 Back
BackChapter 15: Chemical Equilibrium – Mini-Textbook Study Guide
Study Guide - Smart Notes

Chemical Equilibrium
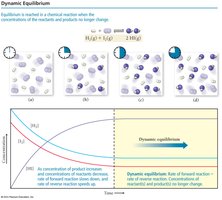
Dynamic Equilibrium
Chemical equilibrium occurs when the rate of the forward reaction equals the rate of the reverse reaction, resulting in constant concentrations of reactants and products. This state is dynamic, meaning reactions continue to occur in both directions, but there is no net change in concentrations.
Key Point 1: At equilibrium, the concentrations of reactants and products remain constant over time, but are not necessarily equal.
Key Point 2: Dynamic equilibrium is achieved when the rates of the forward and reverse reactions are equal.
Example: In the reaction H2(g) + I2(g) ⇄ 2 HI(g), equilibrium is reached when the rate of HI formation equals the rate of HI decomposition.

The Equilibrium Constant (K)
The Law of Mass Action
The equilibrium constant, K, quantifies the ratio of product and reactant concentrations at equilibrium for a given reaction. It is derived from the balanced chemical equation and is unitless.
Key Point 1: For a general reaction: aA + bB ⇄ cC + dD, the equilibrium constant expression is:
Key Point 2: The value of K indicates whether a reaction is product-favored (K >> 1) or reactant-favored (K << 1).
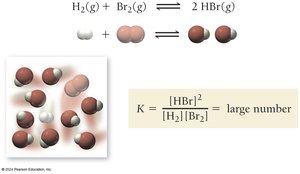
Example: For H2(g) + Br2(g) ⇄ 2 HBr(g), a large K means products predominate at equilibrium.
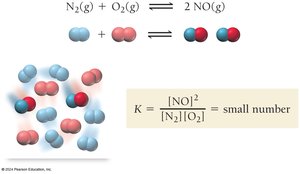
Example: For N2(g) + O2(g) ⇄ 2 NO(g), a small K means reactants predominate at equilibrium.
Manipulating Equilibrium Constants
Reversing and Scaling Chemical Equations
Changes to the chemical equation affect the equilibrium constant:
Key Point 1: Reversing the equation inverts the equilibrium constant:
Key Point 2: Multiplying the equation by a factor n raises K to the nth power:
Key Point 3: Adding reactions multiplies their equilibrium constants:
Equilibrium Constants in Gas Phase Reactions
KP vs. KC
For gas phase reactions, equilibrium constants can be expressed in terms of partial pressures (KP) or concentrations (KC). The relationship is:
Key Point 1: R is the gas constant (0.0821 L·atm/mol·K), T is temperature in Kelvin, and Δn is the change in moles of gas.
Key Point 2: Solids and pure liquids are not included in equilibrium expressions because their activity is 1.
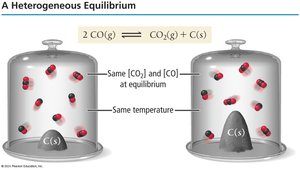
Heterogeneous Equilibria
Equilibria Involving Solids and Liquids
In reactions involving solids or pure liquids, only the concentrations of gases and aqueous species appear in the equilibrium expression.
Key Point 1: The activity of solids and pure liquids is defined as 1.
Example: For 2 CO(g) ⇄ CO2(g) + C(s), the equilibrium expression is
Calculating Equilibrium Constants and Concentrations
ICE Tables and Equilibrium Calculations
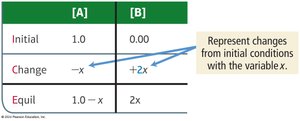
Equilibrium calculations often use ICE tables (Initial, Change, Equilibrium) to organize data and solve for unknowns.
Key Point 1: ICE tables help track changes in concentrations or pressures as a reaction approaches equilibrium.
Key Point 2: Quadratic equations may be required when solving for equilibrium concentrations.
Example: For A(g) ⇄ 2 B(g), if initial [A] = 1.0 M and [B] = 0, the changes are –x for A and +2x for B.
The Reaction Quotient (Q) and Predicting Direction
Comparing Q and K
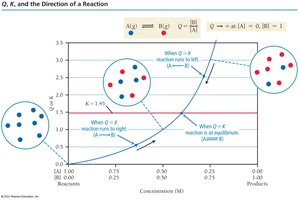
The reaction quotient, Q, is calculated the same way as K but uses current concentrations. Comparing Q to K predicts the direction the reaction will shift to reach equilibrium.
Key Point 1: If Q < K, the reaction shifts right (toward products).
Key Point 2: If Q > K, the reaction shifts left (toward reactants).
Key Point 3: If Q = K, the system is at equilibrium.
Example: For A ⇄ B, if Q < K, more B will form until equilibrium is reached.
Le Châtelier’s Principle
Response to Disturbances
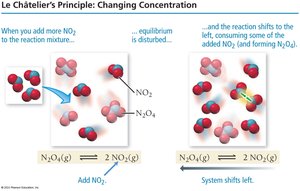
Le Châtelier’s Principle states that a system at equilibrium will shift to counteract any disturbance (change in concentration, pressure, or temperature).
Key Point 1: Adding reactant shifts equilibrium toward products; adding product shifts toward reactants.
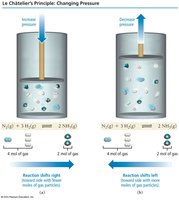
Key Point 2: Increasing pressure (for gases) shifts equilibrium toward the side with fewer moles of gas.
Key Point 3: Increasing temperature shifts equilibrium away from the side with heat (exothermic or endothermic).
Example: For N2O4(g) ⇄ 2 NO2(g), adding NO2 shifts equilibrium left, forming more N2O4.
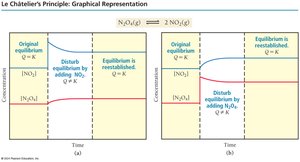

