 Back
BackChapter 14: Solutions – Structure, Properties, and Calculations
Study Guide - Smart Notes

Solutions: Composition and Types
Definition and Components
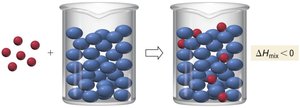
Solutions are homogeneous mixtures composed of two or more substances. The majority component is called the solvent, while the minority component is the solute. Solutions can form spontaneously unless energetically unfavorable. 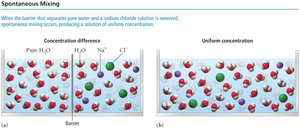
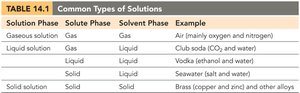
Common Types of Solutions
Solutions can exist in various phases depending on the physical states of the solute and solvent.
Solution Phase | Solute Phase | Solvent Phase | Example |
|---|---|---|---|
Gaseous solution | Gas | Gas | Air (mainly oxygen and nitrogen) |
Liquid solution | Gas | Liquid | Club soda (CO2 and water) |
Liquid solution | Liquid | Liquid | Vodka (ethanol and water) |
Solid solution | Solid | Liquid | Seawater (salt and water) |
Solid solution | Solid | Solid | Brass (copper and zinc) and other alloys |
Spontaneous Mixing and Entropy
Nature’s Tendency to Mix
Mixing of substances is driven by entropy, which is a measure of energy dispersal in a system. Spontaneous mixing occurs when the barrier between two substances is removed, resulting in a uniform concentration. 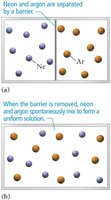
Role of Entropy
- Entropy increases as energy is dispersed over a larger volume. - Formation of a solution does not always lower the system's potential energy, but the increase in entropy can drive the process.
Intermolecular Forces and Solution Formation
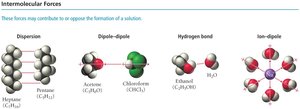
Types of Intermolecular Forces
Intermolecular forces determine the ability of substances to mix and form solutions. The main types are:
Dispersion forces (London forces): Present in all molecules, especially nonpolar ones.
Dipole-dipole interactions: Occur between polar molecules.
Hydrogen bonding: Strong dipole-dipole interaction involving H bonded to N, O, or F.
Ion-dipole interactions: Occur between ions and polar molecules.
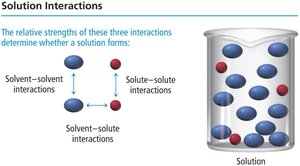
Solution Interactions
Solution formation depends on the relative strengths of three interactions:
Solvent-solvent interactions
Solute-solute interactions
Solvent-solute interactions
Relative Interaction | Outcome |
|---|---|
Solvent-solute > solvent-solvent and solute-solute | Solution forms |
Solvent-solute = solvent-solvent and solute-solute | Solution forms |
Solvent-solute < solvent-solvent and solute-solute | Solution may or may not form, depending on disparity |

Solubility: "Like Dissolves Like"
Solubility is the maximum amount of solute that can dissolve in a given amount of solvent. The general rule is "like dissolves like":
Polar molecules and ionic compounds dissolve in polar solvents.
Nonpolar molecules are more soluble in nonpolar solvents.
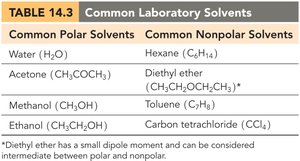
Common Polar Solvents | Common Nonpolar Solvents |
|---|---|
Water (H2O) | Hexane (C6H14) |
Acetone (CH3COCH3) | Diethyl ether (CH3CH2OCH2CH3) |
Methanol (CH3OH) | Toluene (C7H8) |
Ethanol (CH3CH2OH) | Carbon tetrachloride (CCl4) |
Solubility of Vitamins
Vitamins can be classified as water-soluble or fat-soluble based on their molecular structure. Water-soluble vitamins dissolve in body fluids and are easily eliminated, while fat-soluble vitamins can accumulate in fatty tissues. 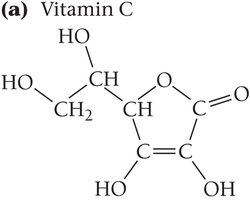
Energetics of Solution Formation
Three-Step Process
Formation of a solution can be visualized in three steps:
Separating solute particles (requires energy, endothermic)
Separating solvent particles (requires energy, endothermic)
Mixing solute and solvent particles (releases energy, exothermic)

Enthalpy of Solution Formation
The overall enthalpy change for solution formation is: For aqueous solutions, and are combined into : 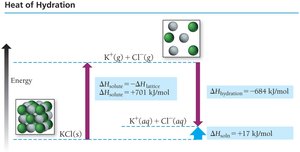
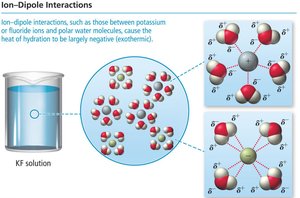
Examples of Enthalpy Changes
Exothermic solution formation:
Endothermic solution formation:
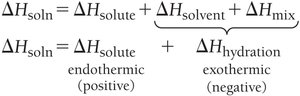
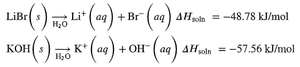
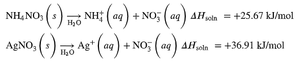
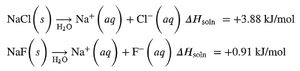
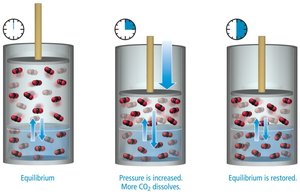
Solution Equilibrium and Solubility Limits
Dynamic Equilibrium
When a solution reaches its solubility limit, the rate of dissolution equals the rate of recrystallization, resulting in dynamic equilibrium. 


