Back
BackChapter 1: Introduction to Matter, Energy, and Measurement
Study Guide - Smart Notes

Introduction to Chemistry
What is Chemistry?
Chemistry is the scientific study of matter, its properties, and the changes it undergoes. It is a foundational science that informs our understanding of biology, physics, environmental science, and many other fields.
Matter: Anything that has mass and occupies space.
Properties: Characteristics that define matter, such as color, density, and boiling point.
Changes: Transformations matter can undergo, including physical and chemical changes.
Classifications of Matter
States of Matter
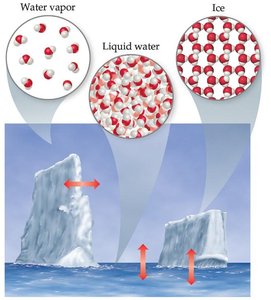
Matter exists in three primary states: solid, liquid, and gas. Each state is characterized by distinct physical properties and molecular arrangements.
Solid: Definite shape and volume; particles are closely packed in a fixed arrangement.
Liquid: Definite volume but no definite shape; particles are close but can move past one another.
Gas: No definite shape or volume; particles are far apart and move freely.
Example: Water can exist as ice (solid), liquid water, or water vapor (gas).
Classification of Matter as Substances
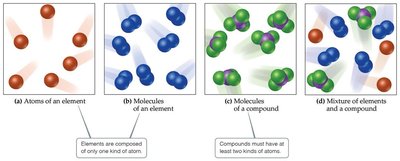
Matter can be classified as substances, which have distinct properties and a composition that does not vary from sample to sample. Substances are further divided into elements and compounds.
Element: A substance that cannot be decomposed into simpler substances. It consists of only one kind of atom.
Compound: A substance composed of two or more elements chemically combined in fixed proportions. It can be decomposed into simpler substances.
Classification of Matter Based on Composition
Atoms are the fundamental building blocks of matter. Each element is made of a unique kind of atom, while compounds are made of atoms from two or more different elements. Groups of atoms bonded together are called molecules.
Molecule: A group of atoms bonded together, representing the smallest unit of a compound.
Example: Water (H2O) is a molecule composed of hydrogen and oxygen atoms.
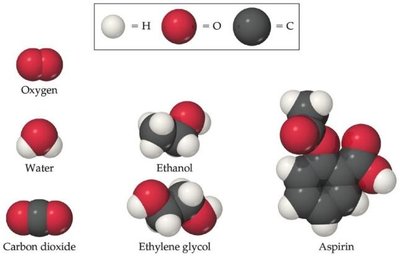
Representing Elements
Chemists use symbols to represent elements. These symbols are one or two letters, with the first letter always capitalized. Some symbols are derived from Latin or Greek names.
Example: Carbon (C), Hydrogen (H), Sodium (Na, from 'natrium'), Iron (Fe, from 'ferrum').
There are currently 118 named elements.
Elements and Composition
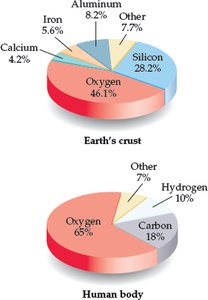
Only a few elements make up most of the Earth's crust and the human body by mass. Oxygen is particularly abundant in both.
Earth's crust: Oxygen, Silicon, Aluminum, Iron, Calcium.
Human body: Oxygen, Carbon, Hydrogen.
Compounds and Composition
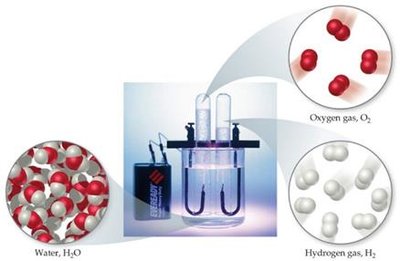
Compounds have a definite composition, meaning the relative number of atoms of each element is the same in any sample. This is known as the Law of Constant Composition (or Law of Definite Proportions).
Example: Water always contains two hydrogen atoms and one oxygen atom.
Equation:
Mixtures
Mixtures are combinations of two or more substances that retain their individual properties. Mixtures can be classified as heterogeneous or homogeneous based on their uniformity.
Heterogeneous mixture: Composition varies throughout the sample (e.g., granite).
Homogeneous mixture (solution): Composition is uniform throughout the sample (e.g., salt water).

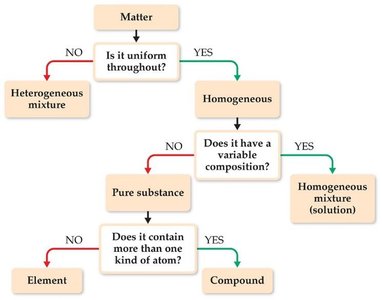
Making a Decision: Classification Scheme
A flowchart can help determine how to classify any type of matter based on its uniformity and composition.
Is the matter uniform throughout?
Does it have variable composition?
Does it contain more than one kind of atom?