 Back
BackChapter 11: Gases – Properties, Laws, and Applications
Study Guide - Smart Notes

Gases
Introduction to Gases
Gases are one of the fundamental states of matter, characterized by their ability to expand and fill any container, low density, and high compressibility. Understanding the behavior of gases is essential for explaining many everyday phenomena and for applications in chemistry and industry.
Kinetic Molecular Theory
Basic Postulates of Kinetic Molecular Theory
The kinetic molecular theory provides a model for understanding the behavior of gases. It is based on several key assumptions:
Constant, straight-line motion: Gas particles are in continuous, random motion.
No attractions or repulsions: Gas particles do not interact except during elastic collisions.
Large spaces between particles: The volume of the particles is negligible compared to the volume of the container.
Kinetic energy and temperature: The average kinetic energy of gas particles is directly proportional to the temperature in kelvin.
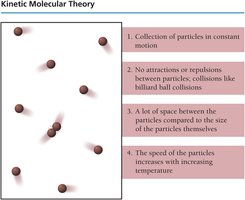
Examples and Applications: The kinetic molecular theory explains why gases are compressible, why they expand to fill their containers, and why they have low densities compared to liquids and solids.
Properties of Gases
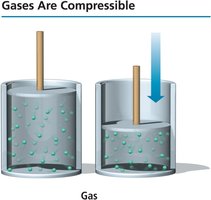
Compressibility
Gases are highly compressible due to the large amount of empty space between particles. When pressure is applied, the particles are forced closer together, reducing the volume.
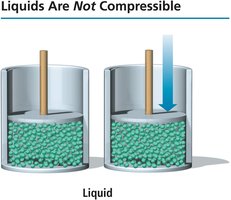
Incompressibility of Liquids
In contrast, liquids are not compressible because their particles are much closer together, leaving little empty space.
Expansion to Fill Container
Gases expand to fill the shape and volume of their container because the attractions between molecules are negligible and the particles are in constant motion.

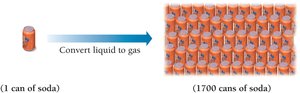
Low Density
Gases have much lower densities than liquids and solids. For example, converting the liquid in a soda can to gas would fill about 1700 cans of the same size.
Pressure and Its Measurement
Definition of Pressure
Pressure is defined as the force exerted per unit area by gas particles as they collide with surfaces. The more frequent and forceful the collisions, the higher the pressure.
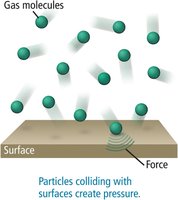
Pressure Differences and Everyday Examples
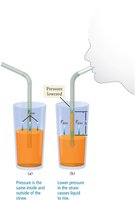
Drinking through a straw works because sucking lowers the pressure inside the straw, allowing atmospheric pressure to push the liquid up.
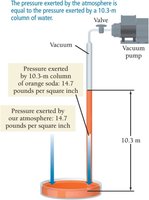
Atmospheric pressure can only push a column of water (or soda) up to about 10.3 meters, which is why extra-long straws do not work beyond this height.
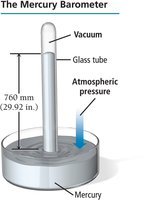
Units of Pressure
Atmosphere (atm): Average pressure at sea level.
Pascals (Pa): SI unit, where 1 Pa = 1 N/m2.
Millimeters of mercury (mm Hg) or Torr: Based on the height of a mercury column in a barometer; 1 atm = 760 mm Hg = 760 Torr.
Pounds per square inch (psi): Common in engineering contexts.
Pressure, Force, and Area
The relationship between pressure, force, and area is given by:
where P is pressure, F is force, and A is area.
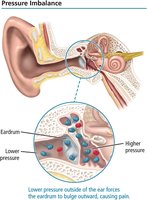
Effect of Altitude on Pressure
Atmospheric pressure decreases with altitude, which can cause discomfort such as ear pain due to pressure imbalances.
Gas Laws
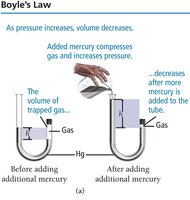
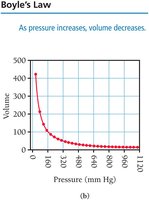
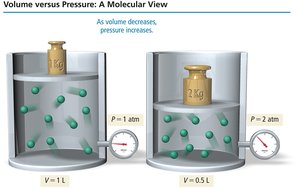
Boyle’s Law: Pressure and Volume
Boyle’s law states that the volume of a gas is inversely proportional to its pressure at constant temperature and amount of gas:
or
For two sets of conditions:
Example: If a gas at 4.0 atm occupies 6.0 L, what volume will it occupy at 1.0 atm? Using Boyle’s law: L.
Charles’s Law: Volume and Temperature
Charles’s law states that the volume of a gas is directly proportional to its temperature (in kelvin) at constant pressure and amount of gas:
or

