 Back
BackChapter 10: Gases – Structure, Properties, and Laws
Study Guide - Smart Notes

Gases: Structure and Properties
What Is a Gas?
Gases are a phase of matter composed of particles (atoms or molecules) that move randomly and rapidly within their container. Gas particles travel in straight lines until they collide with the container wall or another particle, after which they bounce off. These collisions exert force, which is experienced as pressure.
Random motion: Gas particles move in all directions.
Collisions: Collisions with surfaces and other particles create pressure.
Low density: Gases have much lower density than solids or liquids.

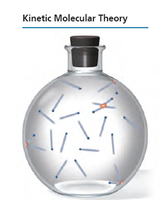
Kinetic Molecular Theory
The kinetic molecular theory is a model describing the behavior of gases as a collection of particles in constant motion. It explains gas properties and the relationships between pressure, volume, temperature, and amount.
Postulates:
Gas particles are always moving.
Attraction between particles is negligible.
Collisions are elastic (no energy loss).
There is much empty space between particles.
Average kinetic energy: Proportional to temperature in Kelvin.
Elastic collisions: Energy is exchanged but not lost overall.
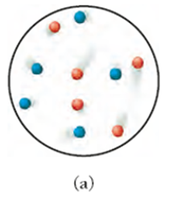
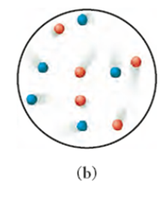
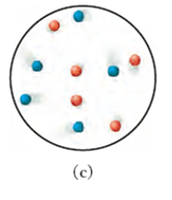
Conceptual Connection: Gas Mixtures
Equal molar amounts of different gases (e.g., argon and xenon) will have equal numbers of particles, but their velocities may differ due to mass differences. The kinetic molecular theory predicts that lighter particles move faster than heavier ones at the same temperature.
Example: Argon (red dots) and xenon (blue dots) in a mixture.
Gas Pressure
Pressure is the force exerted per unit area by gas molecules as they strike surfaces. It depends on the number of gas particles, the volume of the container, and the average speed of the particles.
Pressure formula:
Factors: More particles or higher speed increases pressure; larger volume decreases pressure.
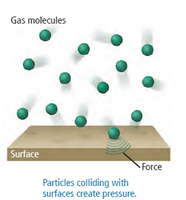
Factors Affecting Gas Pressure
The concentration of gas molecules, number of particles, and volume all affect pressure. Higher concentration or more particles in a given volume increases pressure, while increasing volume decreases concentration and pressure.
Low density: Low pressure.
High density: High pressure.

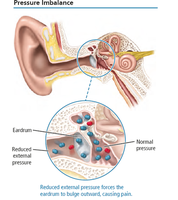
Pressure Imbalance in the Ear
Pressure differences across the eardrum can cause discomfort or injury, such as a "popped eardrum." This occurs when external and internal pressures are not equal.
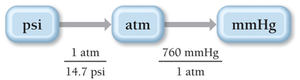
Pressure Units and Conversion
Common units of pressure include millimeters of mercury (mmHg), torr, atmospheres (atm), and pounds per square inch (psi). Conversion between units is essential for calculations.
1 mmHg = 1 torr
760 mmHg = 1 atm
1 atm = 14.7 psi
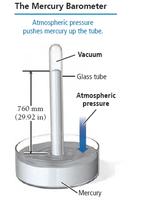
Measuring Gas Pressure: The Manometer
A manometer is a U-shaped tube used to measure the pressure of a gas in a container. The difference in liquid levels indicates the pressure difference between the gas and the atmosphere.
The Simple Gas Laws
Boyle’s Law: Pressure and Volume
Boyle’s law states that the volume of a gas is inversely proportional to its pressure when temperature and amount are constant.
Equation:
Graph: P vs. V is a curve; P vs. 1/V is a straight line.
Application: Diving – as pressure decreases, volume increases.
Charles’s Law: Volume and Temperature
Charles’s law states that the volume of a gas is directly proportional to its absolute temperature (in Kelvin) at constant pressure and amount.
Equation:
Graph: V vs. T is a straight line.
Absolute zero: Theoretical temperature where volume is zero.
Avogadro’s Law: Volume and Moles
Avogadro’s law states that the volume of a gas is directly proportional to the number of moles of gas at constant temperature and pressure.
Equation:
Equal volumes: Equal numbers of molecules, regardless of gas type.
The Ideal Gas Law
Ideal Gas Law: PV = nRT
The ideal gas law combines the simple gas laws into one equation relating pressure, volume, temperature, and amount of gas.
Equation:
Variables: P (pressure, atm), V (volume, L), n (moles), R (gas constant), T (temperature, K)
Kinetic Molecular Theory and the Ideal Gas Law
The kinetic molecular theory provides a quantitative model for the ideal gas law, relating the motion and collisions of gas particles to pressure and volume.
Standard Conditions and Molar Volume
Standard temperature and pressure (STP) are defined as 273 K and 1 atm. At STP, 1 mole of an ideal gas occupies 22.4 L, known as the molar volume.
Density and Molar Mass of Gases
Density of a Gas
Density is the ratio of mass to volume. For gases, density is often given in grams per liter (g/L). The density of a gas can be calculated using the ideal gas law and molar mass.
Equation:
Direct proportionality: Density increases with molar mass and pressure.
Mixtures of Gases and Partial Pressures
Dalton’s Law of Partial Pressures
Dalton’s law states that the total pressure of a gas mixture is the sum of the partial pressures of its components. Each gas behaves independently, and its partial pressure can be calculated using the ideal gas law.
Equation:
Mole fraction:
Partial pressure:
Collecting Gases Over Water
When gases are collected over water, the total pressure includes both the gas and water vapor. The partial pressure of water vapor depends on temperature and must be subtracted to find the pressure of the dry gas.
Molecular Velocities and Kinetic Energy
Temperature and Molecular Velocities
The average kinetic energy of gas molecules depends on their mass and velocity. At the same temperature, lighter molecules move faster than heavier ones.
Root mean square velocity:
R: Gas constant in energy units
M: Molar mass in kg/mol
Diffusion, Effusion, and Graham’s Law
Diffusion and Effusion
Diffusion is the spread of gas molecules from high to low concentration. Effusion is the escape of gas molecules through a small hole into a vacuum. Both rates are related to the root mean square velocity and inversely proportional to the square root of molar mass.
Graham’s Law of Effusion
Graham’s law relates the rates of effusion of two gases to their molar masses:
Equation:
Stoichiometry of Gaseous Reactions
Gases in Chemical Reactions
Stoichiometric calculations involving gases use the ideal gas law to relate volumes, pressures, and temperatures to moles. At STP, 1 mol = 22.4 L.
Real Gases and Deviations from Ideal Behavior
Real Gases
Real gases deviate from ideal behavior at high pressure or low temperature. The ideal gas law assumes no attractions and negligible volume, but real molecules have finite size and intermolecular forces.
Van Der Waals Equation
The van der Waals equation modifies the ideal gas law to account for molecular volume and intermolecular attractions:
Equation:
a: Constant for intermolecular attractions
b: Constant for molecular volume
Summary Review: Properties of Gases
Expand to fill their container
Take the shape of their container
Low density and compressibility
Mixtures are always homogeneous