Example: For N2(g) + 3 H2(g) ⇄ 2 NH3(g), adding H2 shifts equilibrium right, increasing NH3 production.
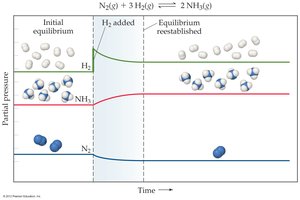
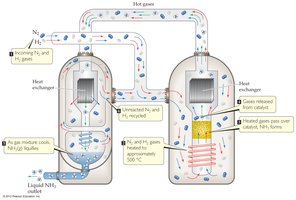
Example: The Haber-Bosch process uses Le Châtelier’s Principle to maximize ammonia yield by manipulating pressure and temperature.
Example: Increasing pressure shifts equilibrium toward fewer gas particles; decreasing pressure shifts toward more gas particles.
Example: For N2O4(g) ⇄ 2 NO2(g), increasing temperature shifts equilibrium right (endothermic reaction).

Example: For Co(H2O)62+(aq) + 4 Cl–(aq) ⇄ CoCl42–(aq) + 6 H2O(l), increasing temperature shifts equilibrium toward the blue product.


Effect of a Catalyst on Equilibrium
Catalysts and Equilibrium Position
Catalysts increase the rate at which equilibrium is reached by speeding up both the forward and reverse reactions equally. They do not affect the equilibrium position or the concentrations of reactants and products at equilibrium.
Key Point 1: Catalysts do not change the value of K or the equilibrium concentrations.
Key Point 2: The system reaches equilibrium faster with a catalyst.
Summary Table: Effects on Equilibrium
Disturbance | Equilibrium Shift | Effect |
|---|---|---|
Add reactant | Right (products) | More product formed |
Add product | Left (reactants) | More reactant formed |
Increase pressure (gas) | Side with fewer moles | Reduces pressure |
Increase temperature (exothermic) | Left (reactants) | Reduces product |
Increase temperature (endothermic) | Right (products) | Increases product |
Add catalyst | No shift | Equilibrium reached faster |
Additional info: This guide covers all major aspects of chemical equilibrium as outlined in Chapter 15, including equilibrium constants, ICE tables, Le Châtelier’s Principle, and the effect of catalysts. Examples and images are provided to reinforce key concepts and applications.
