.
Applications: Chemists develop new products and pharmaceuticals, and chemistry is essential in fields such as medicine, environmental science, and materials engineering.

Chemical | Function |
|---|---|
Calcium carbonate | Used as an abrasive to remove plaque |
Sorbitol | Prevents loss of water and hardening of toothpaste |
Sodium lauryl sulfate | Used to loosen plaque |
Titanium dioxide | Makes toothpaste white and opaque |
Triclosan | Inhibits bacteria that cause plaque and gum disease |
Sodium fluorophosphate | Prevents cavities by strengthening tooth enamel with fluoride |
Methyl salicylate | Gives toothpaste a pleasant wintergreen flavor |

In the kitchen, chemistry is seen in antioxidants in food, metal alloys in utensils, and plastics in containers.

1.2 Scientific Method: Thinking Like a Scientist
The scientific method is a systematic approach used by scientists to explore observations, develop hypotheses, conduct experiments, and formulate theories or laws.
Observation: Gathering facts by observing and measuring events in nature.
Hypothesis: A tentative explanation for the observations.
Experiment: Procedures to test the hypothesis.
Theory: A model that explains observations and experimental results.
Law: A statement that summarizes observed behavior.
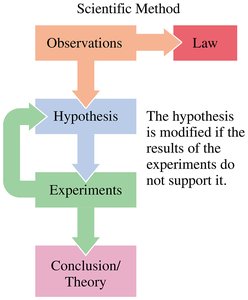
Example: If you sneeze around cats, you might hypothesize you are allergic. Testing this by visiting homes with and without cats and observing your symptoms is an application of the scientific method.
1.3 Learning Chemistry: A Study Plan
Success in chemistry requires active learning and effective study strategies. The textbook provides features such as chapter outlines, glossaries, sample problems, and concept checks to support learning.
Review chapter topics before class.
Engage with sample problems and check solutions.
Participate in study groups and attend office hours.
Use chapter reviews, concept maps, and practice problems for reinforcement.
Step | Description |
|---|---|
1 | Read each Learning Goal for an overview of the material. |
2 | Form a question from the section title. |
3 | Read the section, seeking answers to your question. |
4 | Self-test with Sample Problems and Study Checks. |
5 | Complete Questions and Problems, check answers for odd-numbered problems. |
6 | Repeat for the next section. |

1.4 Key Math Skills for Chemistry
Mathematical skills are essential for solving chemistry problems. Key concepts include place values, positive and negative numbers, percentages, solving equations, and interpreting graphs.
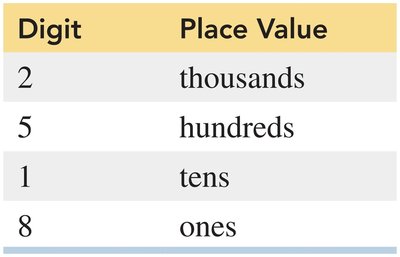
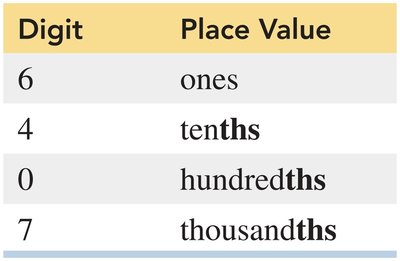
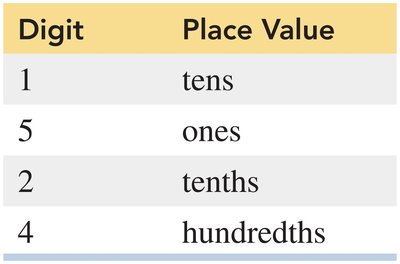
Place Values
Each digit in a number has a specific place value (e.g., thousands, hundreds, tens, ones).
Positive and Negative Numbers
Multiplication/Division: Same signs yield positive results; different signs yield negative results.
Addition: Add values, keep the sign of the larger number.
Subtraction: Change the sign of the number being subtracted, then add.
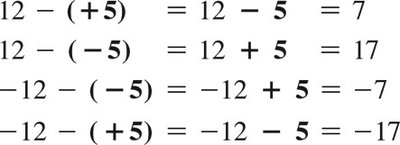
Percentages
Percent (%) = (Part / Whole) × 100%
Example: If a tablet contains 325 mg aspirin in a 545 mg tablet, % aspirin = (325/545) × 100% ≈ 59.6%
Solving Equations
Isolate the variable by performing the same operation on both sides.
Example: To solve , subtract 8 from both sides, then divide by 2: .
For temperature conversion: ; to solve for :
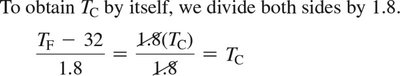
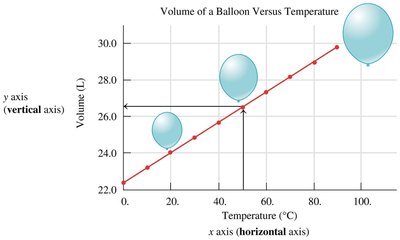
Interpreting Line Graphs
Line graphs show relationships between two variables (e.g., volume vs. temperature).
Direct relationship: As one variable increases, so does the other.
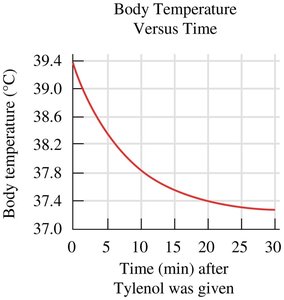
Example: A graph of body temperature versus time after medication shows how temperature decreases over time.
1.5 Scientific Notation
Scientific notation is used to express very large or very small numbers in the form , where and is an integer.
Example: 0.000008 m = m; 100,000 hairs = hairs.
To convert to scientific notation, move the decimal to create a coefficient between 1 and 10, and count the places moved for the exponent.
To convert back, move the decimal according to the exponent.
Standard Format | Scientific Notation |
|---|---|
12,800,000 m | 1.28 × 107 m |
68 kg | 6.8 × 101 kg |
0.0000003 cm | 3 × 10−7 cm |
Calculator Tip: Use the EE or EXP key to enter numbers in scientific notation.
