 Back
BackChapter 16: Chemical Equilibrium – Principles, Calculations, and Applications
Study Guide - Smart Notes

Chemical Equilibrium
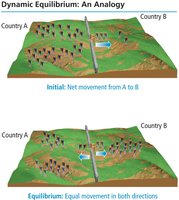
Dynamic Reactions and Equilibrium
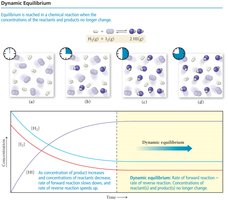
Chemical equilibrium occurs when the rates of the forward and reverse reactions become equal, resulting in no net change in the concentrations of reactants and products. This state is dynamic, meaning that reactions continue to occur, but the overall concentrations remain constant.
Reactant Concentration: Decreases as the reaction proceeds forward.
Product Concentration: Increases as products are formed.
Forward Reaction Rate: Decreases as reactants are consumed.
Reverse Reaction Rate: Increases as products accumulate and can react to reform reactants.
Dynamic Equilibrium: The point at which the rates of the forward and reverse reactions are equal, and concentrations of all species remain constant.
Characteristics of Equilibrium
At equilibrium, the system is not static but dynamic. The rates of the forward and reverse reactions are equal, but the concentrations of reactants and products are not necessarily equal. The position of equilibrium depends on the reaction and conditions.
"Favors Products": Most reactants are converted to products; product concentration is high.
"Favors Reactants": Most reactants remain; product concentration is low.
The Equilibrium Constant (K)
Law of Mass Action
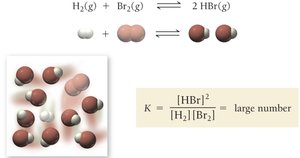
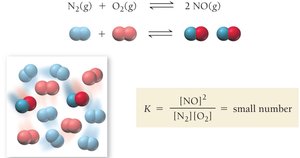
The equilibrium constant, K, expresses the relationship between the concentrations of reactants and products at equilibrium for a given chemical equation. It is a unitless value that provides insight into the position of equilibrium.
General Form: For a reaction , the equilibrium constant is:
K > 1: Products are favored at equilibrium.
K < 1: Reactants are favored at equilibrium.
Manipulating Equilibrium Constants
Reversing the Equation: Invert K ().
Multiplying Coefficients: Raise K to the power of the factor ( if coefficients are multiplied by n).
Adding Equations: Multiply the K values of the individual reactions to get the overall K.
Equilibrium with Gases: KP
For reactions involving gases, the equilibrium constant can be expressed in terms of partial pressures (KP), always using atmospheres (atm) as the unit for pressure.
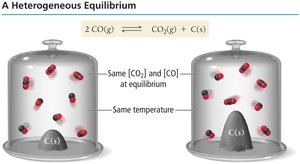
Heterogeneous Equilibria
In reactions involving solids and liquids, their concentrations do not change and are therefore omitted from the equilibrium expression. Only aqueous (aq) and gaseous (g) species are included in K.
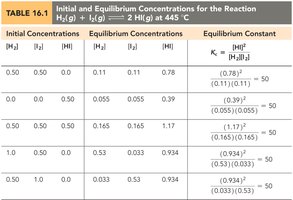
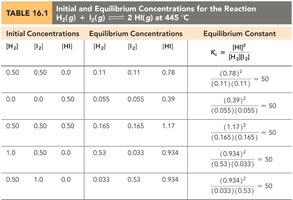
Calculating the Equilibrium Constant
Using Equilibrium Concentrations
To calculate K, use the equilibrium concentrations of all species involved in the reaction. Stoichiometry and ICE (Initial, Change, Equilibrium) tables are often used to determine these values.
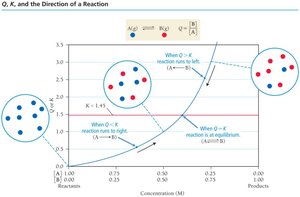
Reaction Quotient (Q) and Predicting Direction
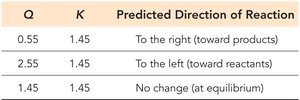
Reaction Quotient (Q)
The reaction quotient, Q, is calculated using the same expression as K but with current (not necessarily equilibrium) concentrations. Comparing Q to K predicts the direction the reaction will proceed to reach equilibrium.
Q < K: Reaction proceeds to the right (toward products).
Q > K: Reaction proceeds to the left (toward reactants).
Q = K: System is at equilibrium; no net change.
Finding Equilibrium Concentrations
Types of Problems
Given K and all but one equilibrium concentration: Solve for the unknown concentration.
Given K and only initial concentrations: Use ICE tables and solve for equilibrium concentrations, often requiring algebraic manipulation.
When K is very small (K << 1), the change in reactant concentration may be negligible, allowing for approximation methods if the change is less than 5% of the initial value.
Le Châtelier’s Principle
Predicting the Effect of Disturbances
Le Châtelier’s Principle states that if a system at equilibrium is disturbed, it will shift in the direction that minimizes the disturbance. Disturbances include changes in concentration, pressure/volume, and temperature.
Adding Reactant: Shifts equilibrium toward products.
Removing Reactant: Shifts equilibrium toward reactants.
Adding Product: Shifts equilibrium toward reactants.
Removing Product: Shifts equilibrium toward products.
Changing Pressure and Volume (for Gases)
Increase Pressure (decrease volume): Shifts equilibrium toward the side with fewer moles of gas.
Decrease Pressure (increase volume): Shifts equilibrium toward the side with more moles of gas.


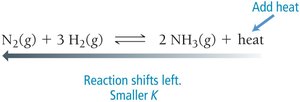
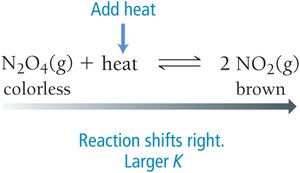
Changing Temperature
Exothermic Reaction (releases heat): Increasing temperature shifts equilibrium toward reactants (K decreases).
Endothermic Reaction (absorbs heat): Increasing temperature shifts equilibrium toward products (K increases).
Summary Table: Le Châtelier’s Principle
Disturbance | System Response |
|---|---|
Increase [Reactant] | Shifts right (toward products) |
Increase [Product] | Shifts left (toward reactants) |
Decrease [Reactant] | Shifts left (toward reactants) |
Decrease [Product] | Shifts right (toward products) |
Increase Pressure | Shifts toward fewer moles of gas |
Decrease Pressure | Shifts toward more moles of gas |
Increase Temperature (exothermic) | Shifts left, K decreases |
Increase Temperature (endothermic) | Shifts right, K increases |
