 Back
BackChapter 12: Chemical Kinetics – Study Guide
Study Guide - Smart Notes

Chapter 12: Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the rates at which chemical reactions occur and the factors that affect these rates. Understanding kinetics is essential for predicting how fast reactions proceed and for controlling industrial, biological, and environmental processes.
Reaction Rate and Its Expression
Basic Information of Reaction Rate
The reaction rate is the change in concentration of a reactant or product per unit time. It is typically expressed in units of molarity per second (M/s).
Rate Law: An equation that relates the reaction rate to the concentrations of reactants.
Rate Constant (k): A proportionality constant specific to each reaction at a given temperature.
Integrated Rate Laws
Integrated Rate Laws: Concentration Changes Over Time
Integrated rate laws describe how the concentration of reactants changes with time for different reaction orders. The variable 't' represents time, '[A]0' is the initial concentration, and '[A]t' is the concentration at time t.
First-order:
Second-order:
Zero-order:
These equations allow calculation of reactant concentrations at any time during the reaction.
![First-order reaction plot: ln[A] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/65693b90_image_2.png)
![Second-order reaction plot: 1/[A] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/65693b90_image_3.png)
![Zero-order reaction plot: [A] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/65693b90_image_4.png)
Graphical Methods for Determining Reaction Order
Graphical Determination of Reaction Order
Reaction order can be determined by plotting concentration data in different ways:
First-order: Plotting vs. time yields a straight line.
Second-order: Plotting vs. time yields a straight line.
Zero-order: Plotting vs. time yields a straight line.

Reaction Half-Life
Half-Life of a Reaction
The half-life (t1/2) is the time required for the concentration of a reactant to decrease to half its initial value. For first-order reactions, the half-life is constant and independent of initial concentration:
First-order:
Second-order:
Zero-order:
Radioactive decay is a classic example of a first-order process.
![Plot of [N2O5] vs. time for three reaction half-lives](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/65693b90_image_6.png)
Theories of Chemical Kinetics
Collision Theory and Concentration
Collision theory states that molecules must collide to react. The rate increases with higher concentration because more collisions occur. The number of collisions is proportional to the product of reactant concentrations.
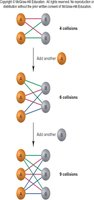
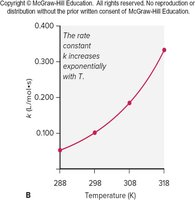
Temperature and the Rate Constant
Temperature significantly affects reaction rates. As temperature increases, the rate constant (k) increases exponentially, as described by the Arrhenius equation:
A = frequency factor, = activation energy, R = gas constant (8.314 J/mol·K), T = temperature in Kelvin
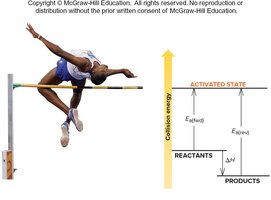
Activation Energy
Activation energy () is the minimum energy required for a reaction to occur. Effective collisions must exceed this threshold. Lower activation energy results in a faster reaction.
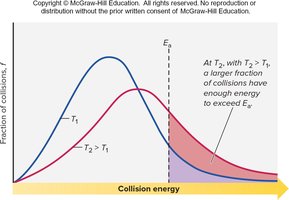
Temperature and Collision Energy
Increasing temperature raises the kinetic energy of particles, increasing the fraction of collisions with energy greater than and thus increasing the reaction rate.
Arrhenius Equation and Activation Energy Calculation
The Arrhenius equation can be used to calculate activation energy if rate constants are known at two temperatures:
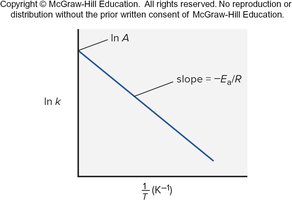
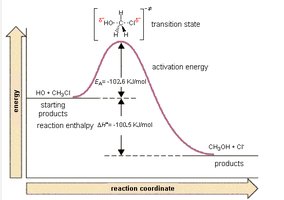
Transition State Theory
Transition State and Activated Complex
The transition state is a high-energy, unstable arrangement of atoms formed during a reaction. It exists at the peak of the energy profile and cannot be isolated. The energy required to reach the transition state is the activation energy.
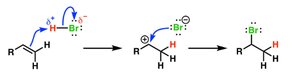
Reaction Mechanisms
Elementary Steps and Molecularity
A reaction mechanism is a sequence of elementary steps. Each step has a molecularity (number of particles involved): unimolecular, bimolecular, or termolecular. Termolecular steps are rare due to the low probability of three particles colliding simultaneously.
Example: Determining Molecularities and Rate Laws
For the reaction:
1. Cl2(g) → 2Cl(g)
2. Cl(g) + CHCl3(g) → HCl(g) + CCl3(g)
3. Cl(g) + CCl3(g) → CCl4(g)
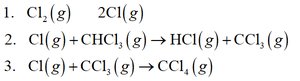
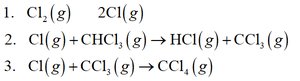
Intermediates
An intermediate is a species formed in one step and consumed in another. It is stable and can sometimes be isolated, unlike the transition state.
Rate-Determining Step
The slowest step in a mechanism is the rate-determining step. The overall rate law is determined by this step, and intermediates must be expressed in terms of reactant concentrations.
Correlating Mechanism with Rate Law
A valid mechanism must:
Sum to the overall balanced equation
Have reasonable elementary steps (usually unimolecular or bimolecular)
Correlate with the observed rate law
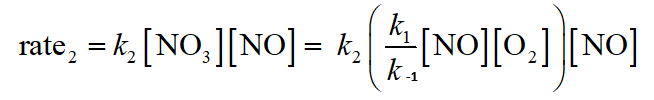
Catalysis: Speeding Up a Reaction
Role of Catalysts
A catalyst increases reaction rate by providing an alternative pathway with lower activation energy. It is not consumed in the reaction and does not affect the overall yield or enthalpy change (ΔH).
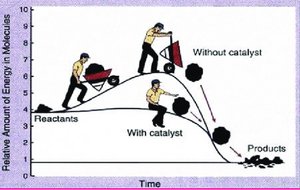

