 Back
BackChapter 1: Matter, Measurement, and Problem Solving – Study Notes
Study Guide - Smart Notes

Exploring the Particulate Nature of Matter
Atoms, Elements, and the Molecular Perspective
The study of chemistry begins with understanding matter at the atomic and molecular level. Atoms are the smallest units of elements, which are pure substances that cannot be chemically or mechanically divided into simpler substances. Since atoms are too small to see directly, chemists use models and theories to represent their behavior and interactions.
Atom: The smallest particle of an element, indivisible by chemical or mechanical means.
Element: A pure substance made of only one kind of atom.
Classes and Properties of Matter
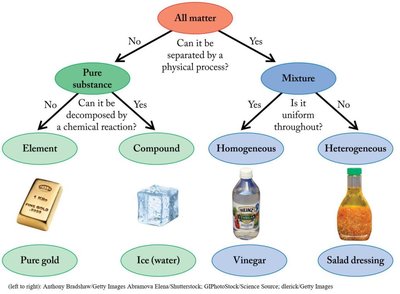
Classification of Matter
Matter is anything that has mass and occupies space. It can be classified based on its composition and properties:
Pure Substance: Has a constant composition and cannot be separated by physical processes. Subdivided into:
Element: Cannot be decomposed by chemical reactions (e.g., gold).
Compound: Can be decomposed by chemical reactions (e.g., water).
Mixture: Combination of two or more pure substances. Can be separated by physical processes.
Homogeneous Mixture: Uniform composition throughout (e.g., vinegar).
Heterogeneous Mixture: Non-uniform composition (e.g., salad dressing).
Physical and Chemical Properties
Properties of matter are divided into physical and chemical properties:
Physical Properties: Observed without changing the substance (e.g., melting point, color).
Chemical Properties: Observed only by changing the substance into another (e.g., reactivity with acids).
Intensive Properties: Independent of the amount (e.g., density, color).
Extensive Properties: Dependent on the amount (e.g., mass, volume).
Physical Change: Does not alter chemical identity (e.g., melting ice). Chemical Change: Converts substances into different substances (e.g., burning wood).
States of Matter
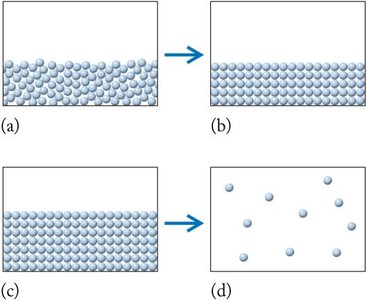
Solids, Liquids, and Gases
Matter exists in three primary states, each with distinct properties:
Solid: Definite shape and volume; particles are closely packed.
Liquid: Definite volume, takes the shape of its container; particles are less tightly packed than in solids.
Gas: No definite shape or volume; particles are far apart and move freely.
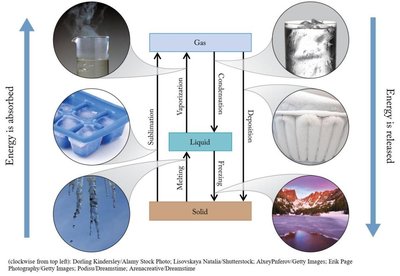
Phase Transitions
Transitions between states of matter involve energy changes:
Melting: Solid to liquid (energy absorbed).
Freezing: Liquid to solid (energy released).
Vaporization: Liquid to gas (energy absorbed).
Condensation: Gas to liquid (energy released).
Sublimation: Solid to gas (energy absorbed).
Deposition: Gas to solid (energy released).
Formulas and Models
Chemical Formulas and Representations
Chemists use formulas and models to represent substances and predict their behavior:
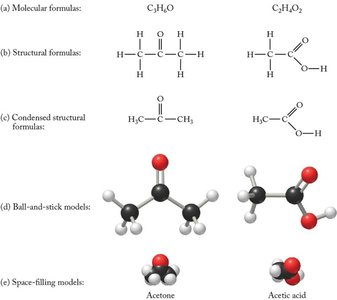
Molecular Formula: Shows the number and type of atoms in a molecule (e.g., H2O).
Empirical Formula: Shows the simplest whole-number ratio of atoms (e.g., CH2O for glucose).
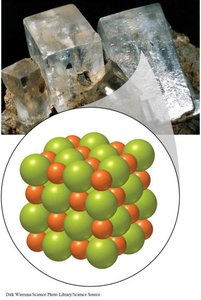
Ionic Compound: Composed of positive and negative ions in a fixed ratio (e.g., NaCl).
Forms of Energy
Energy in Chemistry
Energy is the capacity to do work or transfer heat. In chemistry, energy changes accompany physical and chemical processes:
Kinetic Energy (KE): Energy of motion.
Potential Energy (PE): Stored energy due to position or composition.
Chemical Energy: A form of potential energy stored in chemical bonds.
Coulomb Potential: , where Q1 and Q2 are charges, d is distance.
Law of Conservation of Energy: Energy cannot be created or destroyed, only converted.
Expressing Experimental Results
SI Units and Metric Prefixes
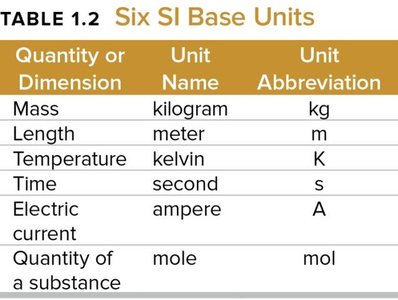
The International System of Units (SI) is used globally for scientific measurements. It includes base units for mass, length, temperature, time, electric current, and amount of substance.
Quantity or Dimension | Unit Name | Unit Abbreviation |
|---|---|---|
Mass | kilogram | kg |
Length | meter | m |
Temperature | kelvin | K |
Time | second | s |
Electric current | ampere | A |
Quantity of a substance | mole | mol |
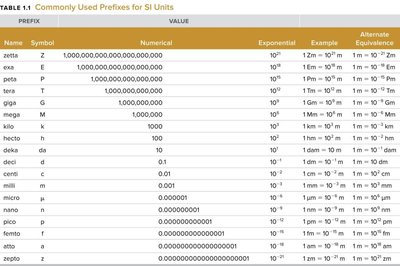
Metric prefixes are used to express multiples or fractions of units:
Name | Symbol | Numerical Value | Exponential |
|---|---|---|---|
kilo | k | 1,000 | 103 |
centi | c | 0.01 | 10-2 |
milli | m | 0.001 | 10-3 |
micro | μ | 0.000001 | 10-6 |
Temperature Scales
Temperature can be measured in Celsius (°C), Kelvin (K), or Fahrenheit (°F). Conversion formulas:

