 Back
BackChapter 10: Gases – Properties, Laws, and Real Behavior
Study Guide - Smart Notes

Gases: Physical Characteristics
General Properties of Gases
Gases are a unique state of matter characterized by their ability to expand and fill containers, high compressibility, and extremely low densities compared to liquids and solids. Most gases are composed of nonmetallic elements with simple formulas and low molar masses. When mixed, gases form homogeneous mixtures regardless of their identities.
Expansion: Gases fill any container they occupy.
Compressibility: Gases can be compressed much more than liquids or solids.
Low Density: Gases have much lower densities than other states of matter.
Homogeneous Mixtures: Multiple gases mix uniformly.
Common Gases at Room Temperature
Several compounds exist as gases at room temperature, each with distinct properties and hazards.
Formula | Name | Characteristics |
|---|---|---|
HCN | Hydrogen cyanide | Very toxic, slight odor of bitter almonds |
H2S | Hydrogen sulfide | Very toxic, odor of rotten eggs |
CO | Carbon monoxide | Toxic, colorless, odorless |
CO2 | Carbon dioxide | Colorless, odorless |
CH4 | Methane | Colorless, odorless, flammable |
C2H4 | Ethene (Ethylene) | Colorless, ripens fruit |
C3H8 | Propane | Colorless, odorless, bottled gas |
N2O | Nitrous oxide | Colorless, sweet odor, laughing gas |
NO2 | Nitrogen dioxide | Toxic, red-brown, irritating odor |
NH3 | Ammonia | Colorless, pungent odor |
SO2 | Sulfur dioxide | Colorless, irritating odor |
Pressure and Its Measurement
Definition of Pressure
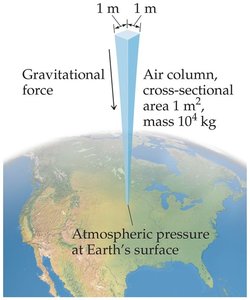
Pressure is defined as the amount of force applied to a given area. All gases exert pressure on surfaces they contact, and atmospheric pressure is the weight of air per unit area at Earth's surface.
Formula:
Atmospheric Pressure: Caused by the gravitational force acting on the air column above Earth's surface.
Units of Pressure
Pressure can be measured in several units, including pascals (Pa), bar, millimeters of mercury (mm Hg or torr), and atmospheres (atm). Conversion between these units is often necessary in calculations.
Pascals (Pa): SI unit of pressure.
Bar: 1 bar = 100 kPa.
mm Hg or Torr: Based on the height difference in a mercury column.
Atmosphere (atm): 1 atm = 760 torr = 760 mm Hg = 101.325 kPa = 1.01325 bar.
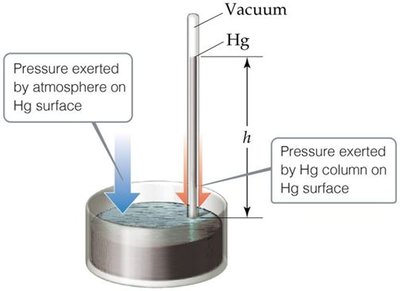
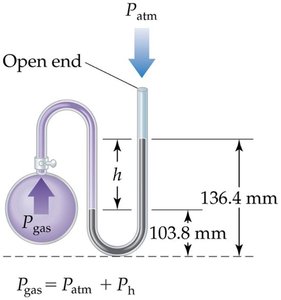
Manometer
A manometer is used to measure the pressure difference between atmospheric pressure and the pressure of a gas in a vessel. The difference in height of the mercury column indicates the pressure.
Standard Pressure
Standard atmospheric pressure at sea level is used as a reference in many calculations. It is equivalent to 1 atm, 760 torr, 760 mm Hg, 101.325 kPa, or 1.01325 bar.
The Gas Laws
Variables Defining Gas State
The physical state of a gas is defined by four variables: temperature, pressure, volume, and amount (moles). The relationships between these variables are described by the gas laws.
Temperature (T): Measured in Kelvin (K).
Pressure (P): Measured in atm, Pa, etc.
Volume (V): Measured in liters (L).
Amount (n): Measured in moles.
Boyle’s Law: Pressure–Volume Relationship
Boyle’s Law states that the volume of a fixed quantity of gas at constant temperature is inversely proportional to the pressure.
Mathematical Relationship:
For two conditions:
Graph: V vs. P is not linear; V vs. 1/P is linear.
Charles’s Law: Temperature–Volume Relationship
Charles’s Law states that the volume of a fixed amount of gas at constant pressure is directly proportional to its absolute temperature.
Mathematical Relationship:
For two conditions:
Graph: V vs. T is linear.

Avogadro’s Law: Quantity–Volume Relationship
Avogadro’s Law states that the volume of a gas at constant temperature and pressure is directly proportional to the number of moles of the gas.
Mathematical Relationship:
At STP: One mole of any gas occupies 22.4 L.