Examples of Catalysis
Homogeneous Catalysis: The catalyst is in the same phase as the reactants. Example: Decomposition of H2O2 catalyzed by Br-.



Heterogeneous Catalysis: The catalyst is in a different phase than the reactants. Example: Metal-catalyzed hydrogenation of ethene.
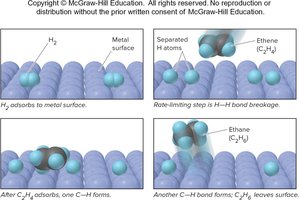
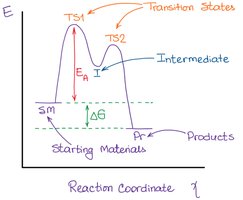
Transition State vs. Intermediate
Differences Between Transition State and Intermediate
The transition state is a fleeting, high-energy configuration at the peak of the reaction coordinate, not isolatable. An intermediate is a relatively stable species that can sometimes be detected or isolated.
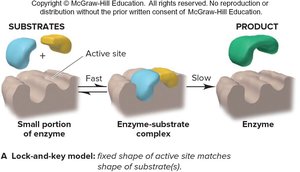
Lock-and-Key Model of Enzyme Action
Enzyme Catalysis
The lock-and-key model describes how enzymes catalyze reactions by providing a specific active site for substrates, lowering activation energy and increasing reaction rate.
Summary Table: Integrated Rate Laws and Half-Lives
Order | Integrated Rate Law | Half-Life Equation |
|---|---|---|
First-order | ||
Second-order | ||
Zero-order |
Key Points for Exam Preparation
Know the integrated rate laws and how to use them for calculations.
Understand how to determine reaction order from plots.
Be able to solve half-life questions.
Recognize the effect of temperature, orientation, and catalysis on reaction rates.
Write rate laws based on reaction mechanisms, using the rate-determining step.
Understand the difference between transition states and intermediates.
Know how catalysts affect reaction rates and energy profiles.
