 Back
BackChapter 1: Matter, Measurement, and Problem Solving – General Chemistry Study Notes
Study Guide - Smart Notes

Chapter 1: Matter, Measurement, and Problem Solving
Chemistry: The Study of Matter
Chemistry is the science that seeks to understand the behavior of matter by studying the actions and interactions of atoms and molecules. Matter is anything that has mass and occupies space, and it is composed of chemicals that make up everything we encounter.
Matter: "Stuff" that has mass and volume.
Chemicals: Substances that compose ordinary things, not just dangerous substances.
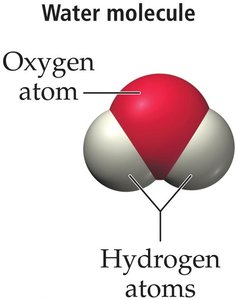
Atoms and Molecules: Fundamental building blocks of matter; atoms rarely exist freely and usually form molecules.
Example: Water is composed of molecules containing two hydrogen atoms and one oxygen atom.

Safety in Chemistry
Safety Data Sheets (SDS) are essential documents that provide information about the hazards, handling, and emergency measures for chemicals used in laboratories.
SDS: Details chemical identity, hazards, and safety precautions.
Example: Capsaicin SDS shows toxicity and eye damage risks.

Quantification and Measurement
Chemistry is a quantitative science, relying on precise measurement and universal standards for reporting numbers. Units are critical for specifying measurements.
Metric System: Used globally; based on powers of ten.
SI Units: International System of Units, derived from the metric system.
Key SI Base Units:
Quantity | Unit | Symbol |
|---|---|---|
Length | Meter | m |
Mass | Kilogram | kg |
Time | Second | s |
Temperature | Kelvin | K |
Amount of substance | Mole | mol |
Electric current | Ampere | A |
Luminous intensity | Candela | cd |
Mass and Weight
Mass is the measure of the quantity of matter in an object, while weight is the measure of the gravitational pull on that matter.
SI unit of mass: kilogram (kg)
1 kg = 2.205 lb
Gram (g): 1/1000 kg
Temperature and Its Measurement
Temperature measures the average kinetic energy of atoms or molecules. The SI unit is the kelvin (K), and temperature determines the direction of heat transfer.
Kelvin scale: Absolute scale, 0 K is absolute zero.
Absolute zero: –273 °C or –459 °F, where molecular motion stops.
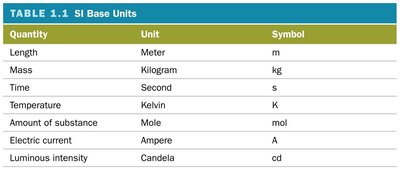
Temperature conversions:
Scale | Freezing Point | Boiling Point | Absolute Zero |
|---|---|---|---|
Fahrenheit | 32 °F | 212 °F | –459 °F |
Celsius | 0 °C | 100 °C | –273 °C |
Kelvin | 273 K | 373 K | 0 K |

Conversion formulas:
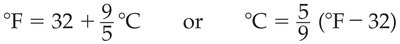
Scientific Notation
Scientific notation is used to express very large or very small numbers conveniently. It consists of a decimal part and an exponential part.
Decimal part: Number between 1 and 10.
Exponential part: 10 raised to an exponent.
Example:
Prefix Multipliers
SI units use prefix multipliers to indicate powers of ten, such as kilo (103), milli (10-3), and micro (10-6).
Example: 1 kilometer (km) = 1000 meters (m)
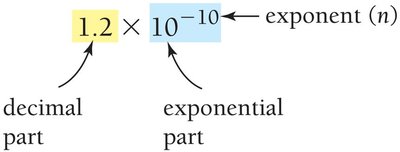
Derived Units: Volume and Density
Derived units are combinations of base units. Volume is measured in cubic centimeters (cm3) or liters (L), and density is the ratio of mass to volume.
Density formula:
Intensive property: Does not depend on the amount of material (e.g., density).
Extensive property: Depends on the amount of material (e.g., mass, volume).
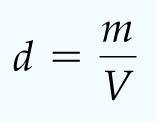
Accuracy, Precision, and Uncertainty
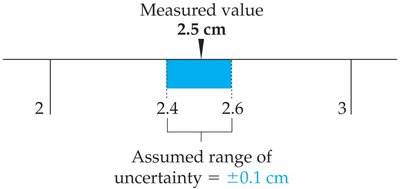
Measurements in chemistry are subject to uncertainty. Accuracy refers to how close a measurement is to the true value, while precision refers to how consistent repeated measurements are.
Uncertainty: Associated with any measured value; the last digit is estimated.
Significant figures: Reflect the precision of a measurement.
Example: 2.5 cm measured on a ruler has uncertainty in the last digit.


