 Back
BackChapter 17: Acids and Bases – Properties, Definitions, and Quantitative Relationships
Study Guide - Smart Notes

Acids and Bases: Properties and Definitions
Common Properties of Acids and Bases
Acids and bases are two fundamental categories of compounds in chemistry, each with distinct physical and chemical properties.
Acids:
Sour taste
Can dissolve metals
Turns litmus paper red
Neutralizes bases
Bases:
Bitter taste
Feel slippery
Turns litmus paper blue
Neutralizes acids
Definitions of Acids and Bases
There are several definitions for acids and bases, each with its own scope and application:
Arrhenius Definition:
An acid produces H+ ions in aqueous solution.
A base produces OH– ions in aqueous solution.
Brønsted–Lowry Definition:
An acid donates an H+ ion in a chemical reaction.
A base accepts an H+ ion in a chemical reaction.
This definition is more broadly applicable than the Arrhenius definition.
Additional info: The Lewis definition (not covered in detail here) further expands the concept by defining acids as electron pair acceptors and bases as electron pair donors.
Identifying Acids and Bases in Reactions
To identify acids and bases in a reaction, look for the species donating and accepting H+:
Example: CH3COOH(aq) + H2O(l) → CH3COO–(aq) + H3O+(aq)
CH3COOH: Acid (donates H+)
H2O: Base (accepts H+)
The Hydronium Ion and Acid Dissociation
The Hydronium Ion (H3O+)
In aqueous solutions, free H+ ions do not exist independently. Instead, they bond to water molecules to form the hydronium ion:
H+ + H2O → H3O+
H3O+ is the actual species present in solution.
Acid Ionization Constant (Ka)
The strength of an acid is quantified by its acid ionization constant, Ka:
For a generic acid HA: HA(aq) + H2O(l) ↔ A–(aq) + H3O+(aq)
The equilibrium expression is:
A larger Ka value indicates a stronger acid (more products at equilibrium).
Strong vs. Weak Acids
Strong acids dissociate completely in water; the reaction goes to completion and is not an equilibrium.
Weak acids only partially dissociate; the reaction is an equilibrium and Ka is used to describe their strength.
Conjugate Acid-Base Pairs
Definition and Identification
Every acid-base reaction involves two conjugate pairs:
When an acid donates a proton, it forms its conjugate base.
When a base accepts a proton, it forms its conjugate acid.
Example: CH3COOH(aq) + H2O(l) ↔ CH3COO–(aq) + H3O+(aq)
CH3COOH / CH3COO–: Acid / Conjugate Base
H2O / H3O+: Base / Conjugate Acid
The weaker the acid, the stronger its conjugate base.
Autoionization of Water and the Ion-Product Constant (Kw)
Autoionization of Water
Water can act as both an acid and a base (amphoteric). When two water molecules react:
2 H2O(l) ↔ H3O+(aq) + OH–(aq)
The equilibrium constant for this reaction is called the ion-product constant for water (Kw):
At 25°C, Kw = 1.0 × 10–14
Acidic, Basic, and Neutral Solutions
If [H3O+] > [OH–]: Acidic
If [H3O+] < [OH–]: Basic
If [H3O+] = [OH–]: Neutral
In pure water at 25°C: [H3O+] = [OH–] = 1.0 × 10–7 M
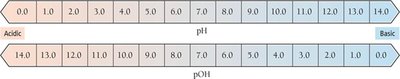
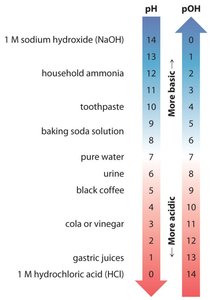
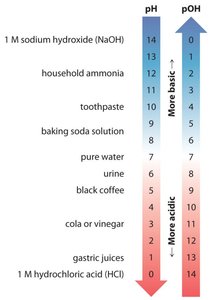
The pH and pOH Scales
The pH Scale
The pH scale is a logarithmic scale used to express the concentration of hydronium ions in solution:
At 25°C:
pH < 7.0: Acidic
pH = 7.0: Neutral
pH > 7.0: Basic
Calculating [H3O+] from pH and Vice Versa
To find [H3O+] from pH:
To find pH from [H3O+]:
The pOH Scale
The pOH scale is defined similarly to pH, but for hydroxide ion concentration:
Relationship Between pH and pOH
At 25°C, the sum of pH and pOH is always 14.00:
pKa and pKb
Definitions and Calculations
pKa is the negative logarithm of the acid dissociation constant:
Similarly, for bases:
Base Strength and the Relationship Between Ka and Kb
Base Ionization Constant (Kb)
For a generic base B: B(aq) + H2O(l) ↔ BH+(aq) + OH–(aq)
The equilibrium expression is:
A larger Kb value indicates a stronger base.
Relationship Between Ka and Kb
For a conjugate acid-base pair:
At 25°C,
If you know Ka, you can find Kb for the conjugate base, and vice versa.
Percent Ionization of Weak Acids
Definition and Calculation
The percent ionization of a weak acid quantifies the fraction of acid molecules that ionize in solution:
Example: For a weak acid with Ka = 4.0 × 10–8 and [HA]in = 0.600 M, [H3O+]eq = 1.5 × 10–4 M:
Effect of Acid Concentration on Percent Ionization
As the concentration of a weak acid increases, the percent ionization decreases.
Calculating pH from Ka
Steps for Calculating pH of a Weak Acid Solution
Write the balanced chemical equation for the acid dissociation.
Write the Ka expression.
Set up an ICE (Initial, Change, Equilibrium) table.
Solve for [H3O+] using the Ka expression.
Calculate pH from [H3O+].
Example: For HNO2 with Ka = 4.6 × 10–4 and [HNO2]initial = 0.200 M, pH = 2.02
Summary Table: Key Equilibrium Constants and Relationships
Constant | Definition | Formula |
|---|---|---|
Ka | Acid ionization constant | |
Kb | Base ionization constant | |
Kw | Ion-product constant for water | |
pH | Hydronium ion concentration | |
pOH | Hydroxide ion concentration | |
Relationship | pH and pOH at 25°C |
