 Back
BackChapter 16: Chemical Equilibrium – Principles, Calculations, and Applications
Study Guide - Smart Notes

Chapter 16: Chemical Equilibrium
Introduction to Chemical Equilibrium
Chemical equilibrium is a fundamental concept in chemistry describing the state in which the concentrations of reactants and products remain constant over time, as the forward and reverse reactions occur at equal rates. Understanding equilibrium is essential for predicting the composition of reaction mixtures and for manipulating chemical processes in laboratory and industrial settings.
The Concept of Dynamic Equilibrium
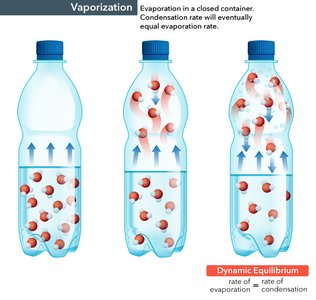
At equilibrium, the reaction does not stop; instead, the forward and reverse reactions proceed at the same rate, resulting in no net change in the concentrations of reactants and products. This state is called dynamic equilibrium because the molecular processes continue, but the macroscopic properties remain constant.
Dynamic Process: Both the forward and reverse reactions are ongoing.
Constant Concentrations: The amounts of reactants and products do not change once equilibrium is reached.
Example: The vaporization and condensation of water in a closed container eventually reach a state where the rate of evaporation equals the rate of condensation.
Describing Equilibrium: The Equilibrium Constant (K)
The equilibrium constant (K) quantitatively expresses the relationship between the concentrations of products and reactants at equilibrium for a given reaction at a specific temperature.
General Form: For the reaction , the equilibrium constant in terms of concentration (Kc) is:
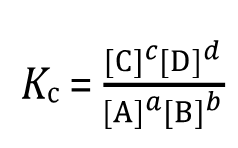
Units: K is usually dimensionless, but the units depend on the reaction stoichiometry.
Significance: The value of K indicates the extent to which a reaction proceeds:
K > 1: Product-favored equilibrium (more products than reactants).
K < 1: Reactant-favored equilibrium (more reactants than products).
K ≈ 1: Comparable amounts of reactants and products.
Manipulating the Equilibrium Constant
The value of K changes in predictable ways when the chemical equation is manipulated:
Multiplying the Equation: If the equation is multiplied by n, the new equilibrium constant is .
Reversing the Equation: The equilibrium constant for the reverse reaction is the reciprocal, .
Combining Equations: When adding equations, multiply their K values to get the overall K.
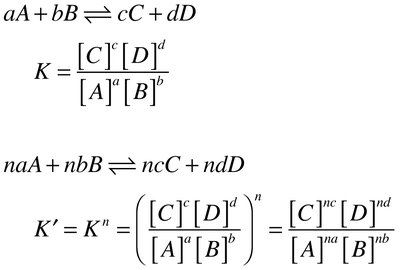
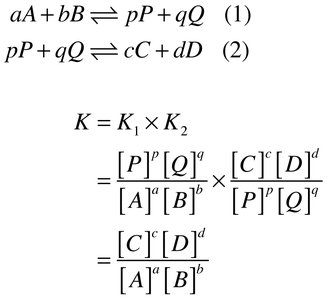
Equilibrium Constants for Gaseous Reactions: Kp
For reactions involving gases, the equilibrium constant can also be expressed in terms of partial pressures (Kp):
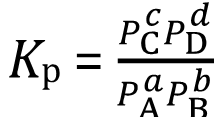
Relationship between Kc and Kp: , where Δn is the change in moles of gas (products minus reactants), R is the gas constant, and T is temperature in Kelvin.
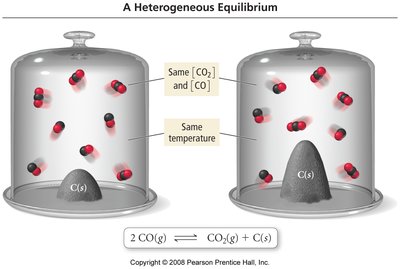
Heterogeneous Equilibria
When pure solids or liquids are involved in equilibrium, their concentrations are considered constant and are omitted from the equilibrium expression. Only aqueous and gaseous species are included.
Example: For , the equilibrium constant is (C(s) is omitted).
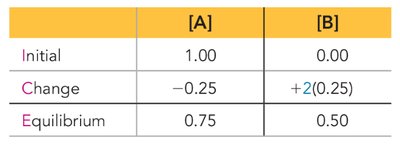
ICE Tables: Calculating Equilibrium Concentrations
ICE tables (Initial, Change, Equilibrium) are systematic tools for solving equilibrium problems. They help track the changes in concentrations or pressures as a reaction approaches equilibrium.
Steps:
List initial concentrations (I).
Define changes in concentrations (C) using variables (e.g., x).
Express equilibrium concentrations (E) in terms of initial values and changes.
Substitute equilibrium values into the K expression and solve for x.
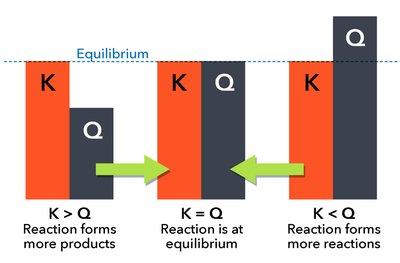
The Reaction Quotient (Q) and Predicting Direction of Change
The reaction quotient (Q) is calculated using the same form as K, but with current (not necessarily equilibrium) concentrations or pressures. Comparing Q to K predicts the direction the reaction will proceed to reach equilibrium:
Q < K: Reaction proceeds forward (toward products).
Q > K: Reaction proceeds in reverse (toward reactants).
Q = K: System is at equilibrium.
Le Chatêlier’s Principle: Response to Disturbance
Le Chatêlier’s Principle states that if a system at equilibrium is disturbed, it will shift in the direction that counteracts the disturbance and restores equilibrium. Common disturbances include changes in concentration, pressure, volume, and temperature.
Change in Concentration: Adding reactant shifts equilibrium toward products; removing reactant shifts toward reactants.
Change in Pressure/Volume (gases): Increasing pressure (decreasing volume) shifts equilibrium toward the side with fewer gas molecules; decreasing pressure (increasing volume) shifts toward more gas molecules.
Change in Temperature: For exothermic reactions, increasing temperature shifts equilibrium left (toward reactants); for endothermic reactions, increasing temperature shifts right (toward products).

Worked Examples and Practice Problems
Example 1: Calculating equilibrium concentrations for acetic acid dissociation using an ICE table and the quadratic formula.
Example 2: Predicting the effect of adding or removing substances, changing pressure, or changing temperature on the position of equilibrium.
Summary Table: Key Equilibrium Concepts
Concept | Description | Key Equation |
|---|---|---|
Dynamic Equilibrium | Forward and reverse reactions occur at equal rates | — |
Equilibrium Constant (Kc) | Ratio of product to reactant concentrations at equilibrium | |
Equilibrium Constant (Kp) | Ratio of product to reactant partial pressures at equilibrium | |
Reaction Quotient (Q) | Ratio of current product to reactant concentrations | |
Le Chatêlier’s Principle | System shifts to counteract disturbances | — |
Additional info: The notes include practice problems and worked examples to reinforce the application of equilibrium concepts, including the use of ICE tables, the calculation of Kc and Kp, and the prediction of equilibrium shifts using Le Chatêlier’s Principle.
