 Back
BackChapter 14: Chemical Kinetics – Mini-Textbook Study Notes
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the speed at which chemical reactions occur and the factors that influence these rates. Understanding kinetics is essential for predicting how quickly reactions proceed and for controlling industrial and biological processes.
Reaction rate: The speed at which reactants are converted to products.
Mechanism: The step-by-step sequence of elementary reactions by which overall chemical change occurs.
Factors Affecting Reaction Rates
Several factors influence how fast a chemical reaction proceeds:
Physical state of reactants: Homogeneous reactions (all reactants in the same phase) are typically faster than heterogeneous reactions (different phases). Increased surface area in solids accelerates reactions.
Reactant concentrations: Higher concentrations lead to more frequent collisions and faster reactions.
Temperature: Raising temperature increases kinetic energy, resulting in more collisions and higher reaction rates.
Presence of a catalyst: Catalysts alter the reaction mechanism and increase rate without being consumed.
Measuring Reaction Rates
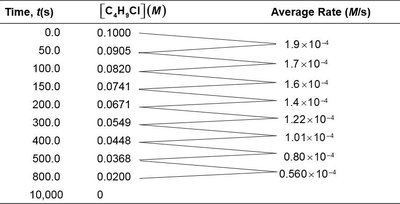
Rate Definitions and Types
Reaction rate is defined as the change in concentration of a reactant or product per unit time:
Average rate: Change over a finite time interval.
Instantaneous rate: Rate at a specific moment, given by the slope of the concentration vs. time curve.
Initial rate: Rate at the very start of the reaction (t = 0).
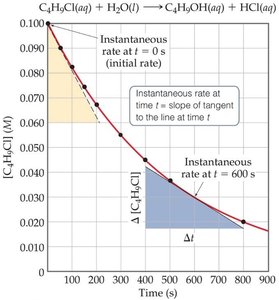
Relative Rates and Stoichiometry
The rate at which reactants are consumed and products are formed depends on the stoichiometry of the reaction. For example, in a 1:1 reaction, the rate of disappearance of reactant equals the rate of appearance of product (with opposite signs). For other ratios, rates are related by stoichiometric coefficients.
Rate Laws and Reaction Order
Determining Rate Laws
Rate laws express the relationship between reaction rate and reactant concentrations. The general form is: where k is the rate constant, and m, n are the orders with respect to each reactant.
Orders are determined experimentally, not from the balanced equation.
The sum of individual orders gives the overall reaction order.
Example: Ammonium and Nitrite Reaction
Experimental data shows that doubling the concentration of either reactant doubles the rate, indicating first-order dependence for each.
Experiment | [NH4+] (M) | [NO2-] (M) | Initial Rate (M/s) |
|---|---|---|---|
1 | 0.0100 | 0.200 | 5.4 × 10-7 |
2 | 0.0200 | 0.200 | 10.8 × 10-7 |
3 | 0.0400 | 0.200 | 21.5 × 10-7 |
4 | 0.200 | 0.0202 | 10.8 × 10-7 |
5 | 0.200 | 0.0404 | 21.6 × 10-7 |
6 | 0.200 | 0.0808 | 43.3 × 10-7 |
Rate Law for the Example
First order in each reactant; second order overall.
k is the rate constant, temperature-dependent.
Types of Reaction Orders
First-Order Reactions
First-order reactions depend on the concentration of one reactant to the first power.
Rate law:
Integrated rate law:
Plot of ln[A] vs. t is linear.
