 Back
BackChapter 1: Matter, Measurement, and Problem Solving – General Chemistry IA Study Notes
Study Guide - Smart Notes

Chapter 1: Matter, Measurement, and Problem Solving
1. Classification of Matter
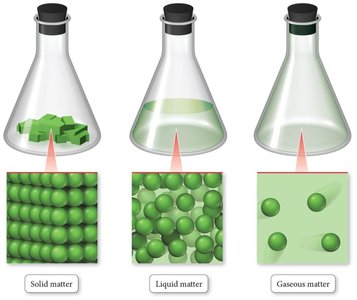
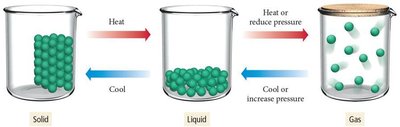
States of Matter
Matter is anything that occupies space and has mass. It consists of atoms and molecules and can exist in three primary states: solid, liquid, and gas. The state of matter depends on conditions such as pressure and temperature.
Solid: Atoms or molecules are packed closely in fixed locations, vibrate in place, and have a fixed volume and rigid shape. Solids can be crystalline (ordered structure) or amorphous (disordered structure).
Liquid: Atoms or molecules are close together but can move past each other. Liquids have a fixed volume but no fixed shape, taking the shape of their container.
Gas: Atoms or molecules are far apart and move freely. Gases have neither fixed volume nor shape and are compressible.
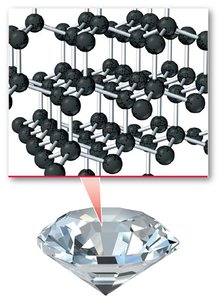
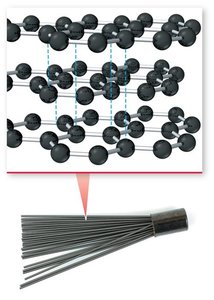
Crystalline vs. Amorphous Solids
Crystalline solids have a well-ordered structure, while amorphous solids lack long-range order. For example, diamond is a crystalline form of carbon, whereas graphite is an amorphous form.
Crystalline: Atoms arranged in a repeating pattern (e.g., diamond).
Amorphous: Atoms arranged randomly (e.g., graphite).
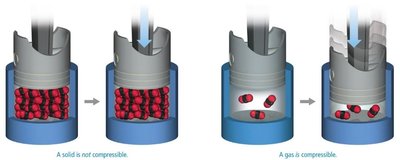
Compressibility of Solids and Gases
Solids are not compressible due to tightly packed particles, while gases are compressible because of the large spaces between particles.
Pressure and Temperature Dependence
The state of matter can change with the addition or removal of energy (heat) or changes in pressure. Heating a solid can turn it into a liquid, and further heating can turn it into a gas.
1.B. Composition of Matter
Purer Substances and Mixtures
Matter can be classified based on its composition as either pure substances or mixtures.
Element: Made up of only one type of atom; cannot be chemically broken down into simpler substances.
Compound: Composed of two or more elements in fixed proportions; can be broken down into simpler substances.
Mixture: Contains two or more substances; can be homogeneous (uniform throughout) or heterogeneous (not uniform).
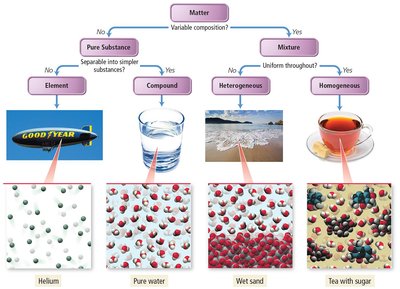
Homogeneous vs. Heterogeneous Mixtures
Homogeneous mixtures (solutions) have uniform composition and properties throughout, while heterogeneous mixtures have regions with different compositions and properties.
Homogeneous: Solution, alloy, colloid.
Heterogeneous: Suspension, visible different components.
1.C. Separating Mixtures
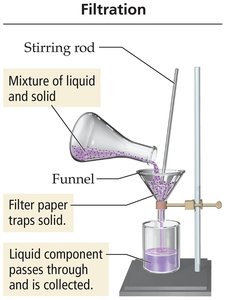
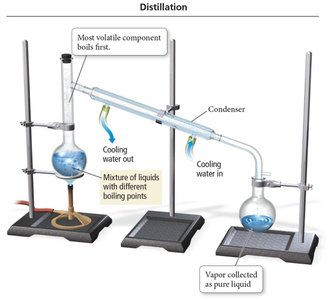
Separation Techniques
Mixtures can be separated by physical methods based on differences in their properties.
Filtration: Separates a heterogeneous mixture of an insoluble solid and a liquid using filter paper.
Distillation: Separates homogeneous mixtures of liquids based on differences in boiling points.
2. Types of Changes and Properties
2.A. Physical and Chemical Changes
Changes in matter can be classified as physical or chemical.
Physical Change: Alters the state or appearance of a substance without changing its composition (e.g., melting, dissolving).
Chemical Change: Alters the composition of matter; atoms rearrange to form new substances (e.g., burning, rusting).

