 Back
BackChapter 14: Solutions – Structure, Properties, and Colligative Effects
Study Guide - Smart Notes

Solutions
Definition and Components
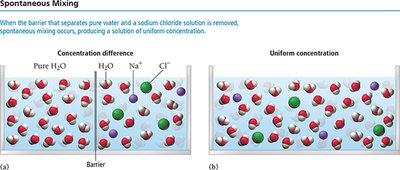
Solutions are homogeneous mixtures in which atoms and molecules intermingle on the molecular and atomic scale. The solvent is the majority component, while the solute is the minority component. Solutions form due to intermolecular forces between solute and solvent particles.
Solvent: The substance present in the largest amount.
Solute: The substance present in a smaller amount.
Homogeneous mixture: Appears as a single substance but contains multiple materials.
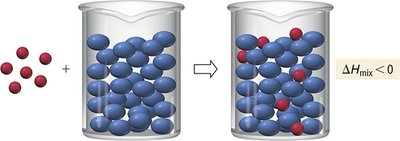
Nature’s tendency: Spontaneous mixing is energetically favorable.
Types of Solutions
Solutions can be formed from various combinations of phases. In aqueous solutions, water is the solvent.
Solution Phase | Solute Phase | Solvent Phase | Example |
|---|---|---|---|
Gas | Gas | Gas | Air (O2 and N2) |
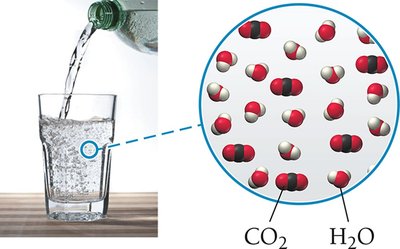
Liquid | Gas | Liquid | Club soda (CO2 in water) |
Liquid | Liquid | Liquid | Vodka (ethanol and water) |
Liquid | Solid | Liquid | Seawater (salt and water) |
Solid | Solid | Solid | Brass (copper and zinc) |
Solubility
Solubility and Miscibility
Solubility refers to the ability of a solute to dissolve in a solvent. If a substance dissolves, it is soluble; if not, it is insoluble. Two liquids that are mutually soluble are miscible; if not, they are immiscible. Solubility depends on the tendency toward mixing and the types of intermolecular forces.
Solubility limit: Maximum amount of solute that can dissolve in a given solvent.
Temperature and pressure: Solubility varies with these conditions.
Rule of thumb: Like dissolves like (polar solutes dissolve in polar solvents, nonpolar in nonpolar).
Common Polar Solvents | Common Nonpolar Solvents |
|---|---|
Water (H2O) | Hexane (C6H14) |
Acetone (CH3COCH3) | Diethyl ether (CH3CH2OCH2CH3) |
Methanol (CH3OH) | Toluene (C7H8) |
Ethanol (CH3CH2OH) | Carbon tetrachloride (CCl4) |

Intermolecular Forces and Solution Formation
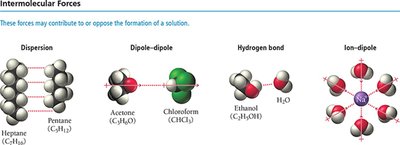
The formation of solutions depends on the types and strengths of intermolecular forces present. These include dispersion, dipole-dipole, hydrogen bonding, and ion-dipole interactions.
Dispersion: Present in all molecules, especially nonpolar.
Dipole-dipole: Occurs between polar molecules.
Hydrogen bond: Strong dipole-dipole interaction involving H bonded to N, O, or F.
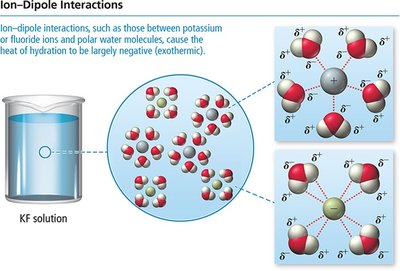
Ion-dipole: Occurs between ions and polar molecules (e.g., Na+ and H2O).
Energetics of Solution Formation
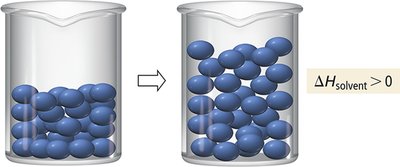
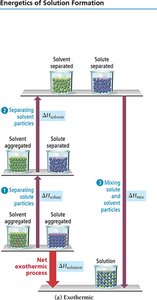
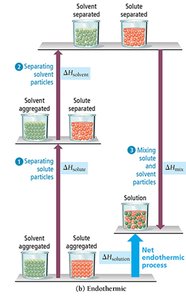
Steps in Solution Formation
Three main steps occur when a solution forms:
Separating solute particles (; endothermic)
Separating solvent particles (; endothermic)
Mixing solute and solvent particles (; exothermic)
The overall enthalpy change is:

