 Back
BackChapter 14: Chemical Kinetics – Study Guide
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the branch of chemistry that studies the rates of chemical reactions and the factors that affect them. Unlike thermodynamics, which tells us whether a reaction is product or reactant favored, kinetics focuses on how fast a reaction proceeds and the mechanism by which it occurs.
Reaction Rate: The speed at which reactants are converted to products, measured as the change in concentration of a reactant or product per unit time.
Kinetics vs. Thermodynamics: Thermodynamics determines if a reaction can occur; kinetics determines how quickly it happens.
Formula:

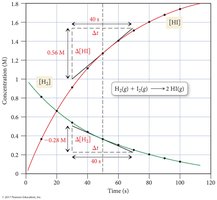
Types of Reaction Rates
Reaction rates can be described as average or instantaneous rates, depending on the time interval considered.
Average Rate: The change in concentration over a finite time interval; a linear approximation of the reaction curve.
Instantaneous Rate: The rate at a specific moment, determined by the slope of the tangent to the concentration vs. time curve.
As time progresses, reaction rates typically decrease due to the reduction in reactant concentration.
Rate Laws and Reaction Order
Rate Laws
The rate law expresses the relationship between the reaction rate and the concentrations of reactants. The form of the rate law must be determined experimentally.
General Rate Law:
Rate Constant (k): A proportionality constant specific to the reaction and temperature.
Reaction Order: The exponent of each reactant in the rate law; the sum of exponents gives the overall order.
Determining Reaction Order
Reaction order is found by analyzing how changes in reactant concentration affect the rate. Common orders are zero, first, and second.
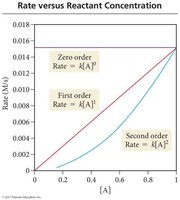
Zero Order: Rate is independent of reactant concentration ().
First Order: Rate is directly proportional to reactant concentration ().
Second Order: Rate is proportional to the square of reactant concentration ().
Experimental Determination of Rate Laws
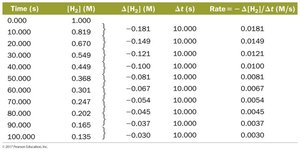
Rate laws are determined by measuring reaction rates at various concentrations and analyzing the data.
Doubling concentration and observing rate changes helps identify order.
Example: If doubling [A] quadruples the rate, the reaction is second order with respect to A.
Integrated Rate Laws
Integrated Rate Laws and Plots
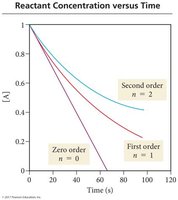
Integrated rate laws relate reactant concentration to time and allow determination of reaction order from concentration-time plots.
If [A] vs. time is linear, the reaction is zero order.
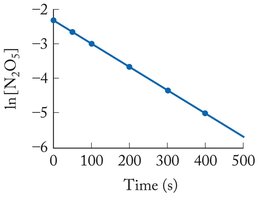
If ln [A] vs. time is linear, the reaction is first order.
If 1/[A] vs. time is linear, the reaction is second order.
![Zero order reaction: [A] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/996c844e_image_9.png)
![First order reaction: ln [A] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/996c844e_image_10.png)
![Second order reaction: 1/[A] vs. time](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/996c844e_image_12.png)
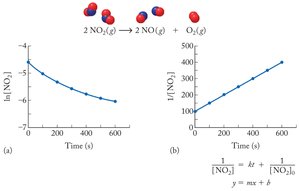
![Second order plot for NO2 decomposition (1/[NO2] vs. time)](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/996c844e_image_14.png)
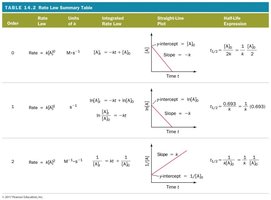
Summary Table of Rate Laws
The following table summarizes the rate laws, integrated rate laws, straight-line plots, and half-life expressions for zero, first, and second order reactions.
Order | Rate Law | Integrated Rate Law | Straight Line Plot | Half-Life Expression |
|---|---|---|---|---|
0 | Rate = k[A]^0 | [A]_t = -kt + [A]_0 | [A] vs. time | t_{1/2} = [A]_0 / 2k |
1 | Rate = k[A]^1 | ln [A]_t = -kt + ln [A]_0 | ln [A] vs. time | t_{1/2} = \ln(2)/k |
2 | Rate = k[A]^2 | 1/[A]_t = kt + 1/[A]_0 | 1/[A] vs. time | t_{1/2} = 1/(k[A]_0) |
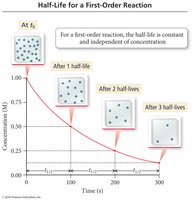
Half-Life of Reactions
Half-Life Concepts
The half-life (t1/2) is the time required for the concentration of a reactant to decrease to half its initial value. The expression for half-life depends on the reaction order.
Zero Order:
First Order:
Second Order:
Factors Affecting Reaction Rates
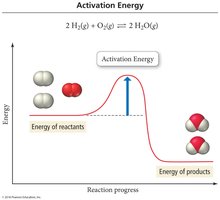
Activation Energy and Energy Diagrams
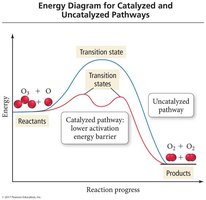
Activation energy (Ea) is the minimum energy required for a reaction to occur. Energy diagrams illustrate the energy changes during a reaction, including the formation of the activated complex (transition state).
Activation Energy: The energy barrier that must be overcome for reactants to convert to products.
Transition State: A high-energy, unstable state with partially formed and broken bonds.
Temperature and the Arrhenius Equation
The rate constant (k) is temperature dependent. The Arrhenius equation relates k to temperature, activation energy, and the frequency factor.
Arrhenius Equation:
Increasing temperature increases the fraction of molecules with enough energy to overcome the activation energy barrier, thus increasing the reaction rate.

