 Back
BackChapter 10: Gases – Properties, Laws, and Molecular Theory
Study Guide - Smart Notes

Chapter 10: Gases
Characteristics of Gases
Gases are one of the fundamental states of matter, exhibiting unique properties that distinguish them from solids and liquids. Understanding these characteristics is essential for studying their behavior and applications.
Compressibility: Gases are highly compressible and expand to fill the entire volume of their containers.
Volume Change with Pressure: When subjected to increased pressure, the volume of a gas decreases.
Homogeneous Mixtures: Gases mix evenly and completely with other gases, forming homogeneous mixtures regardless of their identities.
Independent Molecules: Each gas molecule behaves largely as if other molecules are absent, due to negligible intermolecular forces under typical conditions.
Pressure and Its Measurement
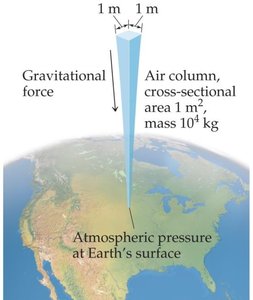
Pressure is a fundamental property of gases, defined as the force exerted per unit area. Atmospheric pressure is the force exerted by the weight of the air above a given area on Earth's surface.
Pressure Formula: , where P is pressure, F is force, and A is area.
SI Unit of Pressure: The pascal (Pa), where .
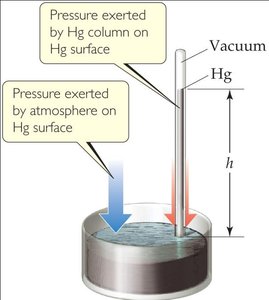
Atmospheric Pressure: Standard atmospheric pressure (1 atm) supports a 760 mm column of mercury (Hg).
Unit Conversions:
The Gas Laws
The behavior of gases can be described by several empirical laws that relate pressure (P), volume (V), temperature (T), and the amount of gas (n).
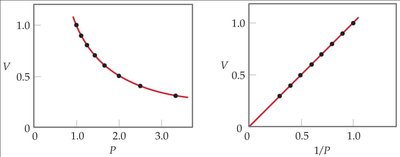
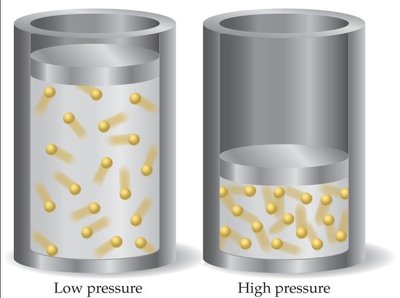
Boyle’s Law (Pressure-Volume Relationship)
At constant temperature and amount of gas, the volume of a gas is inversely proportional to its pressure.
Mathematical Expression: or
Charles’s Law (Temperature-Volume Relationship)
At constant pressure and amount of gas, the volume of a gas is directly proportional to its absolute temperature (in Kelvin).
Mathematical Expression: or
Absolute Zero: The temperature at which the volume of a gas would theoretically be zero is -273.15°C (0 K).
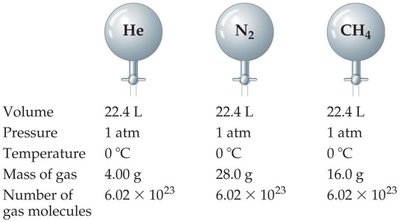
Avogadro’s Law (Quantity-Volume Relationship)
At constant temperature and pressure, the volume of a gas is directly proportional to the number of moles of gas present.
Mathematical Expression: or
Molar Volume at STP: 1 mole of any ideal gas occupies 22.4 L at 0°C and 1 atm.
The Ideal Gas Law
The ideal gas law combines Boyle’s, Charles’s, and Avogadro’s laws into a single equation that describes the state of an ideal gas.
Equation:
Gas Constant (R): (other units available; see table below)
Variables: P (pressure in atm), V (volume in L), n (moles), T (temperature in K)
Unit | Numerical Value |
|---|---|
L·atm/mol·K | 0.08206 |
J/mol·K | 8.314 |
cal/mol·K | 1.987 |
m³·Pa/mol·K | 8.314 |
L·torr/mol·K | 62.36 |
Combined Gas Law
When a gas undergoes changes in pressure, volume, and temperature, the combined gas law is used:
Equation: (for a fixed amount of gas)
Gas Densities and Molar Mass
The ideal gas law can be rearranged to calculate the density or molar mass of a gas.
Density Equation: , where δ is density, P is pressure, M is molar mass, R is the gas constant, and T is temperature in Kelvin.
Molar Mass Equation:
Gas Mixtures and Partial Pressures
In a mixture of gases, each gas exerts a pressure as if it were alone in the container. The total pressure is the sum of the partial pressures of each component (Dalton’s Law).
Dalton’s Law:
Mole Fraction: , where is moles of component 1, is total moles.
Partial Pressure:
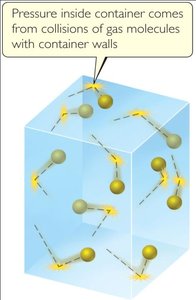
Kinetic-Molecular Theory of Gases
This theory explains the macroscopic properties of gases by considering their molecular composition and motion.
Gases consist of a large number of molecules in constant, random motion.
The combined volume of all molecules is negligible compared to the container volume.
Intermolecular forces are negligible.
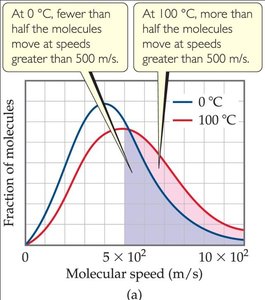
The average kinetic energy of gas molecules is proportional to the absolute temperature.
Root-Mean-Square Speed
The root-mean-square (rms) speed is the speed of a gas molecule with average kinetic energy.
Equation:
Lower molar mass results in higher rms speed at a given temperature.
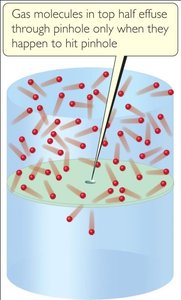
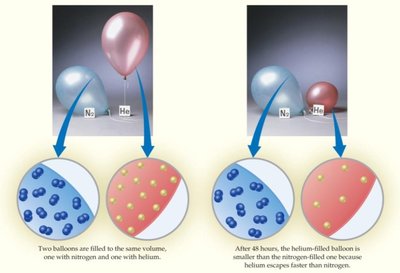
Molecular Effusion and Diffusion
Effusion is the process by which gas molecules escape through a small hole; diffusion is the mixing of gases due to molecular motion.
Graham’s Law of Effusion:
The rate of effusion is inversely proportional to the square root of the molar mass.
Diffusion and Mean Free Path
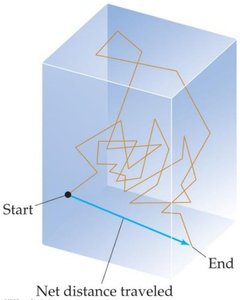
Diffusion is the spread of one substance throughout another, slowed by molecular collisions. The mean free path is the average distance a molecule travels between collisions.
At sea level, the mean free path for air molecules is about 60 nm; at higher altitudes, it increases significantly.
Increasing pressure decreases the mean free path; increasing temperature increases it.
Additional info: These notes cover all major aspects of the behavior of gases as outlined in a standard general chemistry curriculum, including the physical properties, mathematical relationships, and molecular theory explanations.