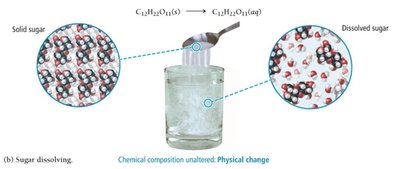

Evidence of Chemical Change
Formation of a precipitate
Gas evolution (bubbles)
Production or absorption of thermal energy
Color change
Emission of light
2.B. Physical and Chemical Properties
Properties of matter are classified as physical or chemical.
Physical Properties: Can be observed without changing the substance's composition (e.g., color, density, melting point).
Chemical Properties: Can only be observed by changing the substance's composition (e.g., flammability, reactivity).
2.C. Energy in Physical and Chemical Changes
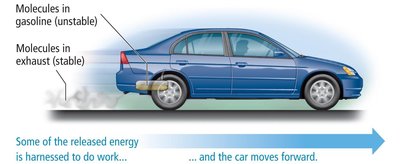
Energy is a fundamental aspect of physical and chemical changes. It is the capacity to do work, defined as the action of a force through a distance.

Potential Energy: Energy associated with position or composition.
Kinetic Energy: Energy associated with motion.
Thermal Energy: A type of kinetic energy related to temperature.

Law of Conservation of Energy: Energy cannot be created or destroyed; it can only be transformed. Systems with high potential energy tend to change to lower potential energy, releasing energy to the surroundings.
3. Units of Measurement
3.A. Standard Units
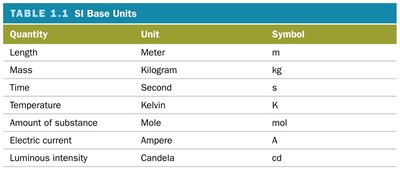
Measurements in science use standard units from the International System of Units (SI). The seven SI base units are:
Quantity | Unit | Symbol |
|---|---|---|
Length | Meter | m |
Mass | Kilogram | kg |
Time | Second | s |
Temperature | Kelvin | K |
Amount of substance | Mole | mol |
Electric current | Ampere | A |
Luminous intensity | Candela | cd |
Length
Measured in meters (m). 1 yard = 36 inches; 1 meter = 39.37 inches. The meter is defined as the distance light travels in a vacuum in second.

Mass
Measured in kilograms (kg). 1 gram (g) = kg. Mass is the quantity of matter; weight is the gravitational pull on matter and depends on location.

Time
Measured in seconds (s). Defined by the radiation events of cesium-133.
Temperature
Measured in Kelvin (K), Celsius (°C), or Fahrenheit (°F). Kelvin is the absolute scale, with 0 K as absolute zero. Temperature determines the direction of heat transfer.
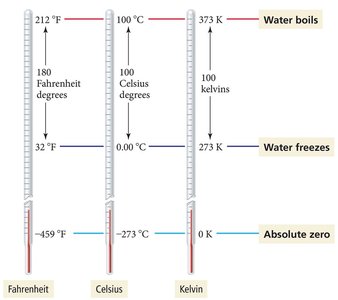
Temperature Conversion Equations
Example: A sick child has a temperature of 40.00 °C. What is the temperature in K and °F?
K
°F
Additional info: Prefix multipliers and derived units (such as volume and density) are also important in scientific measurement, but are not detailed in this excerpt.
