 Back
BackChapter 1: Matter and Energy – Foundations of Chemistry
Study Guide - Smart Notes

Matter and Energy
What is Chemistry?
Chemistry is the scientific study of matter, its properties, and the mechanisms involved in its changes. It explores the composition, structure, and transformations of substances at the atomic and molecular levels.
Matter: Anything that has mass and occupies space.
Atoms: Fundamental building blocks of matter; cannot be chemically broken down into smaller components.
Atoms combine to form molecules and compounds.

The Properties and Transformations of Matter
Matter exists in three primary states: solid, liquid, and gas. Each state is characterized by the arrangement and movement of its particles.
Solids: Particles are closely packed and relatively stationary; definite shape and volume.
Liquids: Particles are close but can move past one another; definite volume but no definite shape.
Gases: Particles are far apart and move rapidly; no definite shape or volume.
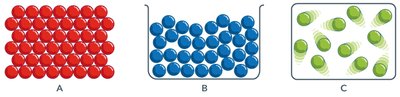
Properties of the Three States of Matter
State | Spacing of Atoms | Mobility | Compressible? | Shape/Volume |
|---|---|---|---|---|
Solid | Touching | Relatively stationary | Slightly | Definite shape and volume |
Liquid | Intermediate | Intermediate | Slightly | No shape, definite volume |
Gas | Very far apart | High velocities | Highly | No shape, no volume |
The Composition of Matter
Matter can be classified as pure substances or mixtures:
Pure Substances: Composed of only one type of atom or molecule; cannot be physically separated.
Elements: Consist of only one kind of atom; cannot be broken down further.
Compounds: Composed of two or more elements chemically combined; can be broken down into elements.
Mixtures: Contain more than one kind of pure substance; can be separated by physical means.
Homogeneous Mixtures (Solutions): Uniform composition throughout.
Heterogeneous Mixtures: Non-uniform composition.


The Properties and Changes of Matter
Properties of matter are classified as physical or chemical:
Physical Properties: Observed without changing the chemical composition (e.g., size, color, melting point).
Chemical Properties: Observed only when a substance undergoes a chemical change (e.g., flammability, acidity).
Changes in matter:
Physical Changes: Do not alter chemical composition; often reversible (e.g., phase changes, mixing).
Chemical Changes: Alter chemical composition; often irreversible (e.g., combustion, rusting).


