 Back
BackChapter 13: Solutions – Structure, Properties, and Colligative Effects
Study Guide - Smart Notes

Solutions: Structure and Properties
Introduction to Solutions
Solutions are homogeneous mixtures composed of two or more substances. The process of solution formation is governed by the interactions between solute and solvent particles, and the tendency of nature toward spontaneous mixing (entropy).
Solute: The substance being dissolved.
Solvent: The substance doing the dissolving, usually present in greater quantity.
Homogeneous mixture: Uniform composition throughout.
Spontaneous mixing: Driven by entropy, leading to uniform distribution of solute.
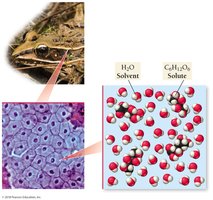
Example: Wood frogs survive freezing temperatures by flooding their blood with glucose, lowering the freezing point of their body fluids.
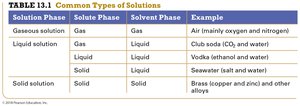
Common Types of Solutions
Solutions can exist in various combinations of solute and solvent phases. The table below summarizes common types:
Solution Phase | Solute Phase | Solvent Phase | Example |
|---|---|---|---|
Gaseous solution | Gas | Gas | Air (O2 and N2) |
Liquid solution | Gas | Liquid | Club soda (CO2 in water) |
Liquid solution | Liquid | Liquid | Vodka (ethanol and water) |
Solid solution | Solid | Solid | Brass (copper and zinc) |
Solubility and Miscibility
Solubility is the maximum amount of solute that can dissolve in a given amount of solvent. Miscible liquids are mutually soluble, while immiscible liquids are not.
Soluble: Salt in water.
Insoluble: Oil in water.
Miscible: Alcohol and water.
Immiscible: Oil and water.
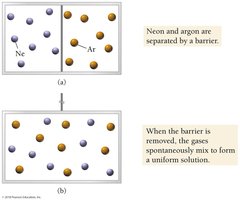
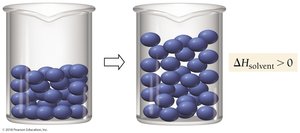
Nature’s Tendency Toward Mixing: Entropy
Entropy and Solution Formation
Mixing increases entropy, which is the measure of energy dispersal in a system. Even when potential energy does not decrease, mixing occurs spontaneously due to entropy.
Entropy: Drives spontaneous mixing.
Example: Mixing of neon and argon gases after removing a barrier.
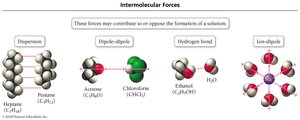
Intermolecular Forces and Solution Formation
Types of Intermolecular Forces
The formation of solutions depends on the types and strengths of intermolecular forces between solute and solvent particles.
Dispersion forces: Present in all molecules, especially nonpolar.
Dipole-dipole forces: Between polar molecules.
Hydrogen bonding: Strong dipole-dipole interaction involving H, N, O, or F.
Ion-dipole forces: Between ions and polar molecules.
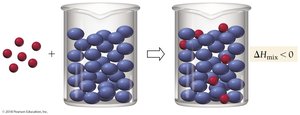
Solution Interactions
To form a solution, solute–solute and solvent–solvent interactions must be overcome, and new solute–solvent interactions must be formed.
Solute–solute interactions: Must be broken (endothermic).
Solvent–solvent interactions: Must be broken (endothermic).
Solute–solvent interactions: Formed (exothermic).
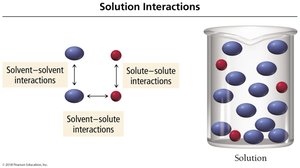
Relative Interactions and Solution Formation
The table below summarizes how the relative strengths of interactions affect solution formation:
Solvent–solute interactions | Solvent–solvent and solute–solute interactions | Result |
|---|---|---|
> | Solution generally forms | |
= | Solution generally forms | |
< | Solution may or may not form |

