 Back
BackChapter 1: Matter, Measurement, and Problem Solving – Structured Study Notes
Study Guide - Smart Notes

Chapter 1: Matter, Measurement, and Problem Solving
The Units of Measurement
Measurement is fundamental in chemistry, and units provide a standardized way to express quantities. The International System of Units (SI) is the global standard, based on the metric system. Understanding units is essential for accurate scientific communication and calculations.
Metric System: Used worldwide, based on powers of ten.
English System: Used primarily in the United States.
SI Units: Standard units for scientific measurement.
SI Base Units:
Quantity | Unit | Symbol |
|---|---|---|
Length | Meter | m |
Mass | Kilogram | kg |
Time | Second | s |
Temperature | Kelvin | K |
Amount of substance | Mole | mol |
Electric current | Ampere | A |
Luminous intensity | Candela | cd |
Length: The meter (m) is the SI unit of length, slightly longer than a yard.

Mass: The kilogram (kg) is the SI unit of mass. Mass measures the amount of matter, while weight measures the gravitational pull on that matter.

Time: The second (s) is the SI unit of time, defined by the radiation events of cesium-133.
Temperature: The kelvin (K) is the SI unit of temperature. Temperature reflects the average kinetic energy of particles and determines the direction of heat transfer. Absolute zero (0 K) is the lowest possible temperature.
Temperature Conversions: The Celsius and Fahrenheit scales are commonly used. Conversion formulas are essential for translating between scales.
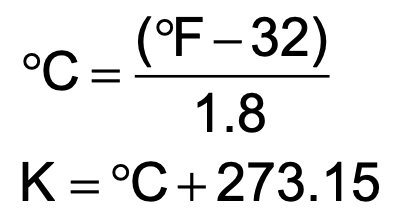
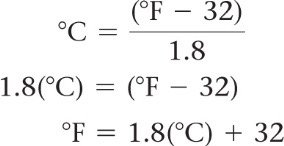
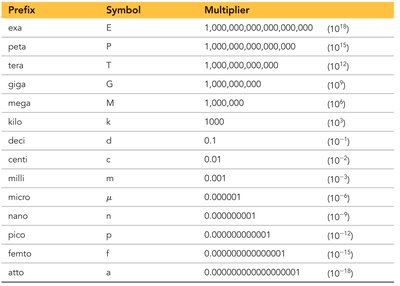
SI Prefix Multipliers
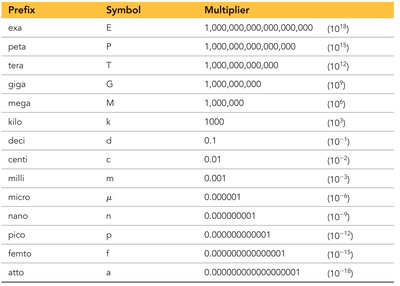
SI units use prefix multipliers to represent quantities that are powers of ten. These prefixes make it easier to express very large or very small numbers.
Prefix | Symbol | Multiplier |
|---|---|---|
exa | E | 1,000,000,000,000,000,000 (1018) |
peta | P | 1,000,000,000,000,000 (1015) |
tera | T | 1,000,000,000,000 (1012) |
giga | G | 1,000,000,000 (109) |
mega | M | 1,000,000 (106) |
kilo | k | 1,000 (103) |
deci | d | 0.1 (10-1) |
centi | c | 0.01 (10-2) |
milli | m | 0.001 (10-3) |
micro | μ | 0.000001 (10-6) |
nano | n | 0.000000001 (10-9) |
pico | p | 0.000000000001 (10-12) |
femto | f | 0.000000000000001 (10-15) |
atto | a | 0.000000000000000001 (10-18) |
Derived Units: Volume and Density
Derived units are combinations of base units. Volume and density are common derived units in chemistry.
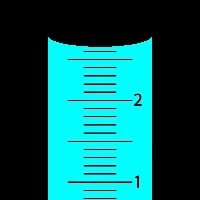
Volume: Measures space, typically in cubic centimeters (cm3) or liters (L).
Density: Ratio of mass to volume, , usually in g/cm3.
Intensive Property: Independent of the amount of substance (e.g., density).
Extensive Property: Dependent on the amount of substance (e.g., mass).
The Reliability of a Measurement
Reliability in measurement is determined by precision and accuracy, as well as the correct use of significant figures.
Significant Figures: Digits that reflect the precision of a measurement.
Exact Numbers: Have unlimited significant figures (e.g., counting objects).
Precision: Consistency among measurements.
Accuracy: Closeness to the true value.

Rules for Counting Significant Figures:
All nonzero digits are significant.
Interior zeroes (between nonzero digits) are significant.
Leading zeroes (before the first nonzero digit) are not significant.
Trailing zeroes after a decimal point are significant.
Trailing zeroes before an implied decimal point are ambiguous.
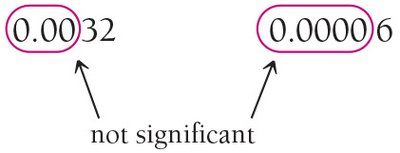
Significant Figures in Calculations
When performing calculations, the number of significant figures in the result depends on the operation:
Multiplication/Division: Result has the same number of significant figures as the factor with the fewest significant figures.
Addition/Subtraction: Result has the same number of decimal places as the quantity with the fewest decimal places.
Rounding: Round down if the digit dropped is 4 or less; round up if 5 or more.
Multistep Calculations: Only round the final answer to avoid rounding errors.