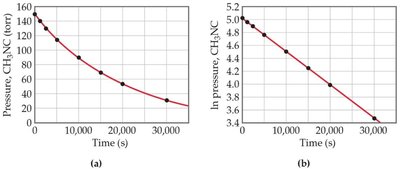
Half-Life of First-Order Reactions
The half-life () is the time required for the concentration of a reactant to decrease by half. For first-order reactions: 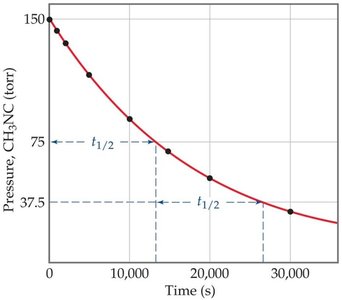
Second-Order Reactions
Second-order reactions depend on the concentration of one reactant squared or two reactants each to the first power.
Rate law:
Integrated rate law:
Plot of 1/[A] vs. t is linear.
![Plot of ln[NO2] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/5d9462b2_image_8.png)
![Plot of 1/[NO2] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/5d9462b2_image_9.png)
Half-Life of Second-Order Reactions
For second-order reactions, half-life depends on initial concentration:
Zero-Order Reactions
Zero-order reactions have rates independent of reactant concentration.
Rate law:
Integrated rate law:
Plot of [A] vs. t is linear.
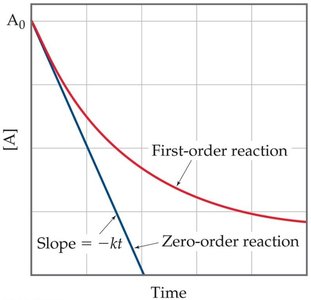
Collision Theory and Activation Energy
Factors Affecting Rate
Temperature: Higher temperature increases rate constant and reaction rate.
Frequency of collisions: More molecules, more collisions, faster rate.
Orientation of molecules: Proper alignment is necessary for effective collisions.
Activation energy (Ea): Minimum energy required for reaction to occur.

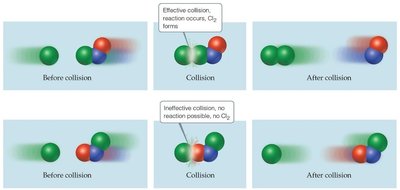
Transition State and Reaction Progress
The transition state (activated complex) is the highest energy point along the reaction pathway. The activation energy is the energy required to reach this state. 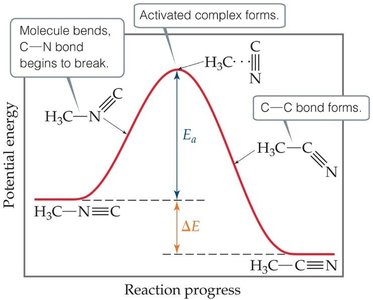
Energy Distribution and Temperature
At higher temperatures, more molecules have sufficient energy to overcome the activation energy barrier. 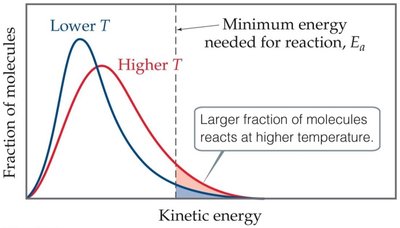
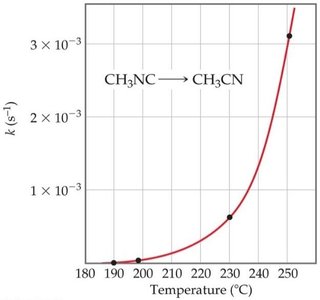
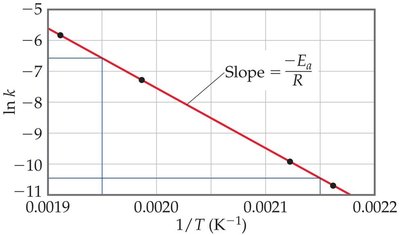
Arrhenius Equation
The Arrhenius equation relates the rate constant to temperature and activation energy:
Graphical form:
Reaction Mechanisms
Elementary Reactions and Molecularity
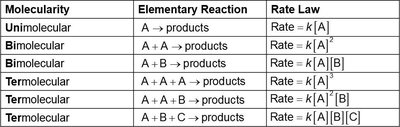
Mechanisms consist of elementary steps, each with a defined molecularity (number of molecules involved).
