 Back
BackCHM 113 ch1
Study Guide - Smart Notes

Introduction to Matter, Energy, and Measurement
Overview
This chapter introduces fundamental concepts in chemistry, including the classification of matter, properties and changes of substances, measurement techniques, and the importance of quantitative analysis. Understanding these basics is essential for further study in chemistry.
Methods of Classification of Matter
States of Matter
Matter exists in three primary states: solid, liquid, and gas. Each state has distinct physical characteristics based on the arrangement and movement of particles.
Solid: Definite shape and volume; particles are closely packed and vibrate in place.
Liquid: Definite volume but no definite shape; particles are less tightly packed and can move past each other.
Gas: No definite shape or volume; particles are far apart and move freely.
Example: Ice (solid), liquid water, and water vapor (gas) are all forms of H2O.

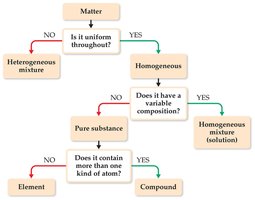
Classification Based on Composition
Matter can be classified as a pure substance or a mixture. Pure substances include elements and compounds, while mixtures can be homogeneous or heterogeneous.
Element: Cannot be decomposed into simpler substances; made of one type of atom.
Compound: Can be decomposed; made of two or more elements chemically combined.
Homogeneous mixture (solution): Uniform composition throughout (e.g., salt water).
Heterogeneous mixture: Non-uniform composition (e.g., salad dressing).
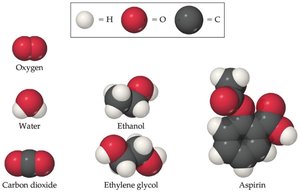
Atoms, Elements, and Compounds
Atoms are the fundamental building blocks of matter. Each element consists of unique atoms, and compounds are formed from atoms of different elements.
Molecule: A group of atoms bonded together, representing the smallest unit of a compound.
Example: Water (H2O) is a molecule made from hydrogen and oxygen atoms.
Properties of Matter
Physical and Chemical Properties
Properties of matter are categorized as physical or chemical.
Physical properties: Can be observed without changing the substance (e.g., color, density, melting point).
Chemical properties: Can only be observed when the substance undergoes a chemical change (e.g., flammability).
Intensive vs. Extensive Properties
Intensive properties: Independent of the amount of substance (e.g., density, boiling point).
Extensive properties: Depend on the amount of substance (e.g., mass, volume).
Physical and Chemical Changes
Changes in matter are classified as physical or chemical.
Physical change: Alters appearance but not composition (e.g., melting, evaporation).
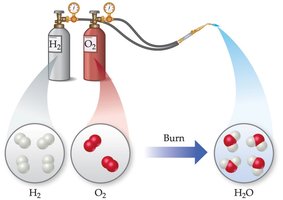
Chemical change: Produces new substances (e.g., combustion, oxidation).
Example: Melting ice is a physical change; burning hydrogen in oxygen to form water is a chemical change.


Separation of Mixtures
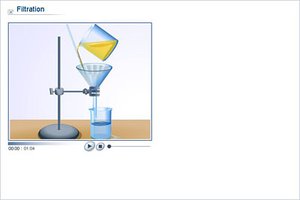
Filtration
Filtration separates solid substances from liquids or solutions based on particle size.
Example: Filtering sand from water.

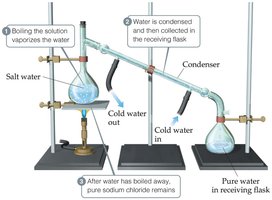
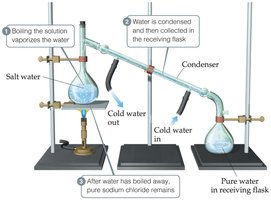
Distillation
Distillation separates components of a homogeneous mixture based on differences in boiling points.
Example: Separating salt from water by boiling and condensing the water vapor.
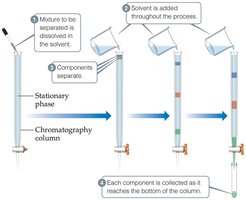
Chromatography
Chromatography separates substances based on their ability to adhere to a solid surface.
Example: Separating dyes in ink using paper chromatography.
Measurement in Chemistry
SI Units and Metric System
Chemistry relies on standardized units for measurement, primarily the SI (International System of Units) and the metric system.
Mass: kilogram (kg) or gram (g)
Length: meter (m)
Time: second (s)
Temperature: Kelvin (K) or Celsius (°C)
Amount of substance: mole (mol)
Volume: liter (L) or cubic centimeter (cm3)