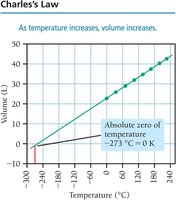
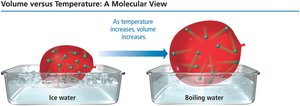
Absolute zero is the temperature at which the volume of a gas would theoretically be zero (0 K or -273°C).
Combined Gas Law
The combined gas law relates pressure, volume, and temperature for a fixed amount of gas:
This law is useful when more than one variable changes.
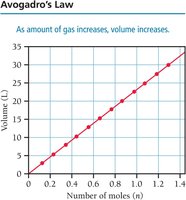
Avogadro’s Law: Volume and Moles
Avogadro’s law states that the volume of a gas is directly proportional to the number of moles (n) at constant temperature and pressure:
or

The Ideal Gas Law
The ideal gas law combines Boyle’s, Charles’s, and Avogadro’s laws into a single equation:
P: Pressure (atm)
V: Volume (L)
n: Moles of gas
R: Ideal gas constant (0.0821 L·atm/mol·K)
T: Temperature (K)
Example: Calculate the volume occupied by 0.845 mol of nitrogen gas at 1.37 atm and 315 K.
L
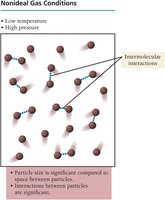
Deviations from Ideal Gas Behavior
Gases deviate from ideal behavior at low temperatures and high pressures, where intermolecular forces and the volume of particles become significant.

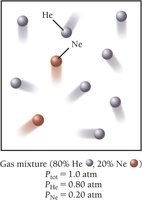
Mixtures of Gases and Partial Pressures
Dalton’s Law of Partial Pressures
In a mixture of gases, each gas exerts its own pressure, called partial pressure. Dalton’s law states that the total pressure is the sum of the partial pressures of all components:
The partial pressure of a component is its fractional composition times the total pressure:
Stoichiometry Involving Gases
Gas Volumes in Chemical Reactions
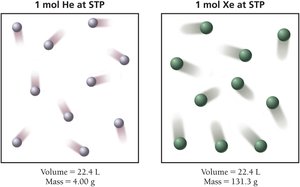
In reactions involving gases, the ideal gas law can be used to relate the volume, pressure, temperature, and amount of gaseous reactants or products. At standard temperature and pressure (STP: 0°C, 1 atm), 1 mol of any ideal gas occupies 22.4 L.
Summary Table: Gas Laws
Law | Relationship | Equation | Constants |
|---|---|---|---|
Boyle’s Law | P ∝ 1/V | n, T | |
Charles’s Law | V ∝ T | n, P | |
Avogadro’s Law | V ∝ n | P, T | |
Combined Gas Law | — | n | |
Ideal Gas Law | — | — |
Chemistry in the Environment: Air Pollution
Major Gaseous Pollutants
Sulfur dioxide (SO2): Emitted from electricity generation and metal refining; causes respiratory irritation and acid rain.
Carbon monoxide (CO): Produced by incomplete combustion; can displace oxygen in blood, causing health risks.
Ozone (O3): Upper atmospheric ozone protects from UV light; ground-level ozone is a pollutant and lung irritant.
Nitrogen dioxide (NO2): Emitted by vehicles and power plants; causes smog and respiratory issues.
Legislation such as the Clean Air Act has significantly reduced pollutant levels in the U.S. over the past decades.
Review
Kinetic molecular theory explains the properties of gases.
Pressure is the result of molecular collisions with surfaces.
Gas laws describe the relationships among pressure, volume, temperature, and amount of gas.
The ideal gas law unifies these relationships in a single equation.
Dalton’s law describes the behavior of mixtures of gases.
Gas stoichiometry allows calculation of reactant and product volumes under specified conditions.
