 Back
BackChapter 14: Solutions – Structure, Energetics, and Solubility
Study Guide - Smart Notes

Solutions
Solution Terminology
Understanding solutions is fundamental in chemistry, as they represent homogeneous mixtures of two or more substances. The terminology associated with solutions helps classify and describe their properties and behaviors.
Solution: A homogeneous mixture of two or more substances.
Solvent: The component present in the greater amount; typically determines the phase of the solution.
Solute: The component present in the lesser amount; dissolved in the solvent.
Example: In an aqueous solution of salt water, water is the solvent and salt is the solute.
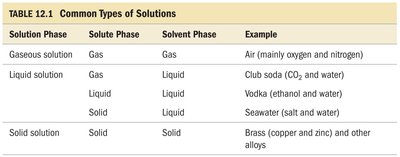
Table Purpose: Classification of solution types based on the phases of solute and solvent, with examples.
Solution Phase | Solute Phase | Solvent Phase | Example |
|---|---|---|---|
Gaseous solution | Gas | Gas | Air (mainly oxygen and nitrogen) |
Liquid solution | Gas | Liquid | Club soda (CO2 and water) |
Liquid solution | Liquid | Liquid | Vodka (ethanol and water) |
Liquid solution | Solid | Liquid | Seawater (salt and water) |
Solid solution | Solid | Solid | Brass (copper and zinc) and other alloys |
Solubility
Solubility quantifies the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature. It is commonly expressed as grams of solute per 100 mL or 100 g of solvent.
Miscible: Liquids that are soluble in each other in all proportions.
Immiscible: Liquids that are not soluble in each other.
Saturated, Unsaturated, and Supersaturated Solutions
Solutions can be classified based on the amount of solute dissolved relative to the equilibrium solubility at a given temperature.
Saturated Solution: Contains the maximum amount of dissolved solute at equilibrium; excess solute remains undissolved.
Unsaturated Solution: Contains less than the equilibrium amount of solute.
Supersaturated Solution: Contains more solute than is normally possible; unstable and prone to crystallization.

