 Back
BackChapter 14: Chemical Kinetics – Study Notes
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the speed (rate) at which chemical reactions occur and the factors that influence these rates. It also investigates the mechanisms by which reactions proceed, providing insight into the stepwise changes that occur from reactants to products.
Reaction Rate: The change in concentration of a reactant or product per unit time.
Mechanism: The sequence of elementary steps by which a chemical reaction occurs.
Reaction Rates
Definition and Calculation of Reaction Rate
The rate of a reaction is defined as the change in concentration of a reactant or product over a specific period of time. For a general reaction:
Average Rate:
The negative sign for reactants indicates their concentration decreases over time.
Example: For the reaction , the rate can be expressed as:

Concentration vs. Time
Reaction rates typically decrease over time as reactant concentrations decrease. The rate can be visualized by plotting concentration versus time for reactants and products.
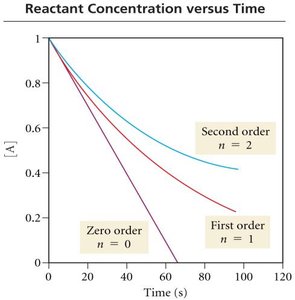
Factors Affecting Reaction Rate
Nature of Reactants
Small molecules react faster than large molecules.
Gases react faster than liquids, which react faster than solids.
Powdered solids are more reactive than blocks due to greater surface area.
Ions react faster than molecules.
Temperature
Increasing temperature increases reaction rate.
Rule of thumb: For each 10°C rise, the rate approximately doubles.
Catalysts
Catalysts increase reaction rate without being consumed.
They provide an alternative pathway with lower activation energy.
Concentration
Higher concentration of reactants generally increases reaction rate due to more frequent collisions.
Rate Laws and Reaction Order
Rate Law Expression
The rate law relates the rate of a reaction to the concentration of reactants, each raised to a power (the order with respect to that reactant):
General form:
k is the rate constant; x and y are the orders with respect to each reactant.
The overall order is the sum of the exponents.
Determining Rate Laws
Rate laws must be determined experimentally, often using the method of initial rates.
By varying the concentration of one reactant at a time, the order with respect to each can be found.
Integrated Rate Laws and Reaction Order
The integrated rate law allows calculation of reactant concentration at any time. The form depends on the reaction order:
Zero Order:
First Order:
Second Order:
The half-life () is the time required for the concentration to decrease to half its initial value. For first-order reactions, half-life is constant and independent of concentration:
(first order)
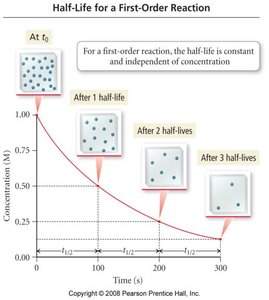
Temperature and the Rate Constant: The Arrhenius Equation
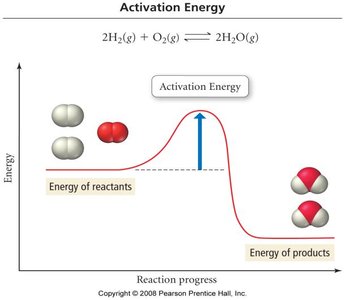
Activation Energy and the Arrhenius Equation
The rate constant depends on temperature, described by the Arrhenius equation:
is the activation energy, is the frequency factor, is the gas constant, and is temperature in Kelvin.
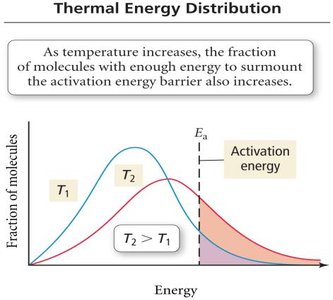
Thermal Energy Distribution
As temperature increases, a greater fraction of molecules have enough energy to overcome the activation energy barrier, increasing the reaction rate.
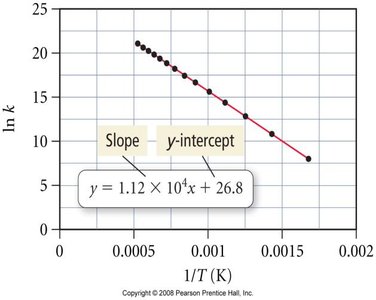
Arrhenius Plot
Taking the natural logarithm of the Arrhenius equation gives a linear form:
A plot of versus yields a straight line with slope and intercept .