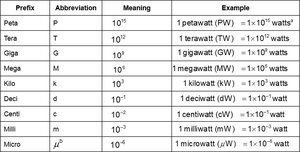
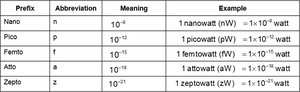
Metric System Prefixes
Prefixes are used to express multiples or fractions of base units.
Prefix | Abbreviation | Meaning | Example |
|---|---|---|---|
Kilo | k | 103 | 1 kilowatt (kW) = 1 × 103 watts |
Milli | m | 10-3 | 1 milliwatt (mW) = 1 × 10-3 watt |
Micro | μ | 10-6 | 1 microwatt (μW) = 1 × 10-6 watt |
Nano | n | 10-9 | 1 nanowatt (nW) = 1 × 10-9 watt |
Pico | p | 10-12 | 1 picowatt (pW) = 1 × 10-12 watt |
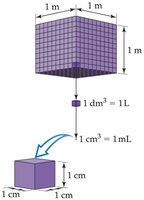
Volume Measurement
Volume is a derived unit, commonly measured in liters (L) and milliliters (mL).
1 L = 1 dm3
1 mL = 1 cm3
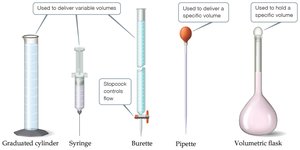
Laboratory Glassware for Measuring Volume
Different types of glassware are used for measuring and delivering volumes in the laboratory.
Graduated cylinder: Used to deliver variable volumes.
Burette: Used to deliver variable volumes with precision.
Pipette: Used to deliver a specific volume.
Volumetric flask: Used to hold a specific volume.
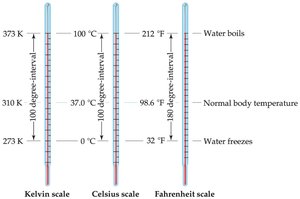
Temperature Measurement
Temperature is measured using Celsius, Kelvin, and Fahrenheit scales. The Kelvin scale is the SI unit and is based on absolute zero.
Celsius: 0°C (freezing point of water), 100°C (boiling point of water)
Kelvin: K = °C + 273.15
Fahrenheit: Not used in scientific measurements
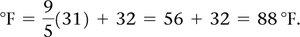
Energy and Density
Energy
Energy is measured in joules (J), a derived unit. The calorie is also used, but 1 cal = 4.184 J. The nutritional Calorie (Cal) is 1,000 cal.
Density
Density is a physical property defined as mass per unit volume.
Formula:
Units: g/mL or g/cm3
Example: Calculate the density of mercury if 1.00 × 102 g occupies 7.36 cm3.
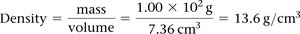
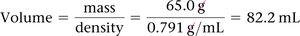
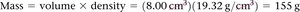
Measurement Accuracy and Precision
Uncertainty in Measurements
All measurements have some degree of uncertainty due to instrument limitations and human error. The last digit measured is considered reliable but not exact.
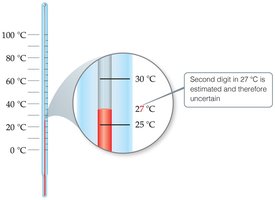
Accuracy vs. Precision
Accuracy: How close a measurement is to the true value.
Precision: How close repeated measurements are to each other.

Significant Figures
Significant figures reflect the precision of a measurement. Rules for determining significant figures:
All nonzero digits are significant.
Zeroes between nonzero digits are significant.
Zeroes at the beginning are not significant.
Zeroes at the end are significant if there is a decimal point.
Significant Figures in Calculations
Addition/Subtraction: Round to the least significant decimal place.
Multiplication/Division: Round to the same number of digits as the measurement with the fewest significant figures.
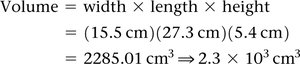
Dimensional Analysis
Unit Conversion
Dimensional analysis is used to convert between units using conversion factors.
Example: To convert pounds to grams, use the conversion factor 1 lb = 453.6 g.

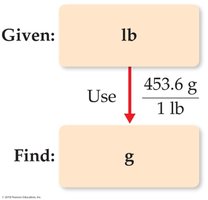
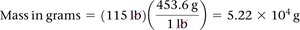
Multiple Conversion Factors
Complex conversions may require multiple steps, such as converting speed from meters per second to miles per hour.
Example:

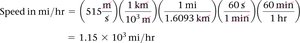
Volume Unit Conversion
Volume conversions may involve powers of conversion factors, such as converting km3 to L.
Example:
Example:
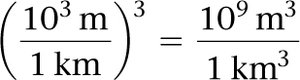
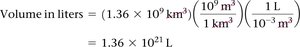
Summary Table: Classification of Matter
Type | Description | Example |
|---|---|---|
Element | Cannot be decomposed; one type of atom | Helium gas |
Compound | Can be decomposed; two or more elements | Pure water (H2O) |
Homogeneous mixture | Uniform composition | Salt water |
Heterogeneous mixture | Non-uniform composition | Salad dressing |
Additional info: These notes expand on brief points from the original materials, providing definitions, examples, and equations for clarity and completeness. Practice exercises and sample calculations are included to reinforce concepts.
