 Back
BackChapter 17: Gibbs Energy and Thermodynamics – Spontaneous Processes and Entropy
Study Guide - Smart Notes

Spontaneous Processes
Definition and Characteristics
Spontaneous processes are fundamental to understanding chemical reactions and physical changes. A process is said to be spontaneous if it occurs without continuous outside intervention. The First Law of Thermodynamics states that energy is conserved, and is mathematically expressed as:
\Delta U: Change in internal energy of the system
q: Heat absorbed by the system or from the surroundings
w: Work done by the surroundings or by the system
However, determining whether a process is favored (spontaneous) cannot rely solely on \Delta U or \Delta H.

Features of Spontaneous Processes
A spontaneous process occurs without any continuous outside intervention.
If a process is spontaneous in one direction, it is not spontaneous in the opposite direction (e.g., dropping a stone is spontaneous, lifting it is not).
Spontaneity can depend on conditions such as temperature (e.g., ice melts spontaneously at T > 0°C, water freezes spontaneously at T < 0°C).

Examples of Spontaneous Processes
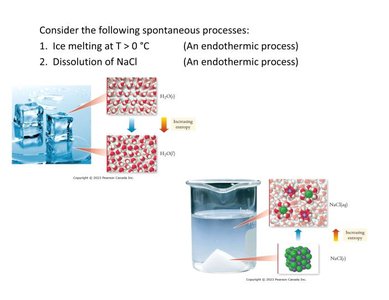
Ice melting at T > 0°C (endothermic process)
Dissolution of NaCl (endothermic process)
Both processes are spontaneous despite being endothermic, illustrating that spontaneity is not solely determined by energy changes.
Entropy and the Second Law of Thermodynamics
Entropy as a Criterion for Spontaneity
After extensive study, it has been concluded that all spontaneous processes share a common characteristic: an increase in entropy. Entropy is a property that quantifies the degree of randomness or disorder in a system.

Entropy: Definition and Probability
Entropy can be viewed as a measure of randomness or disorder. More precisely, it is a measure of the number of arrangements (positions and/or energy levels, called microstates) available to a system in a given state. Entropy is related to probability: nature tends to move toward states with higher probabilities of existing.
Entropy (S): a thermodynamic quantity that increases with the number of energetically equivalent ways to arrange the components of a system to achieve a particular state.

Energy Dispersal and Entropy
Boltzmann's Relation
Ludwig Boltzmann related entropy to the number of possible arrangements of molecules in a system. The Boltzmann equation is:
S: Entropy
k: Boltzmann constant
W: Number of microstates (possible arrangements)
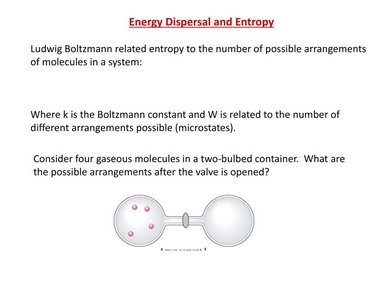
For example, consider four gaseous molecules in a two-bulbed container. The possible arrangements after the valve is opened illustrate the concept of microstates.
Spontaneous Expansion of Gases
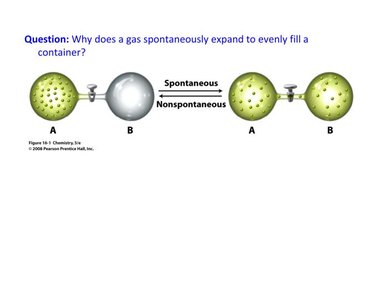
Gases spontaneously expand to evenly fill a container because this increases the number of possible microstates, thus increasing entropy.
Concept Checks: Microstates and Macrostates
Counting Microstates
For three molecules in a two-bulbed container, how many macrostates are possible?
For the macrostate where there are 2 molecules in the left bulb and one in the right bulb, how many microstates are possible?
These questions reinforce the relationship between microstates, macrostates, and entropy.

Factors Affecting Entropy
Energy Levels and Entropy
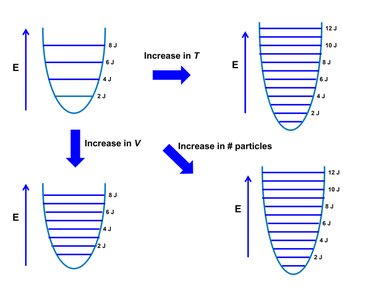
Entropy increases with:
Increase in temperature (T)
Increase in volume (V)
Increase in number of particles
These factors increase the number of accessible energy levels and microstates.
Atomic/Molecular View of Entropy
Microstates and Macrostates
An atom or molecule has a total amount of energy that can be dispersed in various ways (translational, kinetic, rotational, electronic, vibrational). A given set of conditions (P, V, T) defines the total energy (macrostate) of the atom/molecule. The different ways energy can be dispersed are called microstates. Only certain arrangements are allowed due to quantum mechanics.

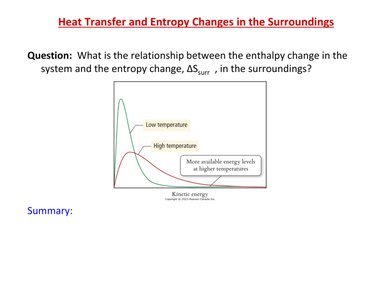
Heat Transfer and Entropy Changes in the Surroundings
Relationship Between Enthalpy and Entropy Change
The relationship between the enthalpy change in the system and the entropy change in the surroundings (\Delta S_{surr}) is important in thermodynamics. At higher temperatures, more energy levels are available, affecting the entropy change.
The Second Law of Thermodynamics
Statement and Implications
The Second Law of Thermodynamics states:
The total entropy of a system and its surroundings always increases for a spontaneous process.
The total change in entropy of the universe can be expressed as the sum of the change in entropy in the system and surroundings:

Temperature Dependence of Entropy Change in Surroundings
Magnitude of \Delta S_{surr}
The magnitude of the entropy change in the surroundings (\Delta S_{surr}) is affected by temperature. At higher temperatures, the same amount of heat transfer results in a smaller change in entropy.

Summary
Spontaneous processes are characterized by an increase in entropy.
Entropy measures the number of possible arrangements (microstates) in a system.
The Second Law of Thermodynamics states that the total entropy of the universe increases for spontaneous processes.
Factors such as temperature, volume, and number of particles affect entropy.
Heat transfer and entropy changes in the surroundings are temperature-dependent.