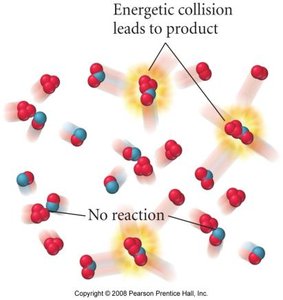
Collision Theory
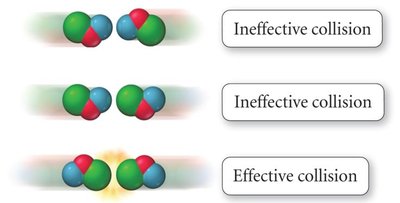
Effective Collisions
For a reaction to occur, reactant molecules must collide with sufficient energy and proper orientation. Only a fraction of collisions are effective and lead to product formation.
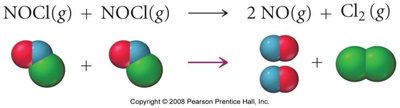
Reaction Mechanisms
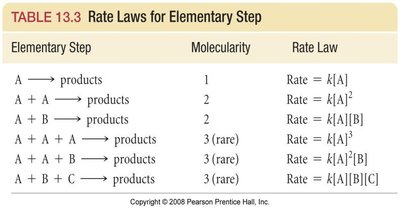
Elementary Steps and Molecularity
A reaction mechanism consists of a sequence of elementary steps. Each step can be classified by its molecularity (number of molecules involved):
Unimolecular: Involves one molecule.
Bimolecular: Involves two molecules.
Termolecular: Involves three molecules (rare).
Elementary Step | Molecularity | Rate Law |
|---|---|---|
A → products | 1 | Rate = k[A] |
A + A → products | 2 | Rate = k[A]^2 |
A + B → products | 2 | Rate = k[A][B] |
A + A + A → products | 3 (rare) | Rate = k[A]^3 |
A + A + B → products | 3 (rare) | Rate = k[A]^2[B] |
A + B + C → products | 3 (rare) | Rate = k[A][B][C] |
Rate-Determining Step
In a multi-step mechanism, the slowest step determines the overall reaction rate (rate-determining step). The rate law for the overall reaction is based on this step.
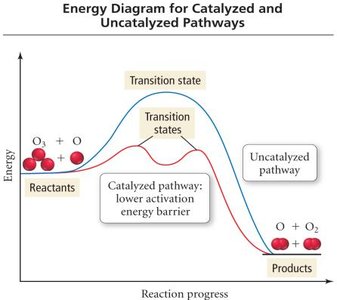
Catalysis
Effect of Catalysts
Catalysts increase the rate of a reaction by providing an alternative pathway with a lower activation energy. They are not consumed in the reaction and can be homogeneous (same phase as reactants) or heterogeneous (different phase).
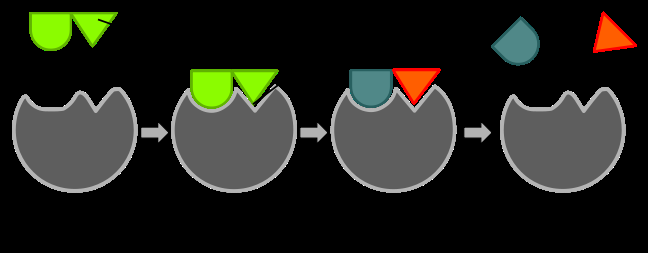
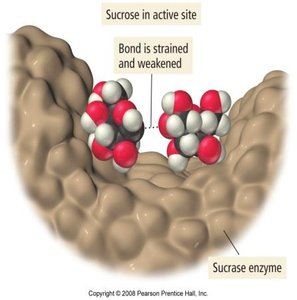
Enzymes
Enzymes are biological catalysts, usually proteins, that speed up biochemical reactions. They work by binding reactants (substrates) at an active site, orienting them for reaction and lowering the activation energy.
Additional info: The study of kinetics is essential for understanding how chemical reactions can be controlled and optimized in industrial, laboratory, and biological settings. Mastery of these concepts is foundational for further study in chemistry and related sciences.