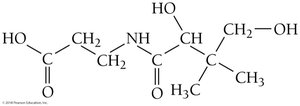
Solubility of Vitamins: Polar vs. Nonpolar
Classifying Vitamins by Solubility
Vitamins can be classified as water-soluble or fat-soluble based on their molecular structure and polarity.
Water-soluble: Highly polar, can hydrogen-bond with water (e.g., Vitamin C, Vitamin B5).
Fat-soluble: Mostly nonpolar, dissolve in fats (e.g., Vitamin K3, Vitamin A).
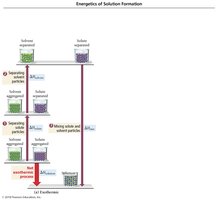
Energetics of Solution Formation
Enthalpy of Solution
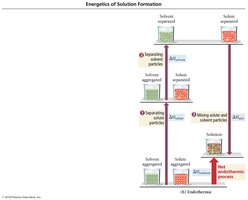
The overall enthalpy change for making a solution depends on three steps:
1. ΔHsolute: Overcome attractions between solute particles (endothermic).
2. ΔHsolvent: Overcome attractions between solvent particles (endothermic).
3. ΔHmix: Form new attractions between solute and solvent (exothermic).
Overall equation:

Exothermic vs. Endothermic Solution Formation
If the energy released in forming solute–solvent attractions is greater than the energy required to break solute–solute and solvent–solvent attractions, the process is exothermic. If less, it is endothermic.
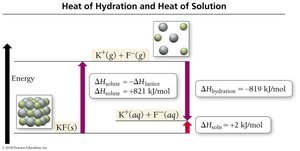
Heats of Hydration
For ionic compounds in water, the heat of hydration combines the energy to overcome water–water attractions and the energy released in forming ion–water attractions.
Lattice energy: Energy holding ions together in a solid.
Heat of hydration: Energy released when ions dissolve in water.
Solution Equilibrium and Solubility Limit
Dynamic Equilibrium
When a solution reaches saturation, the rate of dissolution equals the rate of deposition, resulting in dynamic equilibrium.
Saturated: Maximum solute dissolved.
Unsaturated: Less than maximum solute.
Supersaturated: More than maximum solute; unstable.
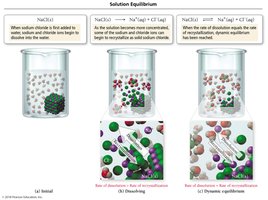

Purification by Recrystallization
Recrystallization is a method to purify solids by dissolving them in hot solvent and allowing them to crystallize upon cooling, leaving impurities behind.

Temperature and Pressure Dependence of Solubility
Solubility of Solids and Gases
Solubility of solids generally increases with temperature, while solubility of gases decreases with temperature.
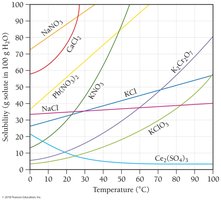

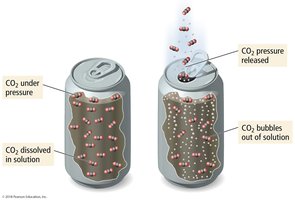
Supersaturation and Soda Pop Example
Supersaturated solutions can be formed by changing temperature or pressure. For example, warm soda pop fizzes more because CO2 is less soluble at higher temperatures.

