 Back
BackChapter 14: Solutions – Structure, Properties, and Formation
Study Guide - Smart Notes

Solutions: Homogeneous Mixtures
Definition and Components
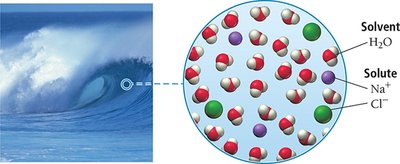
Solutions are homogeneous mixtures composed of a solvent (majority component) and one or more solutes (minority component). The solute is uniformly dispersed throughout the solvent, resulting in a mixture that appears as a single substance, though it contains multiple materials. Most homogeneous materials encountered in daily life, such as air and seawater, are actually solutions.
Solvent: The substance present in the greatest amount.
Solute: The substance(s) present in lesser amounts.
Homogeneous: Uniform composition throughout.
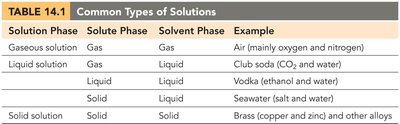
Examples: Air (gaseous solution), seawater (liquid solution), brass (solid solution).
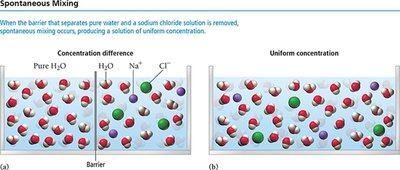
Spontaneous Mixing and Entropy
Nature tends toward spontaneous mixing, which increases the disorder (entropy) of the system. When solutions with different solute concentrations come into contact, they mix spontaneously to form a uniform distribution of solute, driven by entropy rather than a decrease in potential energy.
Mixing is energetically favorable due to increased entropy.
Spontaneous mixing occurs even when attractive forces are negligible, as with ideal gases.
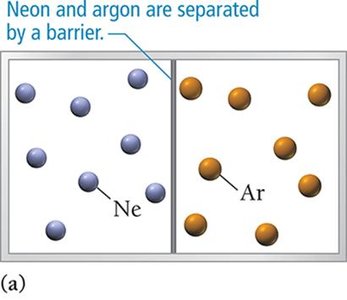
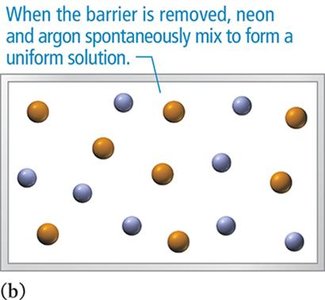
Types of Solutions
Solutions can exist in various phases depending on the states of the solute and solvent. The properties of mixtures depend on the relative amounts of each component.
Solution Phase | Solute Phase | Solvent Phase | Example |
|---|---|---|---|
Gas | Gas | Gas | Air (mainly O2 and N2) |
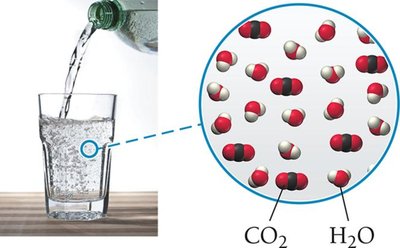
Liquid | Gas | Liquid | Club soda (CO2 and water) |
Liquid | Liquid | Liquid | Vodka (ethanol and water) |
Solid | Liquid | Solid | Seawater (salt and water) |
Solid | Solid | Solid | Brass (copper and zinc) |
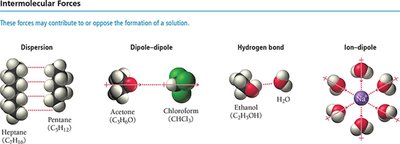
Intermolecular Forces and Solution Formation
Types of Intermolecular Forces
The formation of solutions is influenced by the types and strengths of intermolecular forces between solute and solvent particles. These include:
Dispersion forces: Present in all molecules, especially nonpolar substances.
Dipole-dipole forces: Occur between polar molecules.
Hydrogen bonds: Strong dipole-dipole interactions involving H bonded to N, O, or F.
Ion-dipole forces: Occur between ions and polar molecules, crucial in dissolving ionic compounds in water.
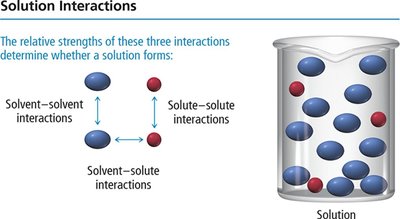
Solution Interactions
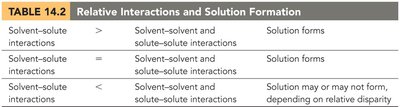
For a solution to form, the relative strengths of solvent-solvent, solute-solute, and solvent-solute interactions must be considered. The process involves overcoming solute-solute and solvent-solvent attractions (endothermic), and forming new solute-solvent attractions (exothermic).
If solvent-solute interactions are stronger than the others, solution forms easily.
If interactions are equal, solution still forms.
If solvent-solute interactions are weaker, solution may not form.
Interaction Comparison | Result |
|---|---|
Solvent-solute > Solvent-solvent and solute-solute | Solution forms |
Solvent-solute = Solvent-solvent and solute-solute | Solution forms |
Solvent-solute < Solvent-solvent and solute-solute | Solution may or may not form |
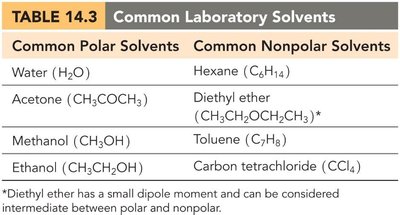
Miscibility and Solubility
"Like dissolves like" is a guiding principle: polar substances dissolve in polar solvents, and nonpolar substances dissolve in nonpolar solvents. Miscible liquids mix in all proportions, while immiscible liquids do not mix.
Miscible: Alcohol and water
Immiscible: Oil and water