Precision, Accuracy, and Significant Figures
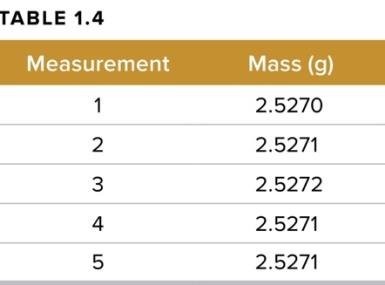
Measurements in chemistry must be both precise and accurate:
Precision: Reproducibility of measurements.
Accuracy: Closeness to the true value.
Significant Figures: All certain digits plus one estimated digit in a measurement.

Rules for significant figures:
All nonzero digits are significant.
Leading zeros are not significant.
Zeros between nonzero digits are significant.
Trailing zeros after a decimal point are significant.
Trailing zeros in a whole number with no decimal point may or may not be significant (use scientific notation to clarify).
For calculations:
Multiplication/Division: Result has the same number of significant figures as the value with the fewest significant figures.
Addition/Subtraction: Result has the same number of decimal places as the value with the fewest decimal places.
Unit Conversions and Dimensional Analysis
Conversion Factors and Problem Solving
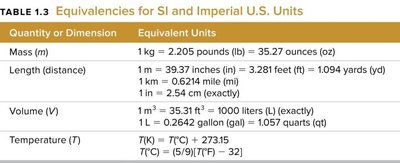
Unit conversions use equivalence statements as ratios (conversion factors) to relate different units. Dimensional analysis ensures that units cancel appropriately, leaving the desired unit.
Example: (exactly)
Conversion factor: or
Sample Problem: If a child weighs 28 lb and needs 75 mg of medication per kg of body mass per day, and the medication is 125 mg/mL, how many mL per dose (given twice daily)?
Convert pounds to kilograms:
Calculate total mg needed, then divide by mg/mL to get mL per dose.
Sample Problem: Convert 134.83 billion gallons of gasoline per year to cubic meters per second.
Use and
Convert years to seconds.
Additional info: Some tables and images have been summarized for clarity and brevity. For full details, refer to the original tables in your textbook or lecture materials.
