 Back
BackChapter 1: Matter, Measurement, and Problem Solving – General Chemistry Study Notes
Study Guide - Smart Notes

Units of Measurement for Physical and Chemical Change
Introduction to Chemistry
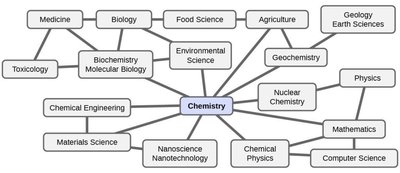
Chemistry is the science that seeks to understand the properties and behavior of matter by studying the properties and behavior of atoms and molecules. It is foundational to many scientific disciplines and has applications in fields such as medicine, engineering, environmental science, and more.
Atoms: Submicroscopic particles that are the fundamental building blocks of all matter.
Molecules: Two or more atoms bonded together in a specific geometric arrangement.
Matter: Any substance that has mass and takes up space.
Chemical Alphabet: Atoms and Elements
Atoms and the Periodic Table
Atoms are composed of protons, neutrons, and electrons. Each element is defined by the number of protons in its nucleus (atomic number). The periodic table organizes elements by increasing atomic number and groups elements with similar chemical properties together.
Element: A substance that cannot be broken down into simpler substances by chemical means.
Periodic Table: A chart that organizes all known elements according to their properties.

Molecules and Compounds
Structure and Properties of Molecules
Molecules are formed when two or more atoms are bonded together. The composition and structure of molecules determine their chemical and physical properties. For example, sodium chloride (NaCl) is a compound formed from sodium and chlorine atoms.
Chemical Bond: The force that holds atoms together in a molecule.
Compound: A substance composed of two or more different elements chemically bonded together.
Example: NaCl (table salt) is made from sodium (Na) and chlorine (Cl).

Physical and Chemical Properties of Matter
Qualitative and Quantitative Observations
Observations in chemistry can be qualitative (descriptive, using senses) or quantitative (measurable, using instruments).
Qualitative: Color, odor, texture, etc.
Quantitative: Mass, volume, temperature, etc.

Physical vs. Chemical Properties
Physical properties can be observed without changing the substance's chemical identity, while chemical properties describe how a substance interacts with other substances.
Physical Properties: State, color, melting point, boiling point, density, luster, etc.
Chemical Properties: Reactivity with oxygen, acidity, flammability, toxicity, etc.

Physical and Chemical Changes
Types of Changes in Matter
Changes in matter are classified as physical or chemical. Physical changes alter the form or appearance but not the composition, while chemical changes result in the formation of new substances.
Physical Change: Melting, freezing, condensation, vaporization, sublimation, deposition, dissolving.
Chemical Change: Rusting, burning, reacting with acids or bases.


Signs of Chemical Change
Bubbles of gas appear
Precipitate (solid) forms
Unexpected color change
Energy change (heat or light)
Change in electrical conductivity
Change in melting/boiling point
Change in odor or taste
Energy and Matter
Types of Energy
Physical and chemical changes are often accompanied by energy changes. Energy can be classified as kinetic (energy of motion) or potential (energy due to position or composition).
Kinetic Energy (KE):
Thermal Energy: Related to temperature and molecular motion.
Potential Energy (PE): (gravitational), (spring)
Chemical Energy: Associated with the arrangement of electrons and nuclei in atoms and molecules.
Law of Conservation of Energy: Energy cannot be created or destroyed, only transformed.
Measurement and Units
SI Units and Measurement Standards
Measurements in chemistry use the International System of Units (SI). Each measurement consists of a number and a unit.
Quantity | Unit | Symbol |
|---|---|---|
Length | meter | m |
Mass | kilogram | kg |
Time | second | s |
Temperature | kelvin | K |
Amount of Substance | mole | mol |
Mass vs. Weight
Mass: Amount of matter in an object (measured in kg).
Weight: Force of gravity on an object (measured in newtons, N).
Weight depends on location (e.g., Earth vs. Moon), mass does not.
Temperature Scales
Celsius (°C): Water freezes at 0°C, boils at 100°C.
Kelvin (K): Absolute temperature scale.
Fahrenheit (°F):
Scientific Notation and Prefix Multipliers
Expressing Numbers
Scientific notation is used to express very large or very small numbers. Prefix multipliers are used in the metric system to indicate powers of ten.
Scientific Notation: (N is a number between 1 and 10, n is an integer)
Prefix Multipliers: kilo (k, ), centi (c, ), milli (m, ), micro (μ, ), etc.
Prefix | Symbol | Multiplier |
|---|---|---|
kilo | k | |
centi | c | |
milli | m | |
micro | μ | |
nano | n |
Mathematical Operations with Scientific Notation
Addition/Subtraction: Convert to same exponent before adding/subtracting.
Multiplication: Multiply digit terms, add exponents.
Division: Divide digit terms, subtract exponents.
Precision, Accuracy, and Significant Figures
Precision and Accuracy
Accuracy: How close a measurement is to the true value.
Precision: How close repeated measurements are to each other.
Significant Figures
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Leading zeros are not significant.
Trailing zeros after a decimal point are significant.
When performing calculations, the result should reflect the precision of the measured quantities:
Multiplication/Division: Result has same number of significant figures as the factor with the fewest significant figures.
Addition/Subtraction: Result has same number of decimal places as the quantity with the fewest decimal places.
Dimensional Analysis and Unit Conversions
Dimensional Analysis
Dimensional analysis is a method of problem solving that uses the units to guide the calculation. Conversion factors are used to change from one unit to another.
Conversion Factor: A ratio that expresses how many of one unit are equal to another unit.
Example: To convert 15 miles to kilometers, use the conversion factor 1 mile = 1.609 km.
Derived Units: Volume and Density
Volume
SI unit: cubic meter (m3), but liters (L) and milliliters (mL) are commonly used in chemistry.
1 L = 1 dm3 = 1000 mL = 1000 cm3
Density
Density (d): (mass divided by volume)
SI unit: kg/m3; commonly used: g/cm3 or g/mL for solids and liquids, g/L for gases.
Density is an intensive property (independent of amount of substance).
Problem Solving in Chemistry
General Problem-Solving Strategy
Sort: Identify given information and what is to be found.
Strategize: Develop a conceptual plan (steps to follow).
Solve: Carry out the plan, paying attention to significant figures and units.
Check: Does the answer make sense?
Order of Operations in Calculations
P – Parentheses
E – Exponents
M – Multiplication
D – Division
A – Addition
S – Subtraction
Summary Table: Properties of Sodium, Chlorine, and Sodium Chloride
Property | Sodium | Chlorine | Sodium Chloride |
|---|---|---|---|
Melting Point (°C) | 97.8 | -101 | 801 |
Boiling Point (°C) | 881 | -34 | 1413 |
Colour | Silver | Greenish-yellow | White |
Density (g/cm3) | 0.97 | 0.0032 | 2.16 |
Additional info: This study guide covers the foundational concepts of matter, measurement, and problem solving in general chemistry, including the structure of matter, types of properties and changes, measurement standards, significant figures, and basic problem-solving strategies.