Common Polar Solvents | Common Nonpolar Solvents |
|---|---|
Water (H2O) | Hexane (C6H14) |
Acetone (CH3COCH3) | Diethyl ether (CH3CH2OCH2CH3) |
Methanol (CH3OH) | Toluene (C7H8) |
Ethanol (CH3CH2OH) | Carbon tetrachloride (CCl4) |
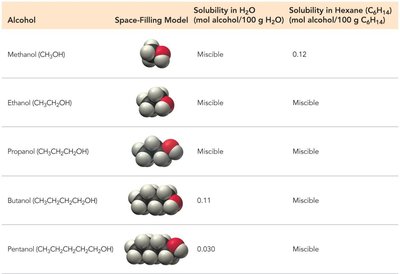
Solubility of Alcohols
The solubility of alcohols in water and hexane depends on their molecular structure. Short-chain alcohols are miscible in water, while longer chains become less soluble.
Alcohol | Solubility in H2O | Solubility in Hexane |
|---|---|---|
Methanol | Miscible | 0.12 |
Ethanol | Miscible | Miscible |
Propanol | Miscible | Miscible |
Butanol | 0.11 | Miscible |
Pentanol | 0.030 | Miscible |
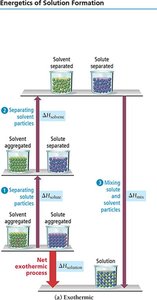
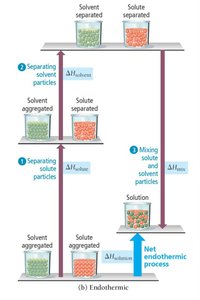
Energetics of Solution Formation
Steps and Enthalpy Changes
Formation of a solution involves three steps:
Separating solute particles (, endothermic)
Separating solvent particles (, endothermic)
Mixing solute and solvent particles (, exothermic)
The overall enthalpy change is:

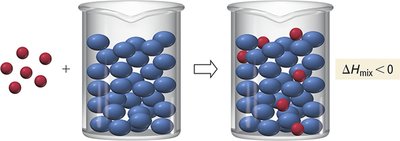
Exothermic vs. Endothermic Solution Formation
If the energy released in forming solute-solvent attractions exceeds the energy required to break solute-solute and solvent-solvent attractions, the process is exothermic. If not, it is endothermic.
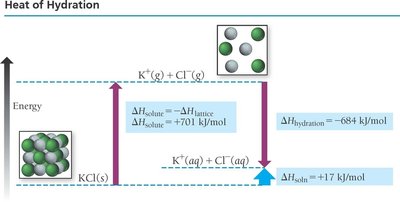
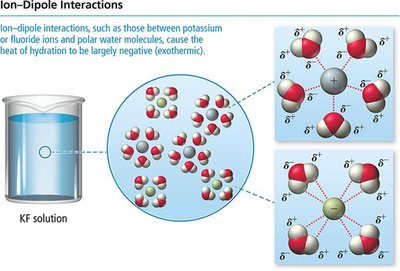
Heat of Hydration and Ionic Solutions
For aqueous solutions of ionic compounds, the energy changes are described by the heat of hydration. The lattice energy (attractive forces between ions) must be overcome, and new ion-dipole attractions with water are formed.
Heat of hydration is usually very exothermic due to strong ion-dipole interactions.
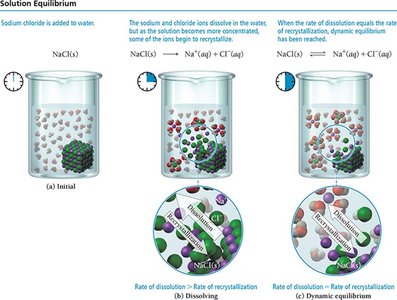
Solution Equilibrium and Solubility
Saturated, Unsaturated, and Supersaturated Solutions
The dissolution of a solute in a solvent is an equilibrium process. A solution is saturated when the rate of dissolution equals the rate of deposition. If less solute is present, the solution is unsaturated. If more solute is present than the solubility limit, the solution is supersaturated.
Saturated: No more solute dissolves.
Unsaturated: More solute can dissolve.
Supersaturated: Excess solute precipitates out.

