 Back
BackChapter 10: Gases – Properties, Laws, and Applications
Study Guide - Smart Notes

Gases: An Overview
Nature and Properties of Gases
Gases are one of the fundamental phases of matter, characterized by particles that move randomly and rapidly within their container. This random motion leads to unique properties such as compressibility and the ability to fill any container.
Particles in Motion: Gas molecules are in constant, random motion, colliding with each other and the walls of their container.
Compressibility: Gases can be compressed much more easily than solids or liquids due to the large amount of empty space between particles.
Expansion: Gases expand to fill the shape and volume of their container.
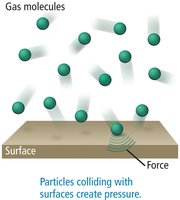
Pressure and Its Measurement
Definition of Pressure
Pressure (P) is defined as the force (F) exerted per unit area (A):
Units: The SI unit of pressure is the pascal (Pa), but atmospheres (atm), millimeters of mercury (mm Hg or torr), and pounds per square inch (psi) are also commonly used.
Factors Affecting Gas Pressure
Concentration of Gas Molecules: Higher concentration (more particles per volume) increases pressure.
Average Speed of Particles: Faster-moving particles exert greater force upon collision, increasing pressure.
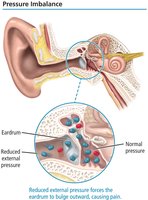
Pressure Imbalance and Its Effects
Pressure differences across membranes, such as the eardrum, can cause discomfort or pain due to the membrane being pushed outward or inward.
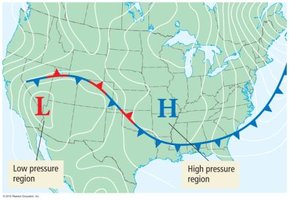
Atmospheric Pressure and Weather
Atmospheric pressure varies with altitude and is responsible for weather phenomena such as wind. Pressure decreases with increasing altitude due to fewer gas particles in a given volume.
Pressure Units and Conversions
Common pressure units include atm, mm Hg (torr), and psi. Conversion between units is often necessary in calculations.
1 atm = 760 mm Hg = 14.7 psi
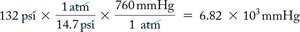
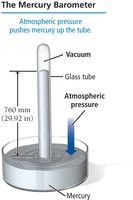
Measuring Pressure: Barometers and Manometers
Mercury Barometer: Measures atmospheric pressure by the height of mercury pushed up a tube by atmospheric force.
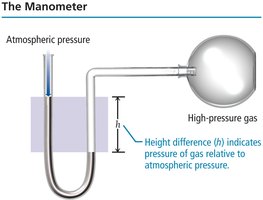
Manometer: Measures the pressure of a gas in a container relative to atmospheric pressure using a U-shaped tube filled with liquid.
Basic Properties of Gases
Four Fundamental Properties
Pressure (P): Measured in atm
Volume (V): Measured in liters (L)
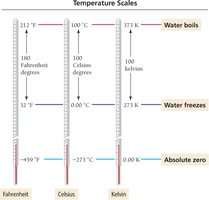
Temperature (T): Measured in Kelvin (K), where
Number of Particles (n): Measured in moles (mol)
The Simple Gas Laws
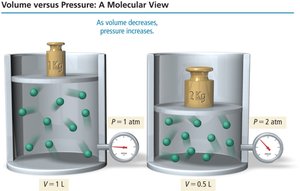
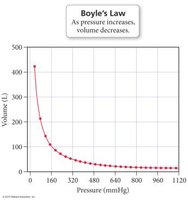
Boyle’s Law: Pressure-Volume Relationship
Boyle’s Law states that the volume of a gas is inversely proportional to its pressure at constant temperature and amount of gas:
or
As pressure increases, volume decreases by the same factor, and vice versa.
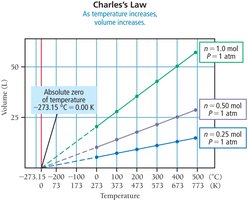
Charles’s Law: Volume-Temperature Relationship
Charles’s Law states that the volume of a fixed amount of gas at constant pressure is directly proportional to its temperature in Kelvin:
or
As temperature increases, volume increases linearly.
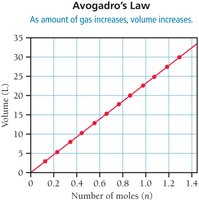
Avogadro’s Law: Volume-Mole Relationship
Avogadro’s Law states that the volume of a gas is directly proportional to the number of moles at constant temperature and pressure:
or
Equal volumes of gases at the same temperature and pressure contain equal numbers of molecules.
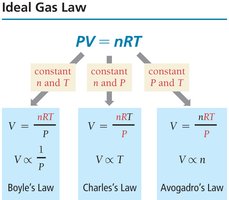
The Ideal Gas Law
Combining the Simple Gas Laws
The ideal gas law combines Boyle’s, Charles’s, and Avogadro’s laws into a single equation:
P: Pressure (atm)
V: Volume (L)
n: Moles of gas
R: Ideal gas constant,
T: Temperature (K)
Standard Temperature and Pressure (STP) and Molar Volume
STP is defined as 1 atm pressure and 273.15 K (0°C) temperature. At STP, one mole of any ideal gas occupies 22.4 L.


Density of Gases
Calculating Gas Density
Density () is the ratio of mass to volume. For gases, density is usually expressed in g/L.
At STP, molar volume is 22.4 L/mol.
For non-standard conditions, use:

Mixtures of Gases and Partial Pressures
Dalton’s Law of Partial Pressures
In a mixture of gases, each gas exerts a pressure independently of the others. The total pressure is the sum of the partial pressures:
The partial pressure of a gas can be calculated from its mole fraction and the total pressure:
Mole fraction () is the ratio of moles of component to total moles in the mixture.
Kinetic Molecular Theory
Postulates of Kinetic Molecular Theory
Gas particles are in constant, random motion.
The volume of individual particles is negligible compared to the total volume.
Collisions between particles (and with container walls) are perfectly elastic (no energy lost).
There are no significant attractive or repulsive forces between particles.
Pressure and Molecular Collisions
Pressure results from collisions of gas particles with the walls of their container.
Temperature and Kinetic Energy
The average kinetic energy of gas particles is directly proportional to the temperature in Kelvin:
As temperature increases, the average speed of particles increases.
Heavier molecules move more slowly at the same temperature.
Diffusion and Effusion
Definitions
Diffusion: The process by which gas molecules spread out from high to low concentration.
Effusion: The process by which gas molecules escape through a small hole into a vacuum.
The rate of effusion is inversely proportional to the square root of the molar mass (Graham’s Law):
Real Gases and Deviations from Ideal Behavior
Limitations of the Ideal Gas Law
At high pressures and low temperatures, real gases deviate from ideal behavior due to intermolecular attractions and the finite volume of gas particles.
Van der Waals Equation
The van der Waals equation modifies the ideal gas law to account for real gas behavior:
a: Accounts for intermolecular attractions (units: atm·L2/mol2).
b: Accounts for the finite volume of molecules (units: L/mol).
Summary Table: Gas Laws and Their Relationships
Law | Relationship | Equation | Variables Held Constant |
|---|---|---|---|
Boyle’s Law | P ∝ 1/V | n, T | |
Charles’s Law | V ∝ T | n, P | |
Avogadro’s Law | V ∝ n | P, T | |
Ideal Gas Law | PV = nRT | — |