Reporting Scientific Numbers and Significant Figures
Scientific numbers must be reported with the correct number of significant figures. The rules for significant figures help ensure that calculations reflect the precision of the measured quantities.
Rules:
All nonzero digits are significant.
Interior zeros are significant.
Trailing zeros after a decimal point are significant.
Leading zeros are not significant.
Significant Figures in Calculations
When performing calculations, the result must reflect the precision of the input values.
Multiplication/Division: Result has the same number of significant figures as the factor with the fewest.
Addition/Subtraction: Result has the same number of decimal places as the quantity with the fewest decimal places.
Do not round intermediate steps; round only the final answer.
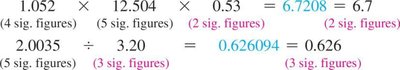
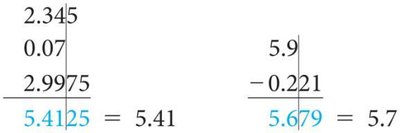
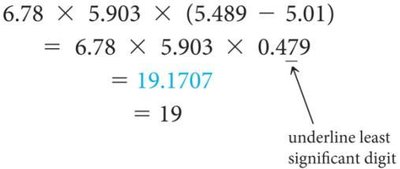
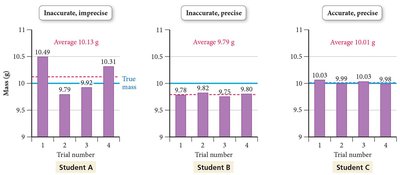
Types of Errors
Errors in measurement can be random or systematic. Random errors fluctuate unpredictably, while systematic errors consistently skew results in one direction.
Random error: Equal probability of being too high or too low.
Systematic error: Consistently too high or too low.
Example: Student A (inaccurate, imprecise), Student B (precise, inaccurate), Student C (accurate, precise).
Dimensional Analysis and Unit Conversion
Dimensional analysis is a method for solving problems using units as a guide. Conversion factors are used to change units, and units should always be included in calculations.
Conversion factor: Ratio used to convert from one unit to another.
Example:
Units raised to a power: Both the number and unit must be raised to the power.

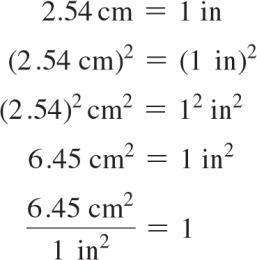
Classification of Matter
Matter can be classified by its physical state (solid, liquid, gas) and by its composition (elements, compounds, mixtures).
States of matter: Solid (fixed shape and volume), liquid (fixed volume, variable shape), gas (variable shape and volume).
Structure determines properties: Arrangement of atoms/molecules affects physical properties.
Crystalline solids: Long-range order (e.g., table salt, diamond).
Amorphous solids: No long-range order (e.g., glass, plastic).

Pure Substances and Mixtures
Pure substances contain only one type of matter, while mixtures contain two or more substances intermingled.
Elements: Basic building blocks, organized in the periodic table.
Compounds: Two or more elements joined together.
Mixtures: Can be homogeneous (uniform composition) or heterogeneous (variable composition).
Separating Mixtures
Mixtures can be separated by physical methods such as distillation (separating liquids by boiling point) and filtration (separating solids from liquids).
Distillation: Separates based on volatility.
Filtration: Separates based on solubility.
Physical and Chemical Changes
Physical changes alter the state or appearance of matter without changing its composition, while chemical changes alter the composition by rearranging atoms.
Physical change: Melting, freezing, vaporization, condensation, sublimation.
Chemical change: Rusting, burning, reactions.
Physical and Chemical Properties
Physical properties can be observed without changing the substance's composition, while chemical properties are observed only during a chemical change.
Physical properties: Odor, taste, color, appearance, melting point, boiling point, density.
Chemical properties: Flammability, corrosiveness, acidity, toxicity.
Energy in Chemistry
Energy is the capacity to do work, and every change in matter involves energy. The total energy of a substance is the sum of its kinetic and potential energy.
Kinetic energy: Energy of motion.
Potential energy: Energy of position or composition.
Forms of energy: Electrical, thermal, chemical, light.
The Scientific Method
The scientific method is a systematic approach to learning that emphasizes observation, experimentation, and the development of theories and laws.
Observation: Measuring or observing nature.
Hypothesis: Tentative interpretation, must be testable and falsifiable.
Theory: Model explaining observations and laws, predictive and well-tested.
Law: Brief statement synthesizing observations, often mathematical.
Law of Conservation of Mass
The law of conservation of mass states that in a chemical reaction, matter (atoms) is neither created nor destroyed.
Example: The total mass of reactants equals the total mass of products in a chemical reaction.
} }