Pressure Dependence: Henry’s Law
The solubility of a gas in a liquid is directly proportional to its partial pressure above the liquid, described by Henry’s Law:
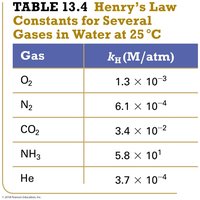
Gas | kH (M/atm) |
|---|---|
O2 | 1.3 × 10–3 |
N2 | 6.1 × 10–4 |
CO2 | 3.4 × 10–2 |
NH3 | 5.8 × 101 |
He | 3.7 × 10–4 |
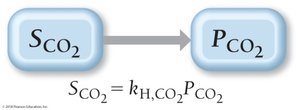
Solution Concentration Units
Common Concentration Units
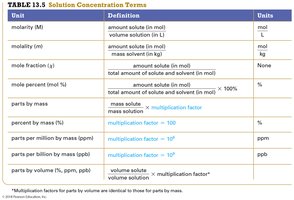
Concentration describes the amount of solute in a given amount of solution. Common units include molarity, molality, mass percent, volume percent, ppm, and ppb.
Unit | Definition | Units |
|---|---|---|
Molarity (M) | amount solute (mol) / volume solution (L) | mol/L |
Molality (m) | amount solute (mol) / mass solvent (kg) | mol/kg |
Mole fraction (χ) | moles of component / total moles | None |
Mass percent (%) | mass solute / mass solution × 100% | % |
Parts per million (ppm) | mass solute / mass solution × 106 | ppm |
Parts per billion (ppb) | mass solute / mass solution × 109 | ppb |
Preparing Solutions
To prepare a solution of known concentration, calculate the required mass of solute and dissolve it in solvent to reach the desired volume.
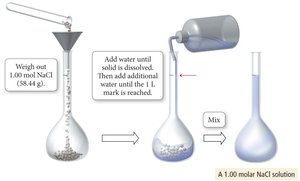
Molarity and Dissociation
Molarity allows calculation of the concentration of ions in solution after dissociation. For example, 1.0 M CaCl2 yields 1.0 M Ca2+ and 2.0 M Cl–.
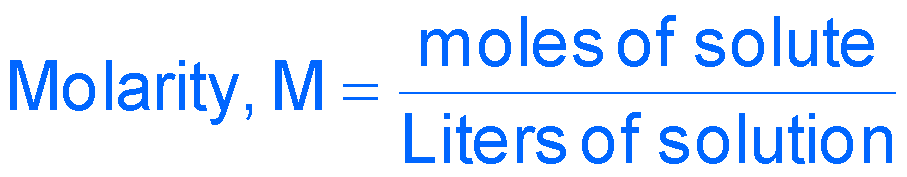
Percent, ppm, and ppb Calculations
Percent, ppm, and ppb are used for very dilute solutions. The formulas are:
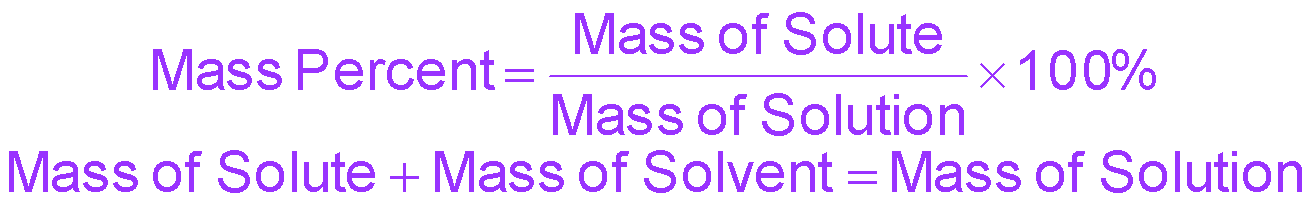
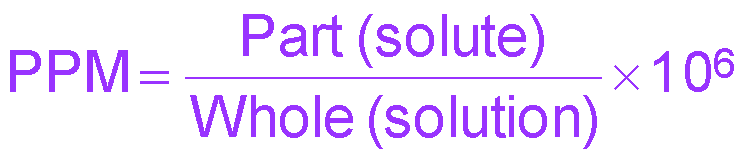
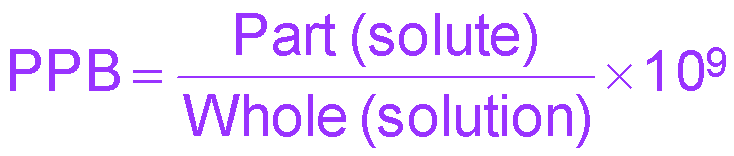
Colligative Properties
Definition and Importance
Colligative properties depend only on the number of solute particles, not their identity. These include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.
Vapor Pressure Lowering and Raoult’s Law
The vapor pressure of a solvent in a solution is always lower than that of the pure solvent. Raoult’s Law describes this relationship:
Freezing Point Depression
The freezing point of a solution is lower than that of the pure solvent. The change is proportional to the molality:
Boiling Point Elevation
The boiling point of a solution is higher than that of the pure solvent. The change is proportional to the molality:
Osmosis and Osmotic Pressure
Osmosis is the flow of solvent from low to high concentration through a semipermeable membrane. Osmotic pressure is given by:
Van’t Hoff Factor
The van’t Hoff factor, i, accounts for the number of particles produced by dissociation of ionic compounds:
Colligative Properties in Medical Solutions
Isosmotic, hyperosmotic, and hyposmotic solutions affect cells by controlling the net flow of water, which is critical in medical applications.
Additional info: This study guide covers all major aspects of Chapter 13: Solutions, including structure, properties, energetics, concentration units, and colligative effects, with relevant examples and formulas for exam preparation.
