 Back
BackChapter 16 Study Guide
Study Guide - Smart Notes

Acids and Bases
Definitions and Properties
Acids and bases are fundamental chemical species with distinct properties and behaviors in aqueous solutions. Acids typically taste sour, turn blue litmus paper red, and increase the concentration of hydrogen ions ([H+]) in solution. Bases taste bitter, feel slippery, turn red litmus paper blue, and increase the concentration of hydroxide ions ([OH-]).
Arrhenius Definition: Acids produce H+ in water; bases produce OH- in water.
Neutralization Reaction: Acid + Base → Salt + Water (e.g., HCl + NaOH → NaCl + H2O).
Brønsted-Lowry Acids and Bases
Proton Transfer Concept
The Brønsted-Lowry theory broadens the definition of acids and bases:
Acid: Proton (H+) donor.
Base: Proton (H+) acceptor.
Amphoteric: A substance that can act as either an acid or a base (e.g., water).
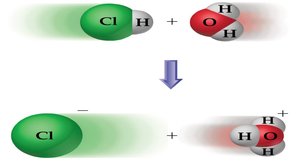
In aqueous solution, the H+ ion is often associated with water molecules, forming hydronium ions (H3O+), and even larger clusters such as H5O2+ and H9O4+.
Conjugate Acid-Base Pairs
Definition and Examples
When an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid. Conjugate acid-base pairs differ by one proton.
Example: HA + H2O ⇌ A- + H3O+
HA and A- are a conjugate acid-base pair.
H2O and H3O+ are a conjugate acid-base pair.
Relative Strengths of Acids and Bases
Strength and Conjugate Relationships
The strength of an acid or base is inversely related to the strength of its conjugate. The stronger the acid, the weaker its conjugate base, and vice versa. In aqueous solution, H3O+ is the strongest acid and OH- is the strongest base that can exist in equilibrium.
Review of Strong Electrolytes
Strong vs. Weak Acids and Bases
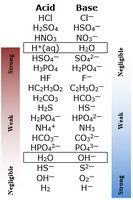
Strong electrolytes (such as strong acids, strong bases, and most salts) dissociate completely in water. Weak acids and bases only partially ionize, often less than 10%.
Strong acids: HCl, HNO3, H2SO4
Strong bases: NaOH, KOH
Weak acids: CH3COOH, HCN
Weak bases: NH3
The Autoionization of Water
Equilibrium and the Ion Product
Water undergoes slight autoionization:
At 25°C,
The equilibrium constant is at 25°C.
The pH and pOH Scales
Definitions and Calculations
The pH and pOH scales provide a convenient way to express acidity and basicity:
At 25°C,
![pH, pOH, [H+], and [OH-] relationship chart](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/26972da0_image_1.png)
In neutral water at 25°C, pH = 7.00. Acidic solutions have pH < 7, and basic solutions have pH > 7.
Ionization Constants for Weak Monoprotic Acids and Bases
Equilibrium Expressions
Weak acids and bases establish equilibrium in water. The acid ionization constant () and base ionization constant () quantify their strengths:
For a weak acid, HA:
For a weak base, B:
Polyprotic Acids
Stepwise Ionization
Polyprotic acids have more than one ionizable hydrogen. Each ionization step has its own , and , etc. For example, phosphoric acid (H3PO4):
Relationship Between Ka and Kb
Conjugate Acid-Base Pairs
For a conjugate acid-base pair:
at 25°C
The stronger the acid (larger ), the weaker its conjugate base (smaller ).
pH of Salt Solutions
Acidic, Basic, and Neutral Salts
The pH of a salt solution depends on the strengths of the acid and base from which the salt is derived:
Strong acid + strong base: Neutral solution (e.g., NaCl).
Strong base + weak acid: Basic solution (e.g., NaC2H3O2).
Weak base + strong acid: Acidic solution (e.g., NH4Cl).
Weak base + weak acid: pH depends on relative and values.
Carboxylic Acids
Structure and Properties
Carboxylic acids are organic acids containing the carboxyl group (COOH). They are generally weak acids. Acetic acid (CH3COOH) is a common example.
Lewis Acids and Bases
Electron Pair Concept
The Lewis definition is the most general:
Lewis acid: Electron pair acceptor (e.g., BF3, metal ions).
Lewis base: Electron pair donor (e.g., NH3, OH-).
Lewis acids often have incomplete octets or vacant orbitals, while Lewis bases have lone pairs available for bonding.
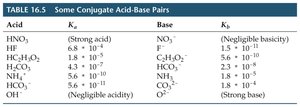
Summary Table: Relative Strengths of Acids and Bases
The following table summarizes the relative strengths of selected acids and bases, as well as their conjugate pairs:
Acid | Ka | Base | Kb |
|---|---|---|---|
HNO3 | Strong acid | NO3- | Negligible basicity |
HF | 6.8 × 10-4 | F- | 1.5 × 10-11 |
HC2H3O2 | 1.8 × 10-5 | C2H3O2- | 5.6 × 10-10 |
HCO3- | 4.3 × 10-7 | CO32- | 1.8 × 10-4 |
NH4+ | 5.6 × 10-10 | NH3 | 1.8 × 10-5 |
HCO3- | 4.3 × 10-7 | CO32- | 1.8 × 10-4 |
OH- | Negligible acidity | Strong base |

Summary Table: Strengths of Acids and Bases
Acid | Base |
|---|---|
HCl, HNO3, H2SO4 | Cl-, NO3-, HSO4- |
H3PO4, HF, CH3COOH | H2PO4-, F-, CH3COO- |
HCO3-, H2O | CO32-, OH- |
H2 | H- |

Additional info: This guide covers all major aspects of acid-base equilibria, including definitions, calculations, and conceptual relationships, suitable for college-level general chemistry.