Extensive and Intensive Properties
Extensive Properties: Depend on the amount of matter (e.g., mass, volume).
Intensive Properties: Independent of the amount of matter (e.g., density, temperature).
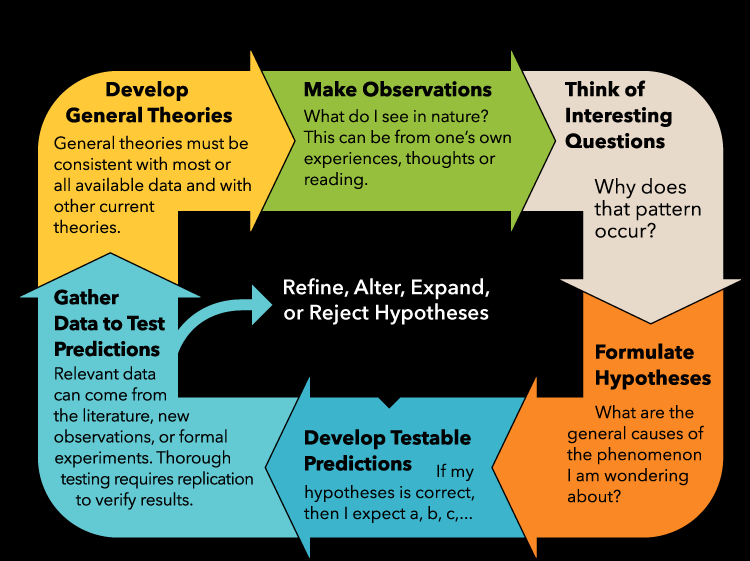
The Scientific Method
The scientific method is a systematic approach to investigating natural phenomena. It involves making observations, forming hypotheses, conducting experiments, and developing theories or laws based on evidence.
Steps: Make observations → Ask questions → Formulate hypotheses → Develop testable predictions → Gather data → Develop theories.
The Units of Measurement
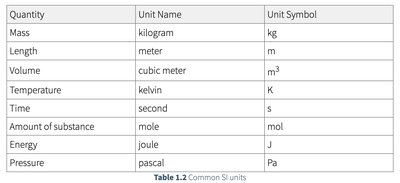
Measurements in chemistry consist of a number and a unit. The SI (International System of Units) is the standard system used in science.
Quantity | Unit Name | Unit Symbol |
|---|---|---|
Mass | kilogram | kg |
Length | meter | m |
Volume | cubic meter | m3 |
Temperature | kelvin | K |
Time | second | s |
Amount of substance | mole | mol |
Energy | joule | J |
Pressure | pascal | Pa |
Metric Prefixes
Prefixes are used to express very large or very small values (e.g., kilo-, centi-, milli-).
1 cm3 = 1 cc = 1 mL
Temperature and Temperature Conversions
Temperature is a measure of the average kinetic energy of molecules. Common scales include Celsius (°C), Kelvin (K), and Fahrenheit (°F).
Kelvin is the SI unit for temperature; absolute zero (0 K) is the lowest possible temperature.
Conversion equations:
Scientific Notation
Scientific notation expresses very large or small numbers as a product of a coefficient and a power of ten.
General form: where and is an integer.
Example: (Avogadro's number).
Accuracy, Precision, and Significant Figures
Accuracy refers to how close a measurement is to the true value; precision refers to the reproducibility of measurements.
Percent error quantifies accuracy:
Significant figures indicate the precision of a measurement.
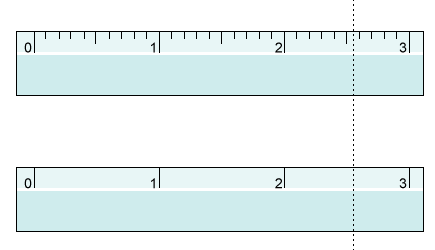
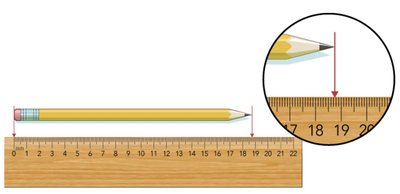
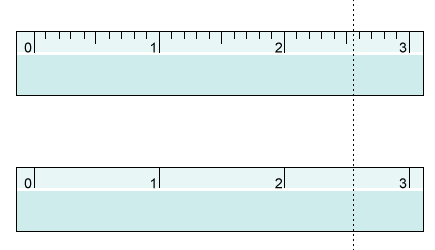
Rules for significant figures:
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Trailing zeros to the right of a decimal are significant.
Leading zeros are not significant.
Exact numbers have infinite significant figures.
For calculations:
Addition/Subtraction: Answer has the same number of decimal places as the value with the fewest decimal places.
Multiplication/Division: Answer has the same number of significant figures as the value with the fewest significant figures.
Dimensional Analysis
Dimensional analysis (unit factor method) is used to convert between units using conversion factors.
Set up conversion factors so that unwanted units cancel, leaving the desired units.
Example: To convert 321 nm to meters:
Density
Density is the ratio of mass to volume and is an intensive property unique to each substance.
At 20°C, the density of water is 0.99823 g/mL.
Substances with density less than water float; those with greater density sink.


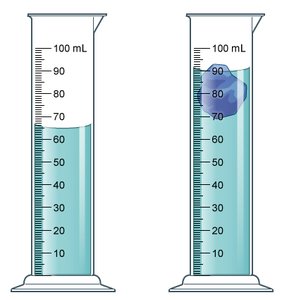
Volume by water displacement: The volume of water displaced by an object equals the object's volume.
Counting Atoms: The Mole
The mole is the SI unit for amount of substance. One mole contains Avogadro's number () of entities (atoms, molecules, etc.).
Energy and Chemistry
Energy is the capacity to do work or produce heat. In chemistry, energy is encountered in various forms:
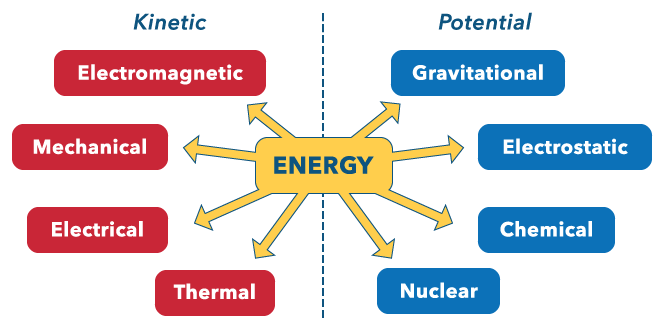
Kinetic Energy (KE): Energy of motion; depends on molecular movement and is related to temperature.
Potential Energy (PE): Energy stored due to position or arrangement of particles.

Examples of energy types: thermal, chemical, nuclear, mechanical, electrical, electromagnetic, gravitational, electrostatic.