Molecularity | Elementary Reaction | Rate Law |
|---|---|---|
Unimolecular | A → products | Rate = k[A] |
Bimolecular | A + A → products | Rate = k[A]2 |
Bimolecular | A + B → products | Rate = k[A][B] |
Termolecular | A + A + A → products | Rate = k[A]3 |
Termolecular | A + A + B → products | Rate = k[A]2[B] |
Termolecular | A + B + C → products | Rate = k[A][B][C] |
Rate-Determining Step
The slowest step in a mechanism limits the overall reaction rate. 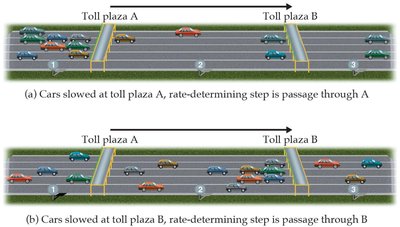
Intermediates
Intermediates are species formed in one step and consumed in another; they are not reactants or products and are often stable enough to be isolated. 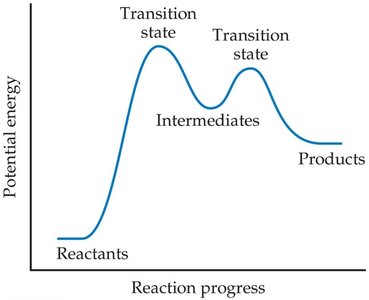
Catalysis
Catalysts and Their Effect
Catalysts increase reaction rates by lowering activation energy and changing the reaction mechanism. 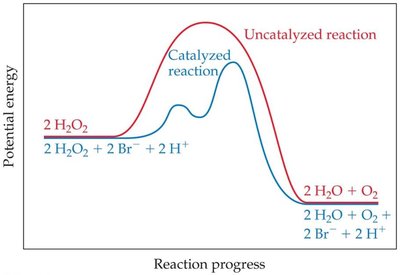
Types of Catalysts
Homogeneous catalysts: Same phase as reactants, often dissolved in solution.
Heterogeneous catalysts: Different phase, typically solid catalyst with gaseous or liquid reactants.
Enzymes: Biological catalysts with specific active sites for substrates.
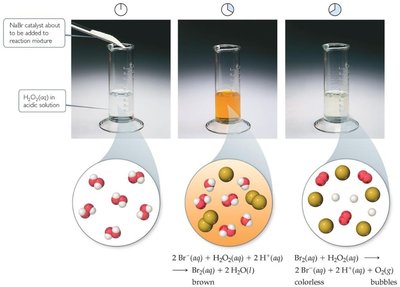
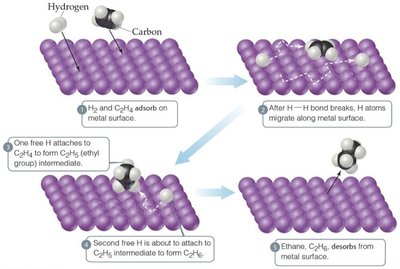

Enzyme Mechanisms
Enzymes operate via a lock-and-key model, where the substrate fits into the enzyme's active site. 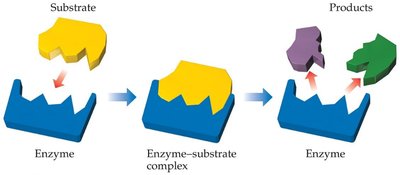
Summary Table: Key Equations
First-order integrated rate law:
Second-order integrated rate law:
Zero-order integrated rate law:
Arrhenius equation:
First-order half-life:
Second-order half-life:
Additional info:
All equations are presented in LaTeX format for clarity and academic rigor.
Images included are directly relevant to the explanation of each paragraph and reinforce key concepts.
