 Back
BackChapter 13: Solutions – Structure, Properties, and Effects
Study Guide - Smart Notes

Solutions
Definition and Components
A solution is a homogeneous mixture composed of two or more substances. The solvent is the majority component, while the solute is the minority component. Solutions form due to intermolecular forces between the solute and solvent, which drive mixing and energy dispersal.
Solvent: The substance present in greater amount.
Solute: The substance present in lesser amount.
Homogeneous mixture: Appears as a single phase, though it contains multiple materials.
Example: Seawater is a homogeneous mixture of salts (solutes) dissolved in water (solvent).
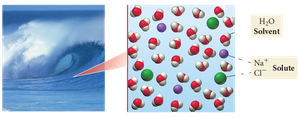
Nature favors spontaneous mixing because uniform mixing is energetically favorable.

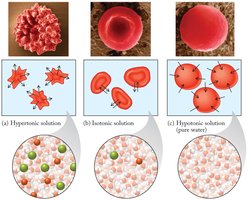
Osmosis
Osmosis is the spontaneous migration of water through semipermeable membranes, which allow water to pass but not solute particles. Water moves from regions of high concentration (low solute) to low concentration (high solute).
Isotonic: Equal solute concentration inside and outside the cell.
Hypertonic: Higher solute concentration outside the cell; water leaves the cell.
Hypotonic: Lower solute concentration outside the cell; water enters the cell.
Solubility
Solubility and Miscibility
Solubility is the maximum amount of solute that can dissolve in a given amount of solvent. Two liquids that are mutually soluble are called miscible. The solubility of a substance depends on the tendency toward mixing and the types of intermolecular forces present.
Polar substances dissolve in polar solvents; nonpolar substances dissolve in nonpolar solvents. This is summarized as "like dissolves like."
Solubility is influenced by temperature and pressure.
Influences on Solution Formation
Mixing is favored by entropy (ΔS), which is a measure of energy dispersal. Most physical systems tend toward lower potential energy, but solution formation is often driven by the increase in entropy rather than a decrease in potential energy.
Intermolecular Forces
Energy changes during solution formation involve differences in attractive forces between particles:
Dispersion forces: Present in nonpolar molecules (e.g., heptane, pentane).
Dipole-dipole forces: Present in polar molecules (e.g., acetone, chloroform).
Hydrogen bonding: Strongest in molecules like ethanol and water.
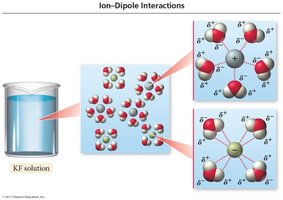
Ion-dipole forces: Occur when ions dissolve in polar solvents like water.
Energetics of Solution Formation
The process of dissolving involves three steps:
Overcoming solute-solute attractions (ΔHsolute, endothermic)
Overcoming solvent-solvent attractions (ΔHsolvent, endothermic)
Forming solute-solvent attractions (ΔHmix, exothermic)
The overall enthalpy change for solution formation is:

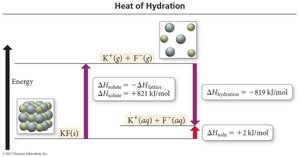
Ion-Dipole Interactions and Heat of Hydration
When ions dissolve in water, they become hydrated, surrounded by water molecules. The formation of ion-dipole attractions releases a significant amount of energy, known as the heat of hydration (ΔHhydration).
For ionic compounds, the lattice energy must be overcome:
Temperature and Pressure Effects on Solubility
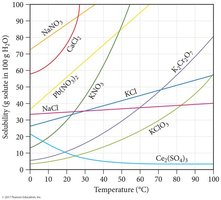
Solubility of Solids
Solubility is often expressed as grams of solute per 100 g of water. For most solids, solubility increases with temperature if the dissolution is endothermic.
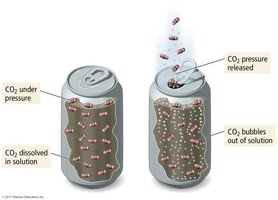
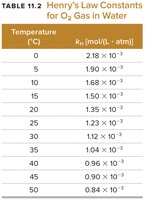
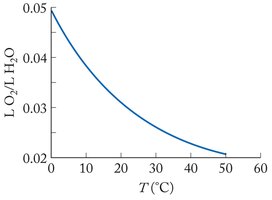
Solubility of Gases
Gases generally have lower solubility in water than ionic or polar solids. For all gases, solubility decreases as temperature increases, and increases as partial pressure increases.

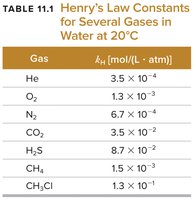
The solubility of a gas is given by Henry's Law:
Sgas: Solubility of the gas
kH: Henry's law constant
Pgas: Partial pressure of the gas
Colligative Properties
Vapor Pressure Lowering
The vapor pressure of a solvent above a solution is lower than that of the pure solvent. This is a colligative property, meaning it depends only on the number of solute particles, not their identity.


Raoult's Law describes this effect:
Xsolvent: Mole fraction of solvent
Psolvent: Vapor pressure of pure solvent
Boiling Point Elevation and Freezing Point Depression
The decrease in vapor pressure leads to an increase in boiling point and a decrease in freezing point of the solution compared to the pure solvent.
Boiling Point Elevation:
Freezing Point Depression:
m: Molality of solute particles
Kb, Kf: Boiling and freezing point constants
Concentration Units
Molarity and Molality
Molarity (M) is moles of solute per liter of solution and is temperature dependent. Molality (m) is moles of solute per kilogram of solvent and is not temperature dependent.
Molarity:
Molality:
Van’t Hoff Factor and Ion Pairing
Van’t Hoff Factor (i)
The van’t Hoff factor (i) accounts for the number of particles produced by an ionic compound in solution. For non-electrolytes, i = 1. For electrolytes, i > 1 due to dissociation.
Boiling point elevation and freezing point depression equations are corrected for electrolytes:
Ion Pairing
Cations and anions may cluster together in solution, forming ion pairs, which reduces the effective number of solute particles.
Osmotic Pressure
Osmotic Pressure (Π)
Osmotic pressure is the pressure required to prevent the flow of water across a semipermeable membrane. It is directly proportional to the molarity of solute particles:
i: van’t Hoff factor
M: Molarity of solute particles
R: Gas constant (0.08206 L·atm/(mol·K))
T: Absolute temperature (K)
Visual Examples and Applications
Freezing Point, Osmosis, and Temperature Effects
Visual examples help illustrate the effects of solute concentration on freezing point, osmotic flow, and temperature changes in solutions.
*Additional info: Academic context and expanded explanations were added to ensure completeness and clarity for exam preparation. All included images are directly relevant to the adjacent paragraphs and reinforce the educational content.*