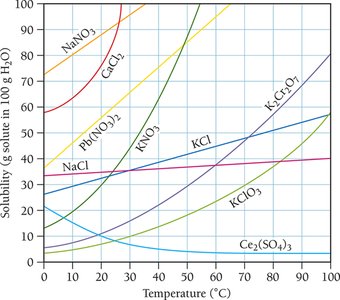
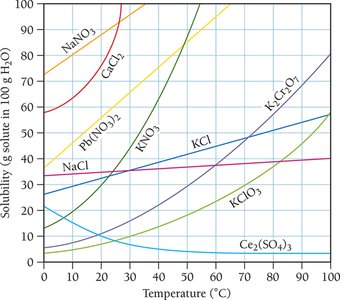
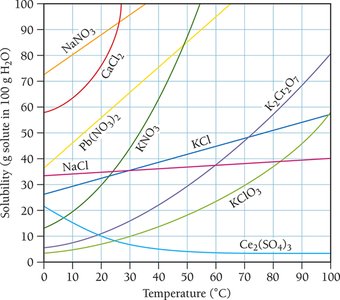
Solubility Curves and Temperature Dependence
Solubility curves show how the solubility of various solutes changes with temperature. Generally, the solubility of solid solutes in liquid solvents increases with temperature, while the solubility of gases decreases.
Purification by Recrystallization
Recrystallization is a method used to purify solid compounds. The solid is dissolved in a hot solvent until saturated, then allowed to cool so the pure compound crystallizes out, leaving impurities behind.
Solubility of Gases: Temperature and Pressure Effects
Temperature Dependence
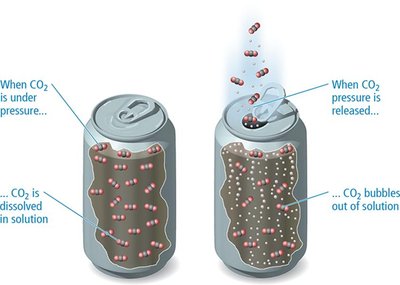
Gases generally have lower solubility in water than ionic or polar solids. The solubility of gases decreases as temperature increases, because the dissolution process is exothermic.

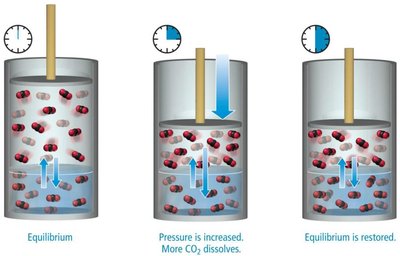
Pressure Dependence and Henry's Law
The solubility of a gas in a liquid increases with increasing partial pressure of the gas above the solution. This relationship is described by Henry's Law:
: Solubility of the gas
: Henry's law constant
: Partial pressure of the gas

Concentration Units and Colligative Properties
Concentration Units
Concentration describes the relative amounts of solute and solvent in a solution. Common units include:
Molarity (M): Moles of solute per liter of solution
Molality (m): Moles of solute per kilogram of solvent
Mass percent: (Mass of solute / Mass of solution) × 100%
Parts per million (ppm):
Parts per billion (ppb):
Colligative Properties
Colligative properties depend only on the number of solute particles, not their identity. Important colligative properties include:
Vapor pressure lowering
Boiling point elevation
Freezing point depression
Osmotic pressure
Vapor Pressure Lowering: Raoult's Law
The vapor pressure of a solvent above a solution is lower than that of the pure solvent. Raoult's Law describes this relationship:
: Mole fraction of solvent
: Vapor pressure of pure solvent
Boiling Point Elevation and Freezing Point Depression
The boiling point of a solution is higher, and the freezing point is lower, than those of the pure solvent. The changes are proportional to the molal concentration of solute particles:
: van't Hoff factor (number of particles per formula unit)
, : Boiling and freezing point constants
Osmosis and Osmotic Pressure
Osmosis is the flow of solvent from a solution of low concentration to one of high concentration through a semipermeable membrane. The osmotic pressure () is given by:
: Molarity of solute particles
: Gas constant ()
: Temperature in Kelvin
Colloids and the Tyndall Effect
Colloidal Dispersions
Colloids are suspensions of particles larger than individual ions or molecules, but too small to settle out by gravity. They exhibit unique properties such as Brownian motion and the Tyndall effect (scattering of light).
Brownian motion: Random movement due to collisions with solvent molecules.
Tyndall effect: Colloid particles scatter light, making colloids appear cloudy or opaque.
Example: Soap forms colloids due to its hydrophilic head and hydrophobic tail, enabling micelle formation and emulsification.
Additional info: All explanations have been expanded for clarity and completeness, and tables have been recreated for study purposes.