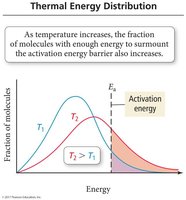
Collision Theory
Principles of Collision Theory
Collision theory states that molecules must collide with sufficient energy and proper orientation to react. Not all collisions result in a reaction.
Effective Collisions: Collisions with enough energy and correct orientation to form products.
Ineffective Collisions: Collisions lacking energy or proper orientation.


Reaction Mechanisms
Elementary Steps and Intermediates
A reaction mechanism is a series of elementary steps that describe how reactants are converted to products. Intermediates are species formed in one step and consumed in another.
Elementary Step: A single event in a mechanism; cannot be broken down further.
Intermediate: Produced in one step, consumed in another; does not appear in the overall equation.
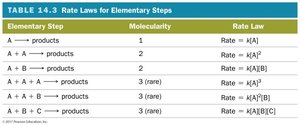
Molecularity: The number of reactant particles involved in an elementary step (unimolecular, bimolecular, termolecular).
Elementary Step | Molecularity | Rate Law |
|---|---|---|
A → products | 1 | Rate = k[A] |
A + A → products | 2 | Rate = k[A]^2 |
A + B → products | 2 | Rate = k[A][B] |
A + A + A → products | 3 (rare) | Rate = k[A]^3 |
A + B + C → products | 3 (rare) | Rate = k[A][B][C] |
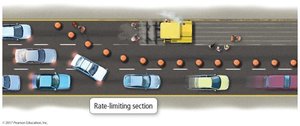
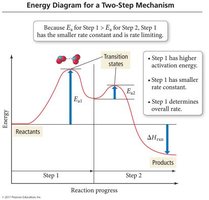
Rate-Determining Step
The slowest step in a reaction mechanism is the rate-determining step (RDS). The rate law for the overall reaction is governed by the RDS.
RDS: The step with the highest activation energy and lowest rate constant.
Mechanisms must sum to the overall reaction and predict the observed rate law.
Catalysis
Catalysts and Reaction Rates
A catalyst increases the reaction rate by providing an alternative pathway with lower activation energy. Catalysts are not consumed in the overall reaction.
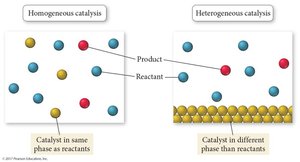
Homogeneous Catalyst: Same phase as reactants; forms a more stable activated complex.
Heterogeneous Catalyst: Different phase; holds reactants in proper orientation.
Summary
Chemical kinetics provides insight into the speed and mechanism of chemical reactions. Understanding rate laws, reaction order, activation energy, and catalysis is essential for predicting and controlling chemical processes.