Types of Solutions Based on Solubility
Saturated: Contains maximum solute at equilibrium; additional solute will not dissolve.
Unsaturated: Contains less solute than saturation; more solute can dissolve.
Supersaturated: Contains more solute than saturation; unstable and excess solute may precipitate.

Solubility and Temperature
Solubility is usually reported as grams of solute per 100 g water. For most solids, solubility increases with temperature (endothermic dissolution). 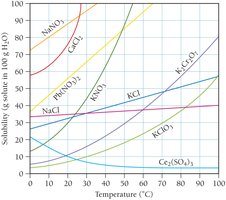
Solubility of Gases
Gases generally have lower solubility in water, and their solubility decreases with increasing temperature (exothermic dissolution). 
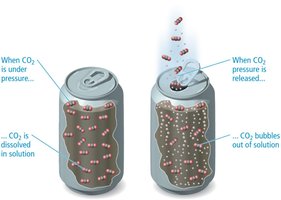
Henry’s Law
The solubility of a gas in a liquid is proportional to the partial pressure of the gas above the liquid: Where is the solubility, is Henry’s law constant, and is the partial pressure. 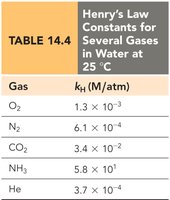
Concentration Units and Calculations
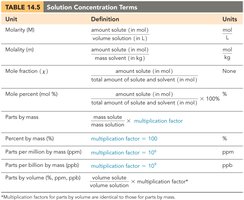
Describing Solution Concentration
Concentration expresses the amount of solute in a given amount of solution. Common units include:
Molarity (M): mol solute / L solution
Molality (m): mol solute / kg solvent
Mass percent: (mass solute / mass solution) × 100%
Parts per million (ppm): (mass solute / mass solution) × 106
Mole fraction (XA): mol component / total mols
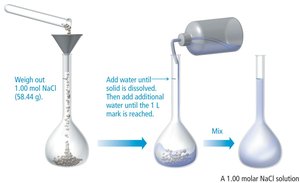
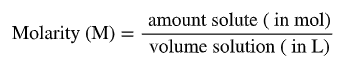
Key Formulas
Molarity:
Molality:
Percent by mass:
Parts per million:
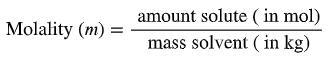
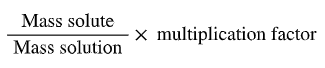
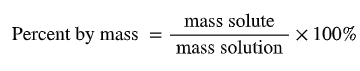
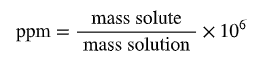
Colligative Properties
Definition and Types
Colligative properties depend on the number of solute particles, not their identity. These include:
Vapor pressure lowering
Freezing point depression
Boiling point elevation
Osmotic pressure
Vapor Pressure Lowering
The presence of a nonvolatile solute lowers the vapor pressure of the solvent.
Freezing Point Depression and Boiling Point Elevation
Adding solute to a solvent lowers its freezing point and raises its boiling point.
Osmosis and Osmotic Pressure
Osmosis is the movement of solvent through a semipermeable membrane from a region of lower solute concentration to higher concentration. Osmotic pressure is the pressure required to stop this flow.
van’t Hoff Factor (i)
The van’t Hoff factor accounts for the number of particles a solute produces in solution, affecting colligative properties.
Mixtures: Solutions, Suspensions, and Colloids
Classification of Mixtures
Solutions: Homogeneous mixtures; particles do not separate on standing.
Suspensions: Heterogeneous; particles separate on standing.
Colloids: Heterogeneous; particles do not separate on standing, but can coagulate and exhibit Brownian motion.
Soap and Emulsification
Soaps have hydrophilic heads and hydrophobic tails, allowing them to form micelles and emulsify nonpolar substances in water.
Tyndall Effect
Colloids scatter light, a phenomenon known as the Tyndall effect.
Summary Table: Key Solution Concepts
Concept | Definition | Example/Application |
|---|---|---|
Solution | Homogeneous mixture of solute and solvent | Salt water, air |
Solubility | Maximum amount of solute that can dissolve | NaCl in water |
Colligative properties | Depend on number of solute particles | Boiling point elevation |
Concentration units | Molarity, molality, mass percent, ppm | Lab calculations |
Additional info: Academic context and explanations have been expanded for clarity and completeness.
