 Back
BackChapter 17: Acids and Bases – General Chemistry II Study Notes
Study Guide - Smart Notes

Acids and Bases
Definitions of Acids and Bases
Understanding acids and bases is fundamental in chemistry. There are three main definitions: Arrhenius, Brønsted-Lowry, and Lewis. Each definition broadens the scope of what can be considered an acid or a base.
Arrhenius Acids and Bases: An Arrhenius acid produces H+ ions in water, while an Arrhenius base produces OH− ions in water.
Brønsted-Lowry Acids and Bases: A Brønsted-Lowry acid is a proton (H+) donor, and a Brønsted-Lowry base is a proton acceptor. Acid-base reactions involve the transfer of a proton from the acid to the base.
Lewis Acids and Bases: A Lewis acid is an electron pair acceptor, and a Lewis base is an electron pair donor. This definition includes more substances, such as metal ions, as acids.
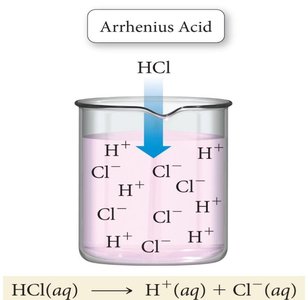
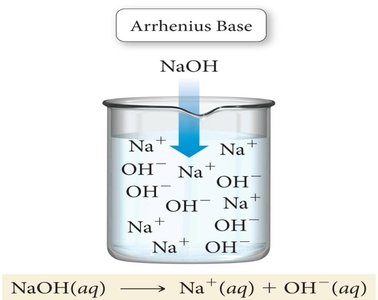
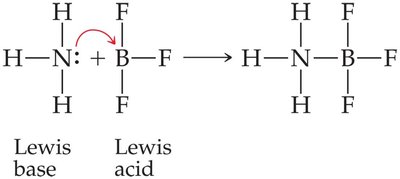
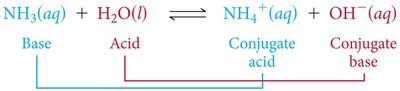
Conjugate Acid-Base Pairs
In Brønsted-Lowry theory, acids and bases always occur in pairs. When an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid.
Every acid has a conjugate base, and every base has a conjugate acid.
Example: NH3 + H2O ⇌ NH4+ + OH−

Water as a Solvent
Auto-Ionization of Water
Water is an amphoteric substance, meaning it can act as both an acid and a base. In pure water, two water molecules react to form hydronium and hydroxide ions:
H2O(l) + H2O(l) ⇌ H3O+(aq) + OH−(aq)
Or simply: H2O(l) ⇌ H+(aq) + OH−(aq)
Ion-Product Constant of Water, Kw
The equilibrium constant for the auto-ionization of water is called the ion-product constant, Kw:
At 25°C,
In neutral water, M$
pKw, pH, and pOH
The pH and pOH scales are logarithmic measures of hydronium and hydroxide ion concentrations, respectively:
at 25°C
at 25°C
pH Scale and Measurements
The pH Scale
The pH scale typically ranges from 0 (very acidic) to 14 (very basic), but values outside this range are possible. Each unit change in pH represents a tenfold change in [H+].
pH < 7: Acidic solution ()
pH = 7: Neutral solution ()
pH > 7: Basic solution ()


Calculating pH and pOH
To find [H3O+] from pH:
To find [OH−] from pOH:
To find pH from [OH−]: Calculate pOH, then use
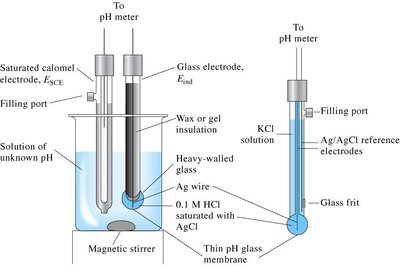
pH Measurement
The pH of a solution is most accurately measured using a pH meter, which uses a glass electrode sensitive to [H3O+]. Less accurate methods include pH paper and indicators.


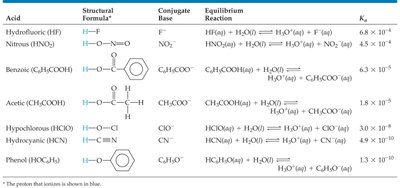
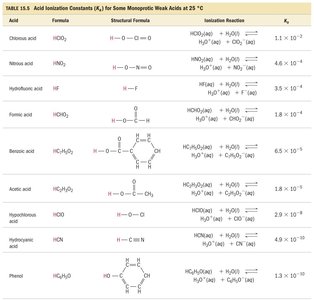
Weak Acids: Acid Dissociation Constant (Ka), % Ionization, and pH Calculations
Strong vs. Weak Acids
Strong acids ionize completely in water, while weak acids only partially ionize. The concentration of H+ in a strong acid solution equals the acid's molarity. For weak acids, equilibrium calculations are required.
Strong acids to memorize: HCl, HBr, HI, HNO3, HClO4, HClO3, H2SO4 (first proton only)
Acid Dissociation Constant (Ka)
The strength of a weak acid is quantified by its acid dissociation constant, Ka:
For HA(aq) + H2O(l) ⇌ H3O+(aq) + A−(aq):
The larger the Ka, the stronger the acid.
pKa is defined as ; smaller pKa means a stronger acid.

% Ionization of a Weak Acid
The percent ionization of a weak acid is the fraction of acid molecules that ionize in solution, expressed as a percentage:
As the acid is diluted, % ionization increases.

Calculating pH of a Weak Acid Solution
To calculate the pH of a weak acid solution, set up an ICE (Initial, Change, Equilibrium) table and solve for [H3O+] using Ka. If % ionization is less than 5%, the change in concentration (x) can be neglected in the denominator for simplification.
Example: For 0.15 M acetic acid (Ka = 1.8 × 10−5):
Set up: , solve for x, then
If % ionization ≤ 5%, the approximation is valid.
Concentration | [HC2H3O2], M | [H3O+], M | [C2H3O2−], M |
|---|---|---|---|
Initial | 0.15 | 0 | 0 |
Change | −x | +x | +x |
Equilibrium | 0.15−x | x | x |
Additional info: The ICE table method is a standard approach for equilibrium calculations in weak acid and base chemistry.