Exothermic vs. Endothermic Solution Formation
If the energy released in mixing is greater than the energy required to separate solute and solvent, the process is exothermic. If less, it is endothermic.
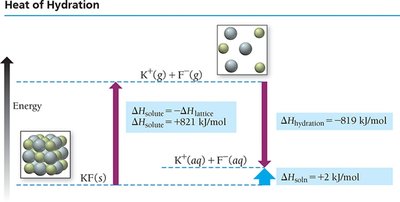
Heats of Hydration (Aqueous Solutions)
For ionic compounds in water, the heat of hydration combines the energy to overcome water-water attractions and the energy released in forming ion-water attractions.
Lattice energy: Energy to separate ions in a solid.
Heat of hydration: Energy released when ions dissolve in water.
Ion-dipole interactions: Strongly exothermic, leading to high solubility of ionic compounds in water.
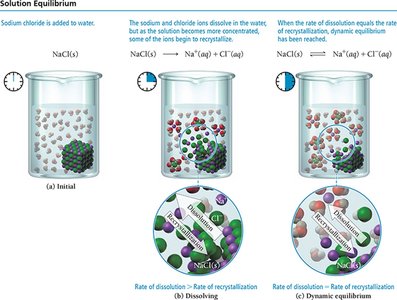
Solution Equilibrium and Solubility Limit
Dynamic Equilibrium
Dissolution is an equilibrium process. When the rate of dissolution equals the rate of deposition, the solution is saturated.
Saturated: Maximum solute dissolved; no more will dissolve.
Unsaturated: Less than maximum solute; more can dissolve.
Supersaturated: More than maximum solute; unstable and will precipitate excess solute.
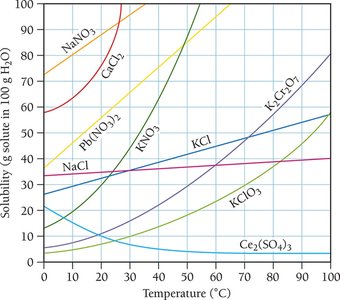
Temperature and Pressure Effects on Solubility
Solubility of Solids
For most solids, solubility increases with temperature. Solubility curves show the relationship between solubility and temperature.
Solubility of Gases
Gas solubility decreases with increasing temperature and increases with increasing pressure. This is important for carbonated beverages and aquatic life.