The Ideal-Gas Equation
Derivation and Formula
The ideal-gas equation combines Boyle’s, Charles’s, and Avogadro’s laws into a single relationship. The proportionality constant R is known as the ideal gas constant.
Formula:
R Values: R has different numerical values depending on units.
Unit | Numerical Value |
|---|---|
L·atm/(mol·K) | 0.08206 |
J/(mol·K) | 8.314 |
cal/(mol·K) | 1.987 |
m3·Pa/(mol·K) | 8.314 |
L·torr/(mol·K) | 62.36 |
Solving Gas Law Problems
To solve gas law problems, tabulate information, convert units, select the correct equation, and use dimensional analysis.
Gas Densities and Molar Mass
Calculating Density
The density of a gas can be calculated using the ideal-gas equation and the molar mass.
Formula:
Variables: M = molar mass, P = pressure, R = gas constant, T = temperature
Calculating Molar Mass
The molar mass of a gas can be determined if the mass, volume, and temperature are known.
Formula:
Volume of Gases in Chemical Reactions
Stoichiometry with Gases
Balanced chemical equations indicate the relative amounts of moles involved in reactions. Unknowns can be determined using stoichiometry and the ideal-gas equation.
If given volume, use stoichiometric ratios.
If given P, V, T, solve for n using , then use mole ratios and convert to desired units.
Gas Mixtures and Partial Pressures
Dalton’s Law of Partial Pressures
When two or more non-reacting gases are combined, each behaves as if it is alone in the container. The total pressure is the sum of the partial pressures of each gas.
Formula:
Mole Fraction and Partial Pressure
The mole fraction of a gas in a mixture is used to calculate its partial pressure.
Mole Fraction:
Partial Pressure:
Kinetic-Molecular Theory of Gases
Purpose and Statements
The kinetic-molecular theory explains why gases behave as described by the gas laws. It is based on several key assumptions:
Gases consist of large numbers of molecules in continuous, random motion.
The combined volume of all molecules is negligible compared to the container volume.
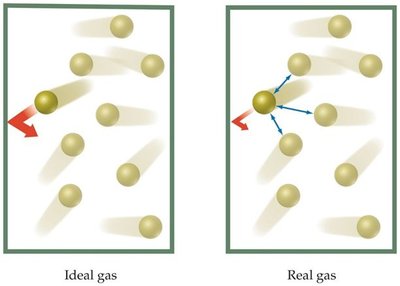
Attractive and repulsive forces between molecules are negligible.
Energy can be transferred during collisions, but average kinetic energy remains constant if temperature is constant.
Average kinetic energy is proportional to absolute temperature; at the same temperature, all gases have equal average kinetic energy.

Distributions of Molecular Speed
Temperature affects the distribution of molecular speeds. Three key speeds are defined:
Most probable speed (vmp): The speed at which the largest number of molecules move.
Average speed (vavg): The mean speed of all molecules.
Root-mean-square speed (vrms): The speed associated with the average kinetic energy.

Application to Gas Laws
Kinetic-molecular theory explains the relationships observed in gas laws:
Increasing volume at constant temperature decreases pressure due to fewer collisions.
Increasing temperature at constant volume increases pressure due to more frequent and forceful collisions.
Molecular Speeds, Effusion, and Diffusion
Effusion and Diffusion
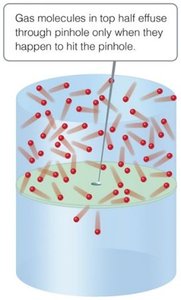
Effusion is the escape of gas molecules through a tiny hole into an evacuated space, while diffusion is the spread of one substance throughout another.
Effusion: Escape through a pinhole.
Diffusion: Mixing and spreading in a space.

Graham’s Law
Graham’s law relates the rate of effusion or diffusion to the molar mass of gases. Lighter gases effuse and diffuse faster than heavier gases.
Formula:

Real Gases: Deviations from Ideal Behavior
Deviations from Ideal-Gas Behavior
Real gases deviate from ideal behavior at high pressures and low temperatures. The assumptions of the kinetic-molecular theory break down under these conditions.
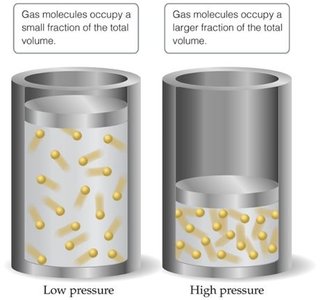
Gas molecules occupy a significant fraction of the total volume at high pressure.
Attractive forces between molecules become significant at low temperature.

Van der Waals Equation
The van der Waals equation corrects the ideal-gas equation for real gases by accounting for molecular volume and intermolecular forces. Each gas has specific van der Waals constants (a and b).
Equation:
a: Corrects for intermolecular attractions.
b: Corrects for molecular volume.
Substance | a (L2·atm/mol2) | b (L/mol) |
|---|---|---|
He | 0.0341 | 0.02370 |
Ne | 0.211 | 0.0171 |
Ar | 1.34 | 0.0322 |
Kr | 2.32 | 0.0398 |
Xe | 4.19 | 0.0510 |
H2 | 0.244 | 0.0266 |
N2 | 1.39 | 0.0391 |
O2 | 1.36 | 0.0318 |
F2 | 1.06 | 0.0290 |
Cl2 | 6.49 | 0.0562 |
H2O | 5.46 | 0.0305 |
NH3 | 4.17 | 0.0371 |
CH4 | 2.25 | 0.0428 |
CO2 | 3.59 | 0.0427 |
CCl4 | 20.4 | 0.1383 |
Additional info: The van der Waals equation is essential for accurately describing gas behavior under non-ideal conditions, such as in industrial processes or extreme environments.