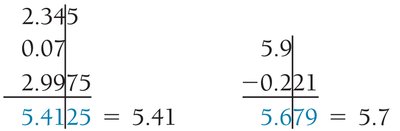
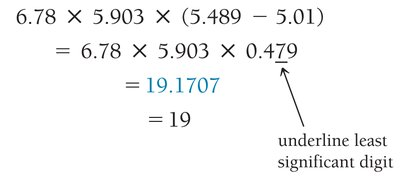

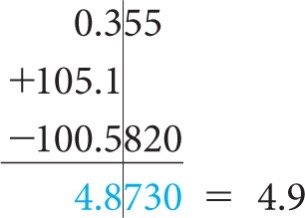
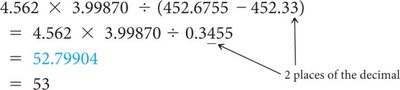
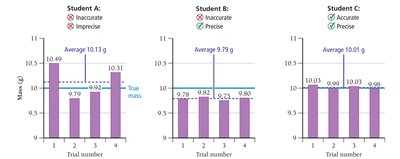
Precision and Accuracy
Precision and accuracy are critical for evaluating the quality of measurements. Random error affects precision, while systematic error affects accuracy.
Random Error: Equal probability of being too high or too low.
Systematic Error: Consistently too high or too low.
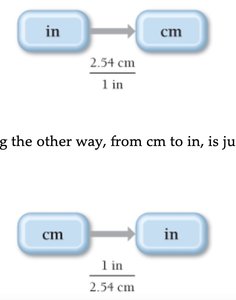
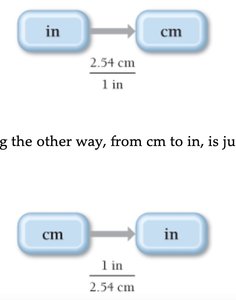
Solving Chemical Problems: Dimensional Analysis
Dimensional analysis is a method for solving unit conversion problems by treating units algebraically. Conversion factors are used to translate between units.
Unit Equation: Statement of two equivalent quantities (e.g., 2.54 cm = 1 in).
Conversion Factor: Fractional quantity used to convert between units.

Unit Conversion Examples
Unit conversion problems require sorting information, strategizing a plan, and applying relationships between units.
Example: Convert 1.76 yards to centimeters.
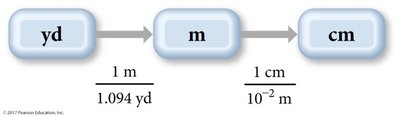
Example: Convert 1.8 quarts to cubic centimeters.
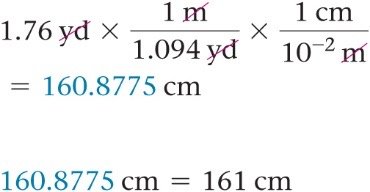
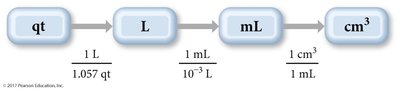
Units Raised to a Power
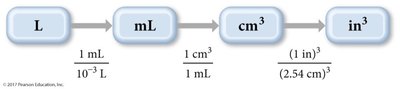
When converting units raised to a power (e.g., area or volume), both the number and the unit must be raised to the power.
Example: Convert liters to cubic inches.
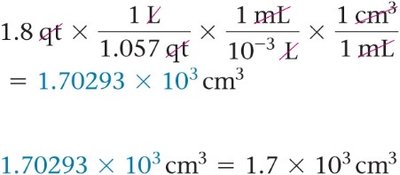
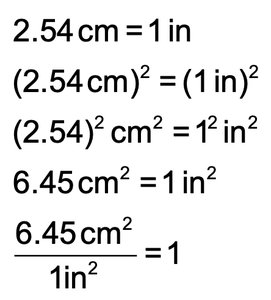
Density as a Conversion Factor
Density can be used as a conversion factor to relate mass and volume. This is useful in practical applications such as calculating the mass of fuel.
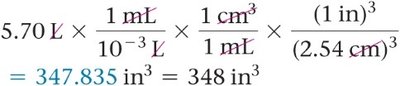
Problems with Equations
Chemistry often requires solving problems using equations, such as finding the radius of a sphere or the density of a cylinder.
Volume of a Sphere:
Volume of a Cylinder:
Density:

Analyzing and Interpreting Data
Data in chemistry is often visualized using graphs. The x-axis represents the independent variable, while the y-axis represents the dependent variable. Understanding how to interpret graphs is essential for analyzing experimental results.
X-axis: Independent variable (manipulated).
Y-axis: Dependent variable (measured).
Slope: Indicates the rate of change.
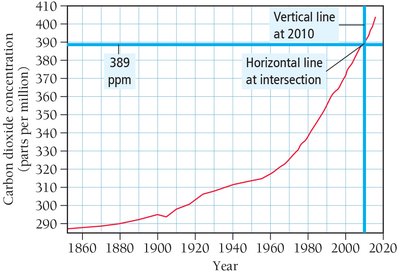
Additional info: These notes cover all major aspects of Chapter 1, including measurement, units, significant figures, dimensional analysis, and data interpretation, providing a comprehensive foundation for general chemistry students.