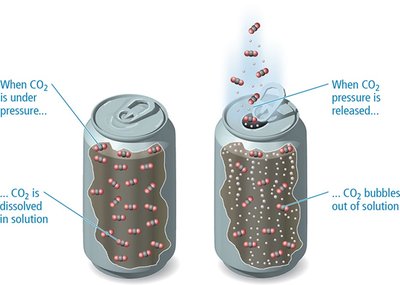
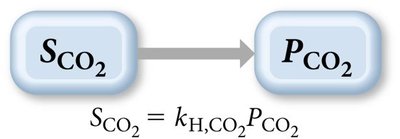
Henry’s Law
The solubility of a gas is directly proportional to its partial pressure:
kH: Henry’s law constant (varies by gas and solvent)
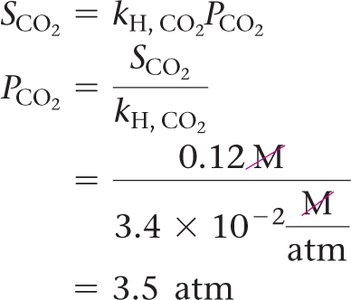
Solution Concentration Units
Molarity (M)
Molarity is the number of moles of solute per liter of solution:
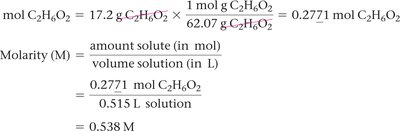
Molality (m)
Molality is the number of moles of solute per kilogram of solvent:
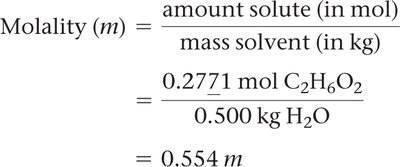
Percent by Mass
Percent by mass is the mass of solute divided by the mass of solution, multiplied by 100%:
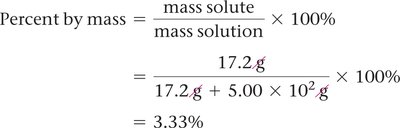
Mole Fraction and Mole Percent
Mole fraction is the fraction of moles of one component in the total moles of all components:
Mole percent is mole fraction multiplied by 100%.
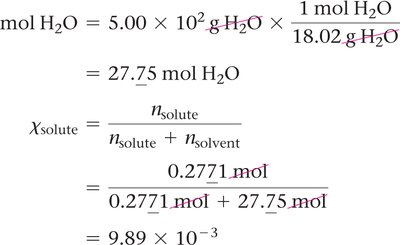
Colligative Properties
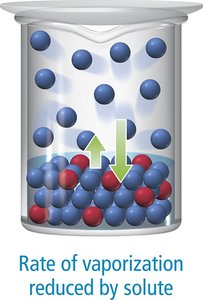
Vapor Pressure Lowering
The vapor pressure of a solvent above a solution is lower than that of the pure solvent. This is due to solute particles occupying surface positions and reducing the rate of vaporization.


Raoult’s Law
Raoult’s Law describes the vapor pressure of a volatile solvent above a solution:
Vapor pressure lowering:
Boiling Point Elevation and Freezing Point Depression
Adding a solute raises the boiling point and lowers the freezing point of a solvent. The changes are proportional to the molality of the solute:
Freezing point depression:
Boiling point elevation:
Osmosis and Osmotic Pressure
Osmosis
Osmosis is the flow of solvent from a solution of low concentration to one of high concentration through a semipermeable membrane. The pressure required to stop this flow is the osmotic pressure:
M: Molarity of solute particles
R: Gas constant ()
T: Temperature in Kelvin
Van’t Hoff Factor and Electrolyte Solutions
Van’t Hoff Factor (i)
The van’t Hoff factor is the ratio of moles of solute particles to moles of formula units dissolved. For electrolytes, is greater than 1 due to dissociation.
Solute | i Expected | i Measured |
|---|---|---|
Nonelectrolyte | 1 | 1 |
NaCl | 2 | 1.9 |
MgSO4 | 2 | 1.3 |
MgCl2 | 3 | 2.7 |
K2SO4 | 3 | 2.6 |
FeCl3 | 4 | 3.4 |
Colloids and Suspensions
Colloid Mixtures
Colloids are heterogeneous mixtures that do not separate on standing. They exhibit the Tyndall effect (scattering of light) and Brownian motion (random movement of particles).
Hydrophilic: Stabilized by attraction to solvent.
Hydrophobic: Stabilized by charged surface repulsions.