Example: If 110 g CuCl2 are added to 100 mL water at 0°C (solubility = 70.6 g/100 mL), only 70.6 g dissolve (saturated), and the rest remains undissolved.
Energetics of Making Solutions
Why Do Molecular Solutions Form?
The formation of a solution depends on the types of intermolecular forces (IMFs) involved and the entropy change during mixing.
IMFs: The strength and type of interactions (solute-solute, solvent-solvent, solute-solvent) determine the enthalpy change.
Entropy: Mixing increases randomness (entropy), favoring solution formation even if the process is endothermic.
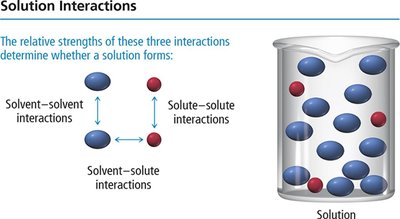
Conditions That Favor Solution Formation
Solution formation is favored when solute-solvent interactions are stronger than solute-solute and solvent-solvent interactions.
Weak solute-solute interactions
Weak solvent-solvent interactions
Strong solute-solvent interactions
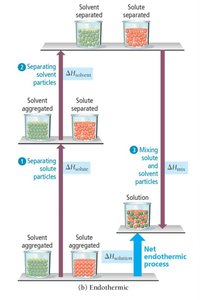
Energy Changes During Solution Formation
The process of solution formation involves three energetically distinct steps:
Breaking solute-solute interactions (, endothermic)
Breaking solvent-solvent interactions (, endothermic)
Mixing solute and solvent particles (, exothermic)
The overall enthalpy change is:
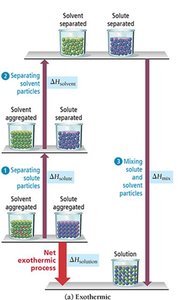
Exothermic: If the energy released in mixing is greater than the energy required to separate solute and solvent, the process is exothermic.
Endothermic: If the energy required to separate solute and solvent is greater than the energy released in mixing, the process is endothermic.
Application of Solution Concepts
Several real-world examples illustrate the principles of solution formation:
Oil and Water: Immiscible due to weak solute-solvent interactions and strong solvent-solvent interactions (hydrogen bonding).
Carbon Tetrachloride and Benzene: Both nonpolar; similar IMFs allow miscibility.
Heptane and Octane: Both nonpolar hydrocarbons; miscible due to similar London Dispersion Forces.
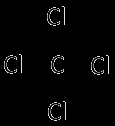
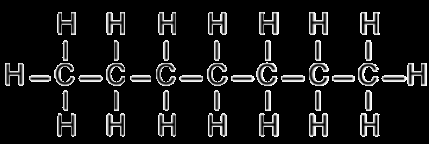
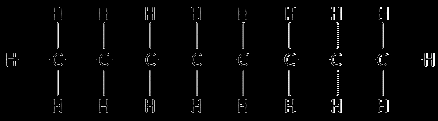
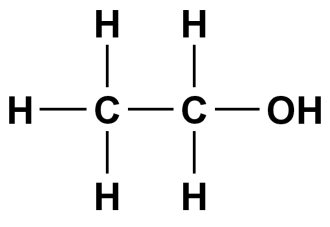
Methanol, Ethanol, and 1,2-Ethylene Glycol: Miscible in water due to hydrogen bonding.
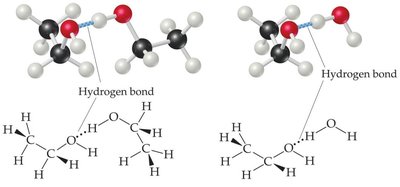
Vitamin C: Polar, water-soluble.
Vitamin A: Nonpolar, fat-soluble.
Key Principle: "Like dissolves like" – polar or ionic solutes dissolve in polar solvents; nonpolar solutes dissolve in nonpolar solvents.
Trends in Alcohol Solubility in Water
As the hydrocarbon chain in alcohols increases, the molecule becomes less polar and its solubility in water decreases.
Alcohol | Formula | Solubility (g/100 g H2O) |
|---|---|---|
Methanol | CH3OH | Miscible |
Ethanol | CH3CH2OH | Miscible |
1-Propanol | CH3CH2CH2OH | Miscible |
1-Butanol | CH3CH2CH2CH2OH | 7.9 |
1-Pentanol | CH3CH2CH2CH2CH2OH | 2.7 |
1-Hexanol | CH3CH2CH2CH2CH2CH2OH | 0.6 |
Ionic Solutions and Enthalpy of Hydration
Ionic Solutions
When ionic compounds dissolve in water, ions are stabilized by hydration due to ion-dipole attractions. Each ion is surrounded by several water molecules.
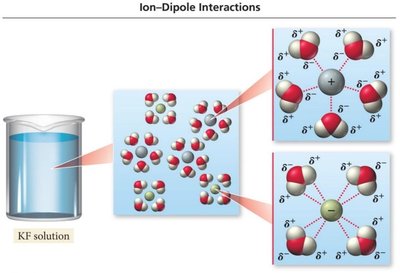
Enthalpy of Hydration
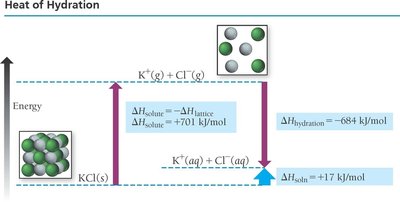
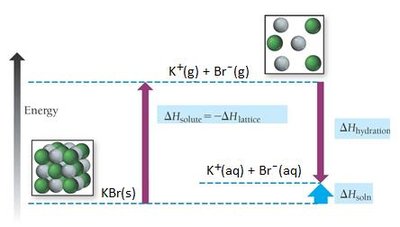
The enthalpy of solution for ionic compounds is given by:
Lattice Energy (): The energy released when one mole of an ionic solid forms from its gaseous ions; highly exothermic.
Enthalpy of Hydration (): The energy released when one mole of gaseous ions is dissolved in water; always exothermic.
The higher the charge density (charge/volume) of the ion, the more exothermic the enthalpy of hydration.
For ionic compounds, the overall solubility depends on the balance between lattice energy and hydration energy.
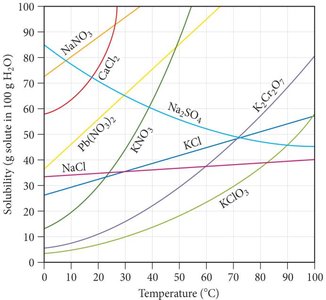
Effect of Temperature and Pressure on Solubility
Temperature and pressure significantly affect solubility:
Solids: Solubility generally increases with temperature, but there are exceptions (e.g., Na2SO4).
Gases: Solubility decreases with increasing temperature (thermal pollution).
Pressure: Solubility of gases increases with partial pressure, described by Henry's Law:
Where is the solubility of the gas, is Henry's Law constant, and is the partial pressure.

Summary Table: Key Concepts in Solution Formation
Concept | Definition | Example/Application |
|---|---|---|
Solution | Homogeneous mixture | Salt water |
Solubility | Maximum solute dissolved | 70.6 g CuCl2/100 mL H2O at 0°C |
Enthalpy of Solution | Energy change in solution formation | |
Lattice Energy | Energy released in ionic solid formation | NaCl(s) from Na+(g) and Cl-(g) |
Hydration Energy | Energy released when ions dissolve | Ca2+(g) + H2O(l) → Ca2+(aq) |
Henry's Law | Gas solubility vs. pressure | CO2 in soda |
Additional info: Academic context and examples were expanded for clarity and completeness. All images included are directly relevant to the adjacent explanations, visually reinforcing key concepts in solution chemistry.
